-
Cancer is an abnormal proliferation of cells that spreads through uncontrolled cell division, evading apoptotic mechanisms, with the ability to invade tissues, causing disruptions in them and generating metastasis in healthy tissues. Today, cancer constitutes one of the greatest challenges of modern medicine[1,2]. According to recent World Health Organization (WHO) estimates, there are more than 19 million new cases of cancer and nearly 10 million deaths annually, making it the leading cause of global mortality. Despite therapeutic advances in recent decades, including precision surgery, combination chemotherapy, advanced radiotherapy, immunotherapy, tyrosine kinase inhibitors, and other types of cellular therapies, cancer treatment continues to face fundamental obstacles such as drug resistance, tumor recurrence, systemic toxicity, and limited cell specificity[1−3]. Faced with this scenario, there are substances that come from nature, such as plants, animals, or minerals, and have not been significantly altered by humans, called natural products, which have been a historical source of bioactive compounds. These are chemical substances with therapeutic properties. When these bioactive compounds are found in foods, they are called functional foods because, in addition to providing nutrients, they offer health benefits. There is evidence that many functional foods and/or their bioactive compounds have beneficial effects in oncology and are capable of intervening at multiple stages of tumor progression[2,4,5]. It is estimated that more than 60% of the antineoplastic drugs approved between 1981 and 2019 are derived directly or indirectly from natural compounds and are distinguished by their multitargeting capacity. These compounds include alkaloids, terpenoids, flavonoids, lignans, polyphenols, coumarins, saponins, and quinones, among others[6]. Although many current synthetic agents act on a single molecular target, natural compounds exert pleiotropic effects, simultaneously regulating multiple signaling pathways involved in proliferation, apoptosis, angiogenesis, invasion, metastasis, and immune evasion[5,7,8]. This multifunctional profile not only broadens its therapeutic spectrum but also counteracts the acquired resistance mechanisms that limit the effectiveness of many targeted treatments. One of the paradigmatic examples is that of the alkaloids derived from Catharanthus roseus (vinblastine and vincristine), widely used in oncology[9]. On the other hand, diets that include bioactive compounds that help prevent cancer have become increasingly important, driven by epidemiological and experimental evidence. Compounds such as curcumin, resveratrol, lycopene, genistein, epigallocatechin gallate (EGCG), capsaicin, and allicin, present in turmeric, grapes, tomatoes, soy, green tea, chili peppers, and garlic, respectively, have demonstrated, mainly in in vitro and in vivo animal models, antitumor activity by modulating critical pathways such as nuclear factor-kappa B (NF-κB), phosphatidylinositol 3-kinase/protein kinase B (PI3K/Akt), signal transducer and activator of transcription 3 (STAT3), mitogen-activated protein kinases (MAPK), and wingless and int-1 (Wnt)/β-catenin[7,10]. On the other hand, apitherapy (the use of bee-derived products) has generated interest as a complementary strategy. Propolis, royal jelly, pollen, and bee venom contain bioactive compounds such as caffeic acid phenethyl ester (CAPE), artepillin C (ARC), 10-hydroxydecenoic acid (10-HDA), melittin, and apamin, with antiproliferative, proapoptotic, antimetastatic, and immunomodulatory properties. Some of these products have shown synergy with chemotherapeutic agents and inhibitory effects on cancer stem cells[5]. It is worth mentioning that advances in biotechnology and synthetic biology have made it possible to overcome the barriers of natural production, developing microbial platforms for the synthesis of complex metabolites, optimizing biosynthetic pathways and improving yields through genetic engineering and metabolic editing[10−12]. However, the low natural abundance of bioactive compounds in plants, as well as the difficulties of their extraction, have motivated the development of advanced biotechnological strategies, including cell cultures, the use of hairy roots, elicitors, the search for endophytic fungi, and genetic engineering to streamline the production and/or extraction of these compounds[13−15]. Though the promise of natural bioactive compounds is substantial, their translation into clinical practice remains challenging. Issues include variability in the natural source materials, low yields, pharmacokinetic barriers, toxicity, regulatory approval, and limited rigorous clinical trials. Addressing these challenges through biotechnological improvements, standardization, and well-designed human studies is essential. This review critically integrates several findings, exploring the therapeutic potential of natural compounds and their biotechnological derivatives (compounds obtained by various techniques that can include genetic modification and overproduction by microorganisms), as well as their molecular mechanisms of action, some in vitro and in vivo studies, productive advances, challenges for clinical translation, and prospects in oncology.
-
The four co-authors conducted a search for articles in Google Scholar, PubMed, Web of Science, and Scopus databases using the keywords 'plant anticancer compounds', 'functional foods against cancer', 'natural products with anticancer activity', and 'vincristine and vinblastine'. Approximately 200 articles in English were retrieved. Regardless of their publication date, these articles included information on biomolecules and/or foods with anticancer activity, reporting tumor reduction or the promotion of cell apoptosis in malignant cases, as well as research on the effect of consuming nutraceutical foods with beneficial effects on cancer treatment. The articles also described the mechanisms of action of bioactive compounds. The articles were reviewed, analyzed, and classified into different categories according to the content structure of the manuscript, resulting in 64 articles. Most of these were published within the last 5 years, although some were older but were deemed necessary for inclusion because of their content. The articles considered report in vitro and in vivo studies with animal models and, in some cases, mention preclinical cases.
-
The research included in this review mentions the natural production of bioactive compounds present in plants, functional foods, and propolis, in which the presence of alkaloids is observed, such as homoharringtonine, colchicine, bisindoles, Vinca alkaloids such as vinblastine and vincristine, camptothecins, flavonoids, polyphenols, caffeic acid, among others. These studies evaluated their mechanisms of action, induction of apoptosis, regulatory mechanisms, and genes involved in synthesis. Table 1 shows some bioactive compounds against cancer.
Table 1. Some bioactive compounds with anticancer activity.
Compound Natural source Mechanism of action Ref. Vinblastine, vincristine Catharanthus roseus Blocks tubulin polymerization, stopping cell division.
Inhibition of microtubules, mitotic arrest, thus halting mitosis in metaphase and promoting apoptosis.[1,28] Paclitaxel, docetaxel Taxus brevifolia Microtubule stabilization, cell apoptosis. [30] Camptothecin Camptotheca acuminata Topoisomerase I inhibition, DNA damage. [4] Caffeic acid phenethyl ester (CAPE) Propolis Inhibition of NF-κB, caspases, angiogenesis. [5] Melittin Bee venom Cytolytic action, inhibition of signaling pathways such as NF-κB, and promoting apoptosis. [5,31] 10-Hydroxy-2-decenoic acid (10-HDA) Royal jelly Regulation of p53, Bax/Bcl-2, and caspases.
Inhibition of angiogenesis.[32] Curcumin Curcuma longa (turmeric) Reduces P-gp expression by inhibiting PI3K/Akt/NF-κB caspase activation.
Regulation of cyclins, CDKs and p21, causing arrest in the G1/S or G2M phase of the cell cycle.[7,13,19] Resveratrol Grapes, red wine and red fruits p53 activation, vascular endothelial growth factor (VEGF) suppression, induction of apoptosis.
Reverses multidrug resistance (MDR).
Inhibits the expression of MDR1 and P-gp at both the transcriptional and protein levels.[7,33,34] Genistein Soybean Inhibition of angiogenesis and tyrosine kinase.
Induction of apoptosis in hormone-dependent tumor cells.[2,5] Glucosinolates Cruciferous vegetables (Cabbage, broccoli) Histone deacetylase (HDAC) inhibition, apoptosis.
Inhibition of CDKs and tumor suppressor gene's expression; also modulates the expression of microRNAs and epigenetic enzymes such as DNA methyltransferases (DNMTs).[33,35] Epigallocatechin gallate (EGCG) Green tea Functional inhibition of P-gp, reduced expression of P-gp, substrate competition, or blockage of adenosine triphosphatase (ATPase) activity.
Inhibitor of proliferation and angiogenesis.
Modulates detoxifying enzymes such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), reducing oxidative damage and modulating cellular redox balance.[2,7,13,34,36] Quercetin Flavonoid (onion, broccoli, apple) Inhibition of key pathways such as PI3K/Akt/mTOR, NF-κB and STAT3, apoptosis by modulating reactive oxygen species metabolism. [37] Naringenin, apigenin, kaempferol, chrysin, luteolin, and quercetin Honey Inhibition of cell proliferation, stimulating apoptosis, blocking NF-κB and promoting cell cycle arrest. [5,38] Cinnamaldehyde Cinnamomum cassia (Chinese cinnamon) Apoptosis is limited to tumor cells and significantly decreases the activity of NF-κB and AP 1, as well as the expression of their target genes (Bcl-2, Bcl-xL). [39] Lycopene Solanum lycopersicum (tomato) Inhibition of the NF-κB pathway, activation of apoptosis, decreasing antiapoptotic proteins, and inhibition of cell proliferation. [2,40] Main mechanisms of action of bioactive compounds with anticancer activity
-
Some natural compounds exhibit a remarkable ability to modulate multiple signaling pathways involved in carcinogenesis, tumor progression, and the initiation of metastasis[1,4,16,17]. The main mechanisms are described below.
Induction of apoptosis
-
Apoptosis is a process of programmed cell death that eliminates damaged, aged, or unnecessary cells in a controlled manner and without causing inflammation. It occurs when internal signals (such as DNA damage activated by p53) or external signals (such as ligands binding to death receptors) trigger a caspase activation cascade, which degrades proteins and DNA. The cell shrinks, forms apoptotic bodies, and is finally eliminated by the macrophages[18,19]. The ability of tumor cells to evade apoptosis is considered one of the hallmarks of cancer, as dysregulation of the apoptotic pathways provides a survival advantage[20]. This ability allows cancer cells to develop multidrug resistance (MDR), a complex tumor phenotype that refers to concurrent resistance to agents with different functions or structures[21].
Cell cycle disruption
-
Several natural agents block the cell cycle at specific phases, inhibiting cell proliferation. Vincristine, for example, stops the cell cycle in the G2/M phase by interfering with the microtubules so the cell cannot initiate DNA replication; therefore, it does not enter the S phase, and this prevents it from duplicating and slows proliferation, whereas quercetin and curcumin regulate cyclins, cyclin-dependent kinase (CDKs), and p21, causing arrest in the G1/S or G2/M phase of the cell cycle[7,13,22]. With this blockage, the cell cannot initiate mitosis; it remains stopped with the DNA already replicated, which can lead to damage repair, senescence, or apoptosis if the damage persists[23].
Inhibition of angiogenesis
-
The formation of new blood vessels is essential for tumor growth and metastasis[7]. Various natural products have demonstrated a potent ability to inhibit tumor angiogenesis, a process that is essential for the growth and spread of cancer[24]. Compounds such as curcumin, resveratrol, EGCG from green tea, and certain saponins and flavonoids act by interfering with key pathways of angiogenesis (e.g., vascular endothelial growth factor [VEGF]/VEGFR), growth regulation, proliferation, survival and metabolism (e.g., PI3K/Akt), hypoxia regulation (e.g., hypoxia-inducible factor-1alpha [HIF-1α)], homeostasis (e.g., fibroblast growth factor [FGF]), and extracellular matrix remodeling (e.g., matrix metalloproteinases [MMPs]). They also interfere with the cellular communication system that regulates the expression of genes related to inflammation, the immune response, cell proliferation, and survival; one example is NF-κB, which blocks the proliferation, migration, and formation of new blood vessels by endothelial cells[25]. Furthermore, many of these compounds induce apoptosis in the tumor's vascular cells and reduce the expression of proangiogenic factors, limiting the supply of oxygen and nutrients to the tumor[7,25].
Epigenetic modulation and reduction of oxidative stress
-
Some natural compounds regulate DNA methylation, histone acetylation, and microRNA expression, restoring the expression of tumor suppressor genes and inhibiting oncogenes[5].
Natural compounds with antioxidant properties, such as polyphenols, terpenoids, and flavonoids, reduce oxidative stress and intervene in epigenetic modulation in cancer cells, counteracting alterations in DNA methylation and histone modifications caused by reactive oxygen species (ROS). They also act by inhibiting epigenetic enzymes such as DNA methyltransferases (DNMTs), histone deacetylases (HDACs), and histone methyltransferases (HMTs), thus restoring the expression of tumor suppressor genes and stopping tumors' progression[26]. On the other hand, antioxidant compounds, such as resveratrol, lycopene, and EGCG, modulate detoxifying enzymes such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), reducing oxidative damage and modulating the cellular redox balance[13].
Bioactive compounds with anticancer activity
-
In traditional medicine, plants, fungi, insects, lichens, algae, etc., have been used since ancient times as remedies or treatments for various diseases, and this empirical knowledge has been passed down across generations. The therapeutic action of many of these natural sources has been verified, the bioactive compounds responsible have been identified, their routes of action have been elucidated. Among the biological activities that have been identified in natural sources are antibacterial, antiviral, immunomodulatory, antidiabetic, hypolipidemic, antioxidant, and anticancer effects, among others. Specifically, among the phytochemicals with anticancer biological activity are the alkaloids, curcuminoids, flavonoids, and coumarins (Table 1).
Plant alkaloids represent one of the most successful classes of antineoplastic agents[1,3,16]. Derived from plants such as Catharanthus roseus, Colchicum autumnale, Taxus brevifolia, and Cephalotaxus harringtonia, these compounds act mainly on the microtubules' dynamics, interfering with cell division[27]. Colchicine, derived from Colchicum autumnale, also alters microtubules' stability and has shown anticancer activity in gastric and breast cancer models, activating caspase-dependent apoptotic pathways and modulating phosphoinositide 3-kinase/protein kinase B/mammalian target of rapamycin (PI3K/Akt/mTOR)[3]. Another relevant alkaloid is homoharringtonine, isolated from Cephalotaxus harringtonia, which inhibits the elongation of the polypeptide chain, interfering with protein synthesis and showing efficacy in leukemias that are resistant to tyrosine kinase inhibitors[2]. Bisindoles, including the Vinca alkaloids vinblastine and vincristine, which are naturally produced by Catharanthus roseus, block tubulin polymerization, arresting mitosis at the metaphase and inducing apoptosis[22,28,29]. Camptothecins, extracted from Camptotheca acuminata, inhibit Topoisomerase I, generating DNA breaks that culminate in apoptosis[5,6]. Vinblastine is used in Hodgkin lymphoma, testicular cancer, Kaposi's sarcoma, choriocarcinoma, breast cancer, and head and neck cancer, whereas vincristine is essential in acute lymphoblastic leukemia, Hodgkin and non-Hodgkin lymphoma, neuroblastoma, Wilms tumor, and rhabdomyosarcoma[2,3,9,27,28].
Flavonoids are polyphenolic compounds and constitute a diverse group of phytochemicals present in fruits, vegetables, legumes, green tea, and red wine[34]. Among the most studied flavonoids are quercetin, kaempferol, luteolin, and apigenin, which exhibit antioxidant, anti-inflammatory, antiproliferative, and proapoptotic effects[1,5,13]. They act by inhibiting key pathways such as the cellular communication system that regulates the expression of genes related to inflammation, the immune response, cell proliferation and survival (NF-κB), the transcription factors involved in cell proliferation (STAT3), and PI3K/Akt, which regulates cellular processes such as growth, proliferation, survival, migration, and metabolism. They have also been shown to inhibit the Wnt/β-catenin cell signaling pathway involved in embryonic development, tissue homeostasis, tissue regeneration, and cell proliferation. They regulate genes that code for proteins that have a crucial role in the regulation of apoptosis (e.g., Bcl-2 and Bax) and the production of caspases (p53 or TP53, which is known as a tumor suppressor gene and regulates the cell cycle and prevents the appearance of cancer). Their actionsIt include the inhibition of VEGF, which encodes a protein that participates in angiogenesis and the formation of new blood vessels, which is an essential process both in normal development and in various diseases, including cancer and macular degeneration, and matrix metalloproteinases (MMPs), which are a family of enzymes involved in the remodeling of connective tissue and the degradation of the extracellular matrix[13,34].
CAPE induces apoptosis by altering the Bcl-2/Bax ratio, releasing Cytochrome c, activating caspases, and fragmenting DNA[5]. Genistein is a soy isoflavone that inhibits tyrosine kinases, blocks angiogenesis, and regulates the expression of the p53 gene, showing efficacy in breast and prostate cancer models[2,6,7]. Resveratrol, present in grapes and wine, exerts antiproliferative, antiangiogenic, and antimetastatic effects, modulating sirtuins, p53, and cellular senescence pathways[7]. Curcumin, derived from Curcuma longa, is one of the most widely investigated compounds, showing the ability to induce apoptosis, inhibit tumor invasion, and reverse MDR, especially in combination with vinblastine[8]. Lycopene, a carotenoid abundant in tomatoes, modulates insulin-like growth factor 1 (IGF-1), a signal that plays an important role in cellular growth, development, and metabolism, and MAPK, a cell signaling cascade that regulates a wide range of cellular processes, including proliferation, differentiation, migration, and cell death, reducing proliferation and increasing apoptosis in tumor cells[2,6,7]. Allicin, derived from garlic, induces apoptosis by modulating the Bcl-2 gene, which encodes a protein that regulates apoptosis and caspases, whereas EGCG from green tea acts by inhibiting the PI3K/Akt/mTOR gene, which is involved in the regulation of cellular growth, survival, proliferation, and metabolism, consequently causing a reduction in cell proliferation and blocking tumor invasion[13].
Functional foods
-
Functional foods are considered to be those products that, when consumed, provide health benefits in addition to nutrients, for the prevention and treatment of diseases[41]. Phytochemicals present in the diet represent an accessible source of bioactive compounds that provide health benefits to those who consume them[35]. It is worth mentioning that in traditional Persian medicine, foods such as Beta vulgaris (beetroot), Brassica oleracea (cabbage), Carthamus tinctorius (safflower), Capparis spinosa (caper), Crocus sativus (saffron), and Ficus carica (fig) have been described as having anticancer properties; such findings have been experimentally validated in modern studies[35]. All these nutraceuticals not only possess direct activity against tumor cells, but also modulate the tumor microenvironment; inhibit inflammatory mediators such as cyclooxygenase-2 (COX-2), interleukin (IL)-6, and tumor necrosis factor (TNF)-α; and strengthen the immune response, suggesting their potential as therapeutic adjuvants[28,35,36].
In recent years, apitherapy has gained relevance as a complementary approach in experimental oncology[5,42]. Propolis, rich in CAPE and Artepillin C, exerts antiproliferative, antiangiogenic, and proapoptotic effects, in addition to modulating the expression of Bcl-2/Bax and caspase activation[5]. Studies have demonstrated its efficacy in breast, colon, and prostate cell lines, as well as in murine models, where a reduction in tumor size and a decrease in the tumor's microvasculature have been observed[43−45]. Royal jelly, whose main bioactive molecule is 10-HDA, has immunomodulatory, antiproliferative, and anti-inflammatory properties, and has shown synergistic effects when combined with conventional chemotherapy, increasing cytotoxicity and reducing side effects[5]. Bee venom, composed of melittin, apamin, and phospholipase A2, acts as a potent cytolytic and NF-κB inhibitor; melittin perforates the cell membranes, induces necrosis, and activates apoptotic pathways. However, its clinical use faces challenges related to its nonspecific toxicity and allergic reactions, so molecular targeting strategies, such as nanoparticles and antibody conjugates, are being investigated[45]. Bee pollen, rich in flavonoids, carotenoids, and phytosterols, has shown antioxidant and antiproliferative effects, improving endogenous antioxidant capacity and reducing oxidative damage in tumor models[5,45].
Recent studies on the anticancer activity of bioactive compounds
-
The robust cytotoxic effects of natural compounds have been reported in a wide variety of tumor cell lines, including breast cancer (MCF-7, MDA-MB-231), colon cancer (HT-29, HCT-116), prostate cancer (PC-3, LNCaP), lung cancer (A549), leukemia (HL-60, K562), and glioblastoma (U87, U251)[3,43,46−48].
In a study conducted with triple-negative breast cancer cells (the MDA-MB-468 cell line), the effect of an ethyl acetate extract derived from Streptomyces coeruleofuscus was evaluated. This extract contained natural compounds such as gallic acid, ellagic acid, and ρ-coumaric acid, which showed potent antiproliferative and proapoptotic activity. The mechanism of action involved the activation of apoptotic pathways, with increased expression of Bax, Fas, or CD95 proteins and caspases, and a reduction in MMP-9, which promoted programmed cell death and inhibited cell migration, suggesting its potential as an anti-metastatic agent[49]. In another in vitro study, the effect of curcumin on colorectal cancer cells (HCT116) was evaluated, focusing on its ability to modulate the expression of microRNAs associated with tumor growth; the ability to inhibit the proteasome and induce apoptosis was observed in the colon cancer cell lines HCT-116 and metastatic SW480, so curcumin could potentially be used for the treatment of both early- and late-stage colon cancer[50].
Several natural compounds, such as propolis, curcumin, and synthetic alkaloid derivatives, have shown to reduce tumors' volume, decrease metastasis, and prolong survival in murine models[43,47,48]. Sameni et al.[51] used BALB/c mice with azoxymethane (AOM)- and dextran sulfate sodium (DSS)-induced colorectal cancer that were treated with propolis in combination with 5-fluorouracil (5-FU). The co-treatment significantly reduced intestinal lesions and necrotic foci and suppressed the expression of COX-2, inducible nitric oxide synthase (iNOS), and β-catenin, indicating a synergistic anti-inflammatory and antiproliferative effect.
Furthermore, an ethanolic propolis extract (EEP) was administered at different concentrations to Wistar rats with dimethylhydrazine (DMH)-induced colorectal cancer. Treatment with EEP increased total antioxidant capacity, reduced oxidative stress, and significantly decreased tumor markers such as proliferating cell nuclear antigen (PCNA), carcinoembryonic antigen (CEA), and platelet-derived growth factor (PDGF), while elevating the expression of the adenomatous polyposis coli (APC) suppressor gene. These effects resulted in a significant reduction in histological damage and tumor progression[52,53].
Although most of the reviewed compounds are still in the preclinical stages, some, such as vincristine, vinblastine, CAPE, and homoharringtonine (an alkaloid from Cephalotaxus spp.), have been evaluated in clinical trials, showing efficacy against leukemia, lymphoma, and other solid tumors. However, challenges related to bioavailability, standardization, and large-scale clinical validation remain.[5,6,9,54]. Saghatelyan et al.[30] conducted a randomized, double-blind, placebo-controlled clinical trial evaluating the weekly administration of intravenous curcumin (CUC-1®, 300 mg) together with paclitaxel (80 mg/m2) in 150 women with advanced or metastatic breast cancer. After 12 weeks of treatment (with 3 months of follow-up), the objective response rate was significantly higher in the curcumin group than in the placebo group (51% vs 33%). Among those who completed the protocol, the difference was even more marked (61% vs 38%). Furthermore, patients who received curcumin reported less fatigue and better physical performance, without any detriment to quality of life, and no serious adverse effects related to curcumin were observed.
Biotechnological strategies for the production of bioactive compounds
-
One of the main challenges for the pharmacological development of plants' secondary metabolites is their low concentration in their tissues of origin. For example, vinblastine and vincristine are found in Catharanthus roseus at levels of only 0.0003%–0.0005% of dry weight, which limits their large-scale extraction and raises the production costs[12,29,55]. In response to this limitation, biotechnological strategies have been implemented that allow researchers to increase its performance, guaranteeing a sustainable supply.
Plant cell cultures and transformed roots (hairy roots), induced by Agrobacterium rhizogenes, offer a stable platform to produce high-value secondary metabolites[56]. It has been shown that the application of elicitors such as jasmonic acid, salicylic acid, yeast extract, and chromium can significantly increase the accumulation of vinblastine in embryogenic cultures of Catharanthus roseus, since these treatments induce the activation of key genes such as TDC, STR, D4H, and DAT and enzymes such as APX, GPX and CAT, which are involved in the final stages of alkaloid biosynthesis[57−59].
Maqsood and Abdul[10] used extracts of endophytic fungi, such as Piriformospora indica, Trichoderma tomentosum, and Fusarium oxysporum, as elicitors in Catharanthus roseus cultures, and observed that the plant–fungus interaction not only increased biomass but also stimulated the expression of genes related to the synthesis of vinblastine and vincristine, demonstrating the potential of using microbial elicitors as a productive intensification strategy. Genetic engineering has opened new opportunities for the microbial production of complex plant metabolites. Zhang et al.[60] achieved, for the first time, the complete synthesis of vinblastine in Saccharomyces cerevisiae, integrating more than 30 heterologous enzymatic reactions, thus generating a new paradigm for pharmaceutical production based on synthetic cellular platforms. This advance not only made it possible to overcome plant-related extraction barriers but also created a scalable platform for pharmaceutical production, with the possibility of optimizing metabolic fluxes and maximizing yield through precision genetic engineering. In turn, endophytic fungi, which are microorganisms that colonize plant tissues without causing harm, have been identified as alternative producers of the metabolites of interest. Khan and Gond[61] isolated strains of Alternaria alternata and Aspergillus clavatonanicus from Catharanthus roseus that are capable of synthesizing vinblastine and its precursors, thus opening an innovative biotechnological avenue based on microbial systems. This approach could reduce the production costs and minimize the environmental impact associated with mass plant harvesting.
Scientific attention has also been paid to developing structural analogs derived from natural products with the aim of optimizing their pharmacological activity and reducing any adverse effects[62]. Modified derivatives of the Vinca alkaloids (such as 14,15-cyclopropanovincristine and 14,15-cyclopropanovinblastine), bisindole-steroid hybrids, and triphenylphosphine conjugates have been shown to yield molecules with superior cytotoxicity to the original compounds in a panel of 60 cell lines by the National Institutes of Health (NIH). These derivatives not only exhibit greater potency but also an improved pharmacokinetic profile, with greater cellular penetration, lower efflux pump-mediated resistance, and reduced toxicity to nontransformed cells[48,60]. These advances underscore the importance of rational modifications to amplify the therapeutic potential of natural compounds.
On the other hand, it is important to mention the 'drug repurposing' process that allows us to identify new therapeutic applications for approved drugs, shortening the times and reducing the costs associated with the development of new chemical entities[63]. De Witt et al.[47] evaluated mebendazole, a Food and Drug Administration (FDA)-approved benzimidazole anthelmintic, as an alternative to vincristine in pediatric gliomas. This drug showed the ability to induce mitotic arrest and apoptosis, with an efficacy similar or superior to vincristine, but without its characteristic neurotoxicity described by Moudi et al.[16]. Mebendazole acts by destabilizing the microtubules, inhibiting tubulin polymerization, generating mitotic dysfunction, and activating apoptotic pathways; moreover, its ability to cross the blood–brain barrier makes it a promising candidate for tumors of the central nervous system[47,64]. These findings illustrate the value of drug repurposing as a strategy to expand the oncology therapeutic arsenal.
Challenges and perspectives
-
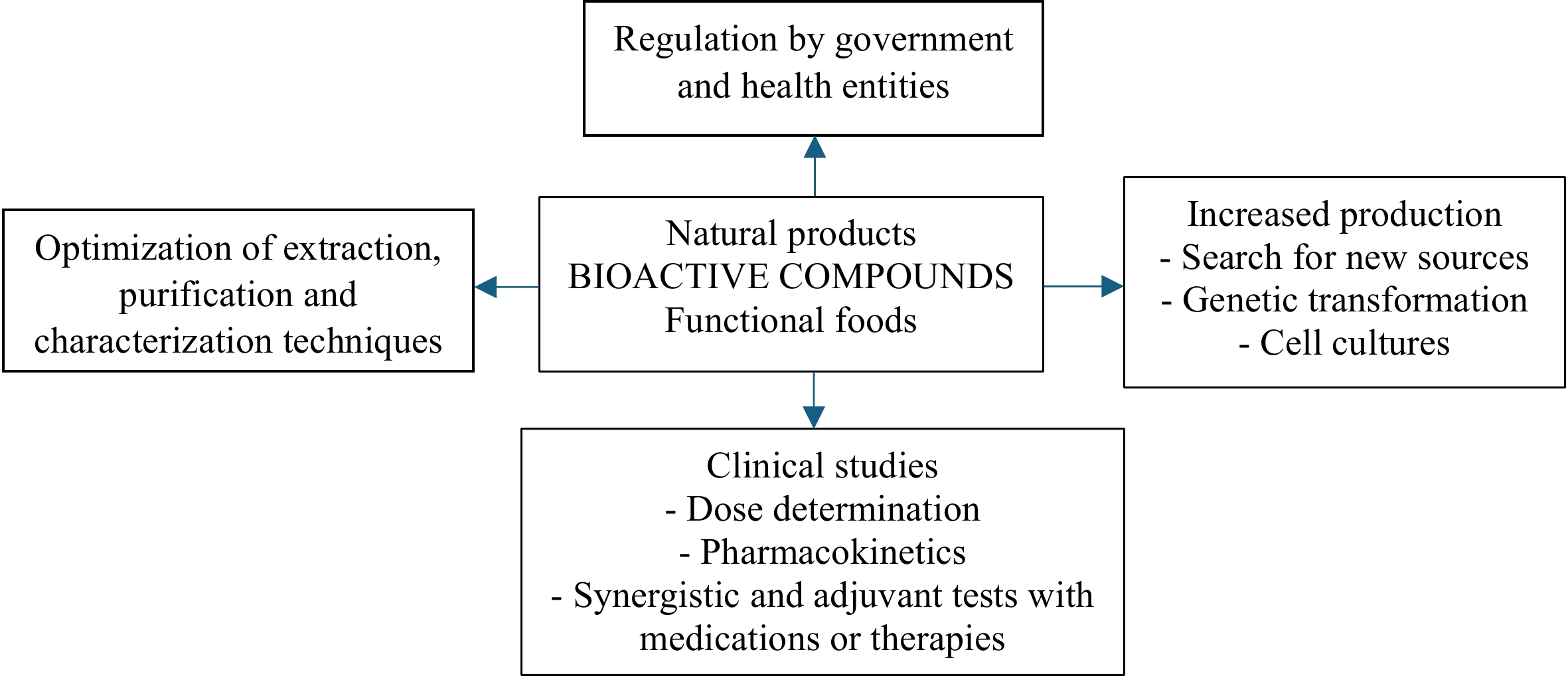
There are many studies, mainly in vitro and in vivo using animal models, and a few preclinical studies that show the potential of natural products in the treatment and control of cancer; however, there are many challenges and perspectives (Fig. 1). The large-scale production of secondary metabolites is limited by the availability of plant material, low natural yields, and the environmental impacts associated with mass collection[59]. Biotechnological strategies, such as cell cultures, transformed roots, endophytic fungi, and synthetic microbial platforms, offer sustainable solutions to overcome these barriers[11,61]. Despite the remarkable potential shown by natural compounds in preclinical studies, their clinical translation faces multiple challenges that limit their practical application and regulatory approval. Many natural compounds have pharmacokinetic limitations, such as low water solubility, rapid metabolism, chemical instability, and poor oral bioavailability[54]. Examples include curcumin, resveratrol, and EGCG, whose therapeutic effects observed in vitro are not easily translated into in vivo results because of their rapid elimination from the body[7,13]. To overcome these barriers, advanced formulations based on nanotechnology (nanoparticles, liposomes, micelles), controlled release systems, targeted conjugates (such as antibody–drug combinations), and chemical modifications that improve stability and bioavailability are being developed.
On the other hand, the chemical composition of natural extracts can vary significantly depending on factors such as the geographical origin, growing conditions, time of collection, and extraction methods[54]. This heterogeneity makes standardization and quality control difficult, complicating the reproducibility of the results[5]. The development of international standards and the detailed characterization of phytochemical profiles are fundamental steps to ensure the safety and efficacy of these agents. It is also important to consider that natural products can interact with conventional medications, altering their metabolism, bioavailability, or toxicity[16]. Therefore, it is crucial to perform detailed pharmacokinetic and pharmacodynamic studies that evaluate possible synergies, antagonisms, or unexpected adverse effects[54].
-
The growing need to prevent and treat various diseases makes biotechnology through biomedicine look for compounds present in natural sources with biological activity effective against pathologies of the autoimmune type; of the circulatory, endocrine, digestive, and respiratory systems; and organs and tissues such as the lung, kidney, liver, colon, breast, pancreas, stomach, bladder, and skin, etc. As a result, it has been possible to verify the positive effect on human health of compounds such as polyphenols, flavonoids, carotenoids, and other phytochemicals such as the alkaloids of the Vinca family (specifically vincristine and vinblastine) present in Catharanthus roseus, the caffeic acid present in propolis, and curcumin from turmeric, which stand out for their antioxidant, anti-inflammatory, antiproliferative, and antiangiogenic properties. These have shown their efficacy in preclinical studies against various types of cancer and seem to have selectivity towards cancer cells. Resveratrol, quercetin, lycopene, and isoflavones have been associated with anticancer activity in in vitro and in vivo studies, preventing cell proliferation, signaling, epigenetics, and tumor metabolism. However, the existing evidence varies, depending on several aspects, including the method of extracting the bioactive compounds, their method of administration to the patient, the sample size, the time of exposure to treatment, and the frequency of administration. Therefore, to justify the use of these biocompounds as single-use therapies or as an adjuvant with beneficial effects, further research is required. This should include standardized operating procedures, larger patient samples, defining the frequency and duration of treatments, and finally, evaluating and determining the optimal concentration of each compound, the administration mechanism, and the possible synergistic effect with chemical medications used by the patient during treatment.
-
Natural products represent a fundamental pillar in the history of oncology, having given rise to some of the most successful antineoplastic agents, such as vinblastine, vincristine, taxanes, and epipodophyllotoxins. Beyond these classic molecules, a vast arsenal of compounds derived from plants, fungi, functional foods, and bee products continues to offer new therapeutic opportunities. It is important to highlight these significant advances in the understanding of the molecular mechanisms of action of these compounds, their synergistic potential with conventional chemotherapy, and the biotechnological strategies that allow for optimizing their production and sustainability. However, for these agents to effectively reach clinical practice, critical barriers related to bioavailability, standardization, scalability, and clinical validation must be overcome. The integration of emerging technologies, such as nanotechnology, synthetic biology, and pharmacogenomics, along with personalized oncology approaches, will be key to maximizing their therapeutic impact in the 21st century. Natural products and their biotechnological derivatives not only represent a valuable source of antineoplastic agents but also embody a multidisciplinary paradigm that unites biodiversity, biotechnology, pharmacology, and medicine in the fight against cancer.
-
Not applicable.
-
The authors confirm contribution to the paper as follows: study conception and design: Díaz R, data collection: Díaz R, Rodríguez-Requenes ME, analysis and interpretation of the results: Díaz R, Díaz-Godínez G, Narvaez-Padilla JA; draft manuscript preparation: Rodríguez-Requenes ME, Díaz-Godínez G. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing is not applicable to this review as no datasets were generated or analyzed during the current study.
-
To Secretariat of Science, Humanities, Technology and Innovation (SECIHTI) for the scholarship to study for a master's degree awarded to Moisés Eluzaí Rodríguez-Requenes (Grant No. 4032210).
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of Nanjing Agricultural University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Rodríguez-Requenes ME, Díaz-Godínez G, Narvaez-Padilla JA, Díaz R. 2026. Main bioactive compounds against cancer and their mechanisms of action. Food Materials Research 6: e004 doi: 10.48130/fmr-0026-0005
Main bioactive compounds against cancer and their mechanisms of action
- Received: 20 September 2025
- Revised: 20 January 2026
- Accepted: 29 January 2026
- Published online: 23 March 2026
Abstract: Human health is a privilege that is extremely difficult to maintain because of the epigenetic conditions that cause the development of diseases of different natures, including immunological, respiratory, hepatic, renal, digestive, and glandular diseases, among others. Cancer is a pathology characterized by the rapid multiplication of abnormal cells and is the leading cause of death worldwide. For this reason, science makes great efforts to find treatments that help eradicate or control the disease in order to maintain or prolong the life of the patient. For this, medications and therapies of chemical origin, such as radiotherapy and chemotherapy, have been developed, whose purpose is to eliminate cancer cells. However, during this process, the quality of life can be degraded, in addition to multiple ailments suffered by patients, so it is necessary to find alternatives of natural origin that can contribute to the elimination of cancer cells, minimizing adverse side effects. Among these alternatives are bioactive molecules produced primarily by plants. In this regard, this manuscript reviews in vitro studies of the cellular mechanisms of action of bioactive compounds and their sources, including functional foods, investigating the molecular origin of these substances at the genomic level and finally offering future perspectives and the potential challenges to overcome in establishing their possible application as adjuvants in cancer treatments.
-
Key words:
- Bioactive compounds /
- Cancer /
- Functional foods /
- Natural products /
- Phytochemicals