-
The rapid increase in demand for fruit trees has led to growing interest in research into tissue culture. This process enables the nutritional organs to undergo dedifferentiation and form callus tissue[1]. Besides micro-propagation, in vitro callus induction and bud regeneration has also been applied in research including gene transformation and polyploidy induction in a number of fruit trees, such as banana[2,3], kiwifruit[4,5], apple[6,7], orange[8,9], and plum[10]. Although it offers high efficiency and rapid multiplication, a considerable number of limitations still exist. For example, the long duration from the induction of tissue-cultured plantlets to their field transplantation, and the requirement to maintain a sterile environment[11−13]. These factors substantially inhibit progress in research related to genetic transformation and mutation breeding.
In vivo callus formation is a common phenomenon after wounding, grafting, or girdling in fruit crops. However, callus induction under natural field conditions is very rare. To date, callus induction in vivo has been reported on jujube, living lime trees, grape, and pomegranate[14−18]. Recent studies have shown that inducing mutations via callus tissue formed under natural field conditions has become a key technical strategy for germplasm innovation[19]. Compared to conventional in vitro callus induction and bud regeneration methods, the in vivo system can offer significant advantages, including a simpler induction system, shorter time duration, and lower costs in terms of both resources and labor. It is noteworthy that both jujube and pomegranate have achieved significant progress by refining the processes for field callus induction, resulting in the effective production of pure polyploid[11,17]. Using this method for Chinese jujube breeding, polyploid plants regenerated from in vivo culture could bloom within the experimental year, eliminating the need for sterile environments and subculture passages required by in vitro systems, thereby saving at least 2 to 3 years in the polyploid breeding process of this species[18]. These advancements in research and technology have substantially reduced the duration of the traditional breeding cycle, thereby providing innovative technical approaches and theoretical frameworks for breeding in fruit trees.
Actinidia arguta is one species of deciduous woody vine of the genus Actinidia. It is distributed in cold temperate areas in Asia (e.g., Northeastern China)[20−22]. Compared to Actinidia deliciosa and Actinidia chinensis, the fruit of A. arguta has a much thinner exocarp, a smoother and hairless surface, and has higher nutritional value. The vitamin C content of A. arguta fruit on average is 430 mg/100 g[23−25]. During grafting experiments, we observed that callus tissue formed at the grafting incision site and differentiated into adventitious buds; however, the frequency of adventitious bud differentiation was extremely low. This phenomenon inspired us to explore alternative methods to enhance adventitious bud regeneration efficiency (Supplementary Fig. S1). Therefore, the induction of callus tissue in vivo for A. arguta, along with the establishment of a bud regeneration system, has the potential to improve breeding efficiency. This approach will establish a groundwork for the genetic transformation and polyploid breeding of A. arguta under in vivo conditions.
-
Six-year-old A. arguta 'Kui Lv' was used as the experimental material, grown on the campus of the Jilin Agricultural University (43°48'48'' N, 125°24'15'' E). The annual precipitation is 661 mm, and the highest and lowest temperatures are 36 and −25 °C, respectively. Thirty plants of 'Kui Lv' were selected. Plant materials were of uniform size. Each experiment was divided into three subgroups as biological replicates, with each subgroup containing 10 new branches. From each plant, we selected new branches with a similar size (stem diameter of 0.6−0.8 cm) from a vine at the same height. The management history of plant materials is uniform.
New branches were cut from the internode with a razor blade in early June. These new branches were pruned to create a smooth cut surface, which was then covered with absorbent cotton for subsequent treatments (Supplementary Fig. S2a). Then, we immediately added related solutions (2 ml) onto the cut surface that was covered with absorbent cotton (Supplementary Fig. S2b). Finally, we wrapped them with different coverings (Supplementary Fig. S2c).
Effects of different coverings on the formation of callus
-
Distilled water was added to the cut surface that was covered with absorbent cotton. Two experiments were conducted to optimize covering conditions for callus induction.
Experiment 1: Single-covering materials. Cut surfaces without any covering served as the control. Three single-layer materials were tested: white plastic bags (WP), black plastic bags (BP), and kraft paper bags (KP).
Experiment 2: Combined-covering methods. Based on results from experiment 1, BP was selected as the base material. Cut surfaces covered with BP alone served as the control. Three combined methods were evaluated: (1) KP + BP (BP inner layer, KP outer layer); (2) BP + humid mud + BP (BP inner, humid mud middle, BP outer); and (3) WP + BP (WP inner, BP outer). The callus induction rate was investigated every five days over a one-month period. The growth grade of the callus was recorded at 15 and 30 d after treatment to determine the most effective covering method.
Effects of growth regulator and AgNO3 on callus formation and bud regeneration
-
To determine the conditions for bud regeneration in vivo, the most effective covering method was employed. Three experiments were carried out to determine the in vivo bud regeneration conditions, including plant growth regulators, silver nitrate (AgNO3), and their dark processing duration. The AgNO3 solution was freshly prepared and stored in a light-proof container (brown glass bottle). In field applications, we covered the treatment site with black plastic immediately after application.
Experiment 1: Optimization of plant growth regulators. Different concentrations of auxin (NAA, IBA, 2,4-D [1, 2, 4, 8 mg/L]) and cytokinin (TDZ, ZT, 6-BA, KT [1, 2, 4, 8, 10 mg/L]) were respectively applied to the cut surface to induce callus and bud regeneration. In the control, plant growth regulators were replaced with distilled water. Different concentrations of mixed solutions of cytokinin together with auxin were applied to optimize the combined treatments (Supplementary Table S1). We added 2 ml of various concentrations of plant growth regulators to the absorbent cotton for the different treatments. Application frequency was a single dose, not in multiple drops over time.
Experiment 2: Optimization of AgNO3 concentration. To inhibit browning and hardening of callus tissue, the optimal hormone concentration (determined in experiment 1) was applied together with AgNO3. Concentrations of 1–6 mg/L were applied to identify the most effective level. The number of regenerated buds was recorded 20 d after callus induction.
Experiment 3: Optimization of dark culture duration. To determine the optimal dark culture period, cut surfaces were first covered with absorbent cotton, followed by application of the optimal hormone and AgNO3 concentrations (determined in experiments 1 and 2). The treated branches were then covered with a double-layer wrapping of black plastic (BP, outer layer) and white plastic (WP, inner layer) to create a dark, high-humidity environment. Six dark culture durations were tested: 5, 10, 15, 20, 25, and 30 d. A control treatment without dark culture (WP only, natural light) was included for comparison. After the dark culture period, all treatments were transferred to natural light conditions for continued culture, and the WP + BP covering was replaced by WP alone.
Investigation of callus grade and bud regeneration rate
-
The percentage of callus induction was calculated using the following formula: Callus induction rate (%) = (Number of branches with callus / Total number of treated branches) × 100%. The evaluation of callus grades is based on the methods of Li and Jiang, and optimized through a preliminary experiment on this basis[17,25] (Table 1, Fig. 1). Additionally, adventitious bud regeneration rate was assessed after full leaf expansion (defined as development of two leaves), using the following formula: Bud regeneration rate = (Total number of regenerated buds / Total number of treated branches) × 100%.
Table 1. Grading standards for callus.
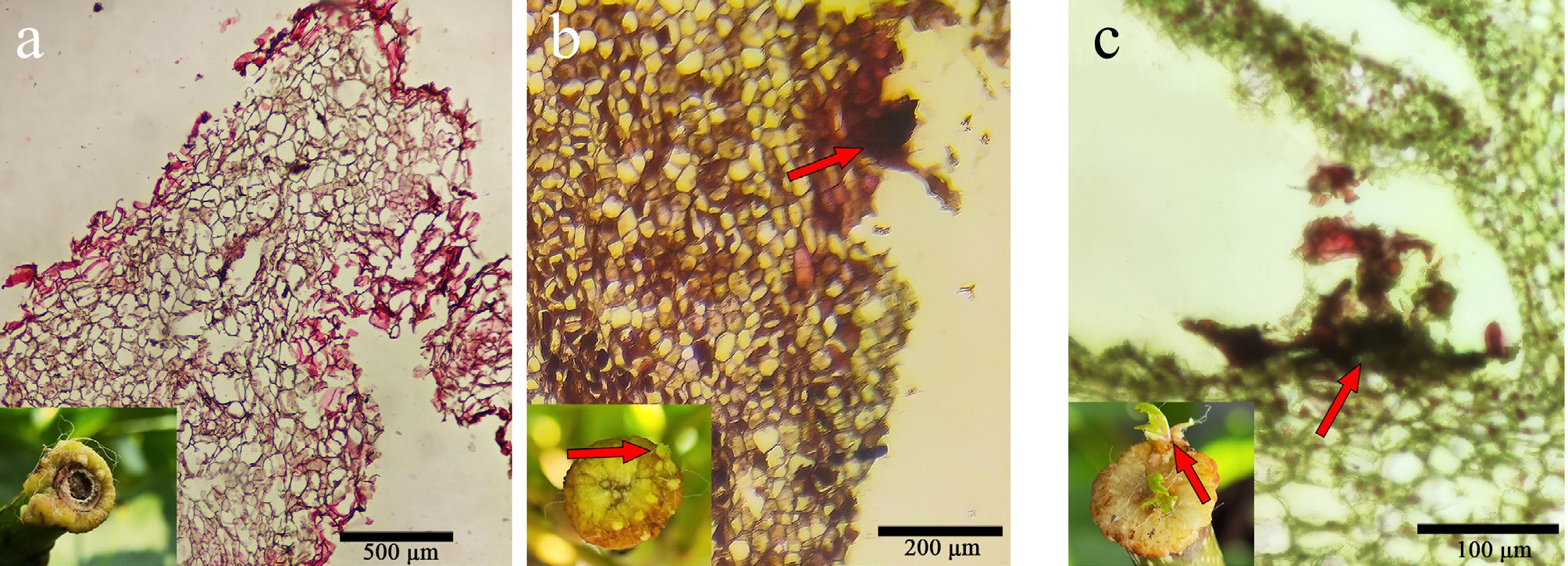
Grade Typical characters 0 The presence of calluses was not observed (Fig. 1a) 1 A cambium-derived callus became observed (Fig. 1b) 2 Callus has grown beyond the cut section and the callus circle has become thicker (Fig. 1c) 3 The callus circle becomes significantly thicker with its width accounting for between one-fifth and one-half of the radius of the cut surface (Fig. 1d) 4 The width of the callus circle is approximately equivalent to one-half of the radius of the cut surface (Fig. 1e) 5 The width of the callus circle accounts for more than one-half of the radius of the cut surface (Fig. 1f) 6 Callus fully covered the cut surface (Fig. 1g) 7 Callus proliferated and overflowed the cut surface (Fig. 1h) 
Figure 1.
Callus grading of branch sections of A. arguta. (a) Grade 0 (treatment for 1 d). (b) Grade 1 (treatment for 5 d). (c) Grade 2 (treatment for 10 d). (d) Grade 3 (treatment for 15 d). (e) Grade 4 (treatment for 20 d). (f) Grade 5 (treatment for 25 d). (g) Grade 6 (treatment for 40 d). (h) Grade 7 (treatment for 60 d).
Data analysis
-
Statistical analysis was conducted with SAS (JMP 6.0, SAS). In data sets with parametric distributions, significant differences between treatment group means were measured using the Student's t-test (p < 0.05). Callus induction rate and bud regeneration rate was measured as the average of three replicates per experiment. Every replicate contained ten new branches.
-
This study aimed to investigate the effect of different covering materials on callus induction. The results showed that there were obvious differences in the callus growth grade and induction rate under different covering treatments. Among the covering materials, the callus growth grade under BP coverage was markedly higher than that of the control (Fig. 2a). Additionally, the BP covering achieved the highest callus induction efficiency and the shortest induction time, significantly outperforming other treatments (Fig. 2b). By contrast, in the control group, callus growth was poor and essentially ceased (Fig. 2c). Callus of WP (Fig. 2d), and KP (Fig. 2e) treatments exhibited mostly yellowish-brown coloration, hard texture, dark and dull surfaces, and marginal cracks accompanied by brown spots, along with frequent browning and growth cessation. Under treatment BP, the callus presented a relatively smooth surface with yellow-white coloration. The tissue was soft in texture and displayed a distinctly granular morphology (Fig. 2f).
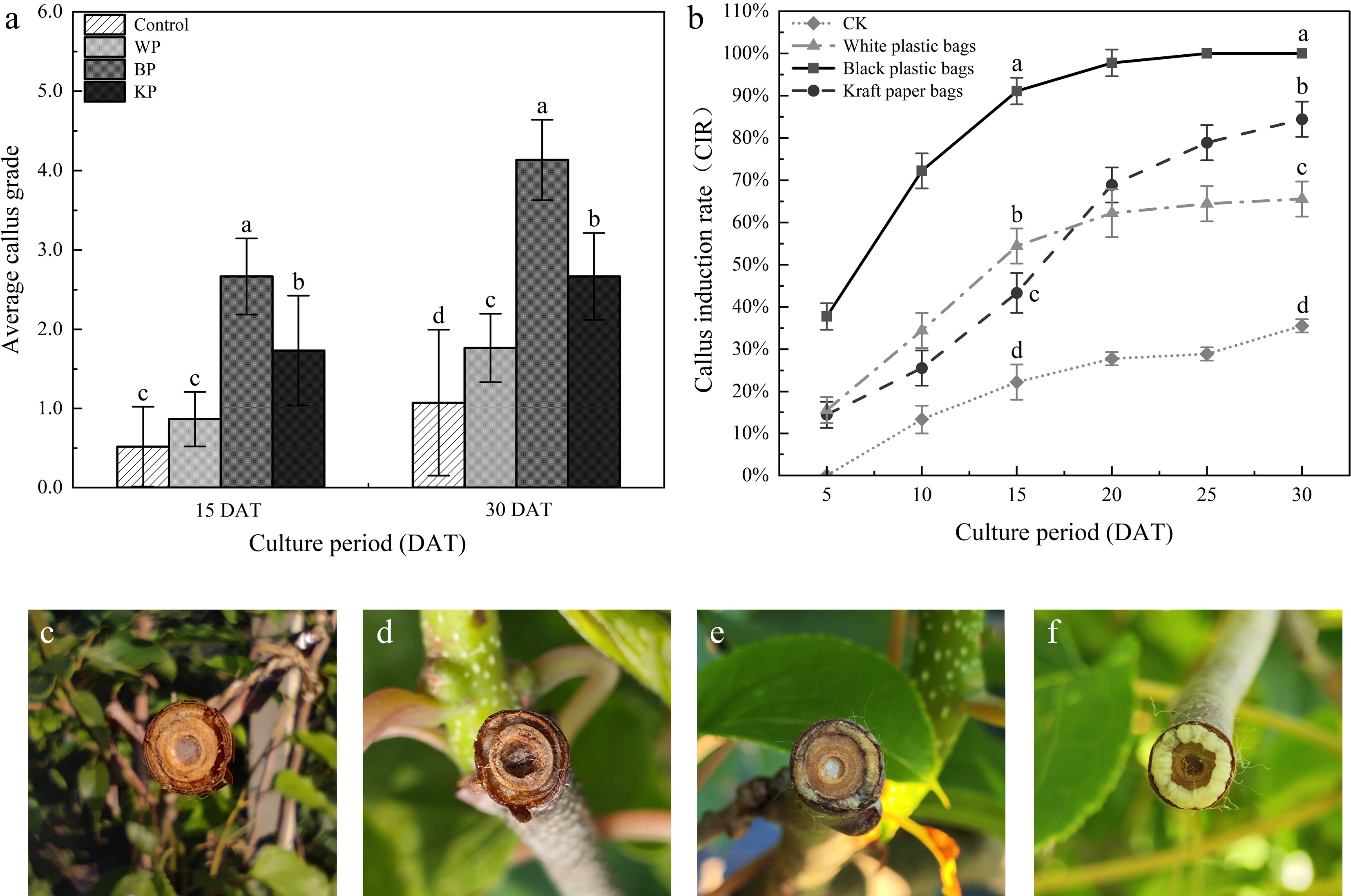
Figure 2.
Effect of different covering materials on the growth grade and morphology of callus. CK, without any covering. (a) Investigation on callus growth grade. (b) Investigation on callus induction rate. (c) Without any covering. (d) WP. (e) KP. (f) BP. Note: different letters indicate significant differences (p < 0.05).
Optimizing the effects of different covering treatments on callus formation
-
To optimize the callus-inducing performance of BP as a covering material, we designed three combination treatments. The results showed that the callus growth rating for WP + BP was significantly higher than other treatments (Fig. 3a). BP and BP + humid mud + BP showed lower callus induction (Fig. 3b, d). In contrast, the callus surface of KP + WP exhibited dry and brown spots (Fig. 3c). Following WP + BP, the callus was milky-white, firm, and granular, and the callus surface remained slightly moist (Fig. 3e). Based on callus growth data under different coverage conditions, we identified WP + BP as the optimal treatment.
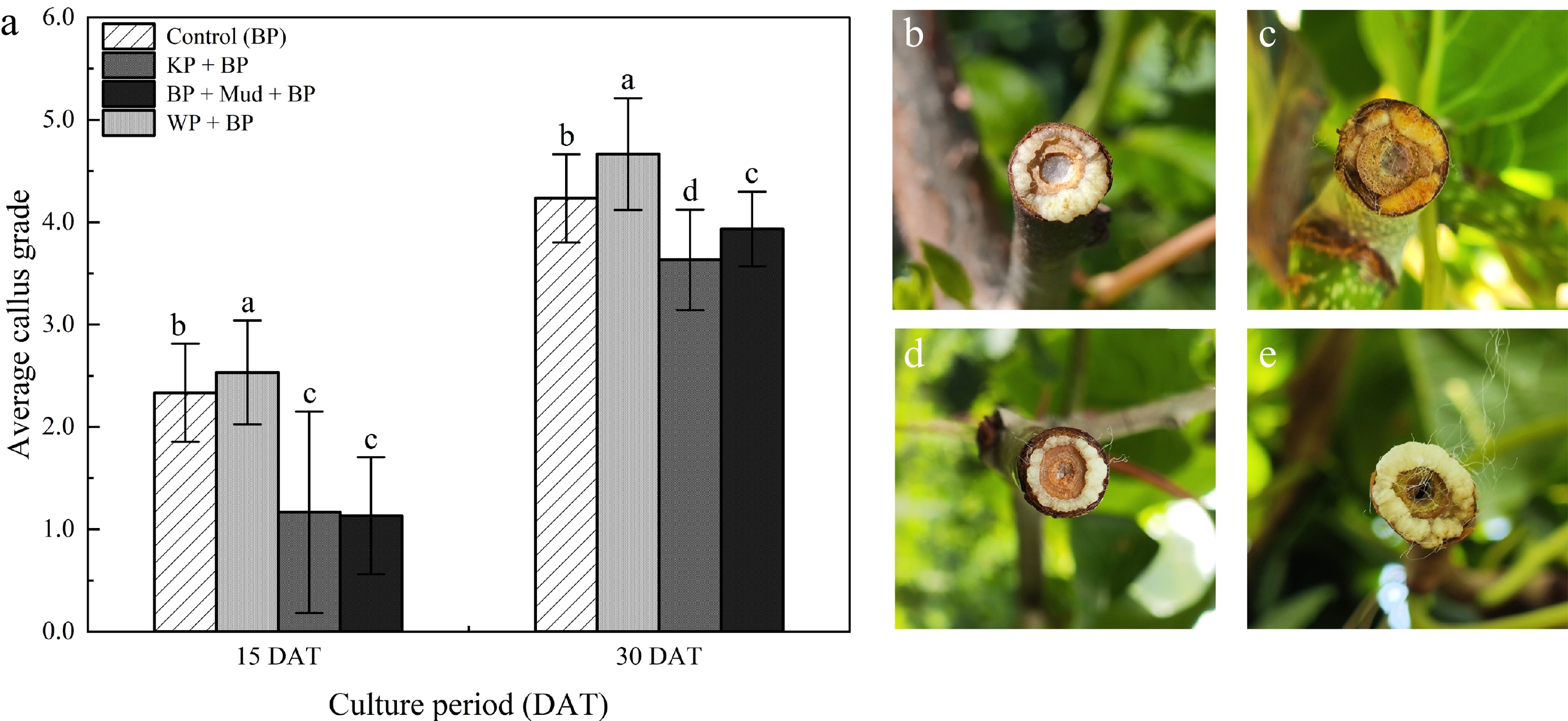
Figure 3.
Optimize the effects of different combination of covering materials on the growth rate and morphology of callus. Using the BP covering as the control. (a) Investigation on callus growth grade. (b) BP. (c) KP + BP. (d) BP + humid mud + BP. (e) WP + BP. Note: different letters indicate significant differences (p < 0.05).
In vivo callus induction and adventitious bud regeneration
Effects of different auxin concentrations on callus induction and adventitious bud regeneration
-
Auxin analogs can upregulate cell-cycle-related genes, thereby promoting callus formation and subsequent organogenic development. To increase the induction rate of callus tissue and shorten the induction time, different concentrations of NAA, IBA, and 2,4-D were applied on the cut surface.
After 20 d of treatment, it was found that, except for the IBA group, all other treatment groups achieved a 100% callus induction rate. The average callus grade initially increased, and then gradually decreased with a concentration increase of NAA and 2,4-D, but the trend of the callus growth grade maintained a decrease with an increase in IBA concentration. The optimal concentrations were 4.0 mg/L NAA, 4.0 mg/L 2,4-D, and 1.0 mg/L IBA.
At these optimal concentrations, the average callus grade of 2,4-D was significantly higher than IBA and NAA (Fig. 4a). Under the coverage treatments applied in this study, induced embryogenic callus and initial differentiation were detected on callus surfaces at 52 d for the 4.0 mg/L 2,4-D treatment, and at 58 d and at 75 d for the 1.0 mg/L IBA treatment, respectively (Fig. 4b–d). In contrast, adventitious bud formation was not observed under other auxin treatments.
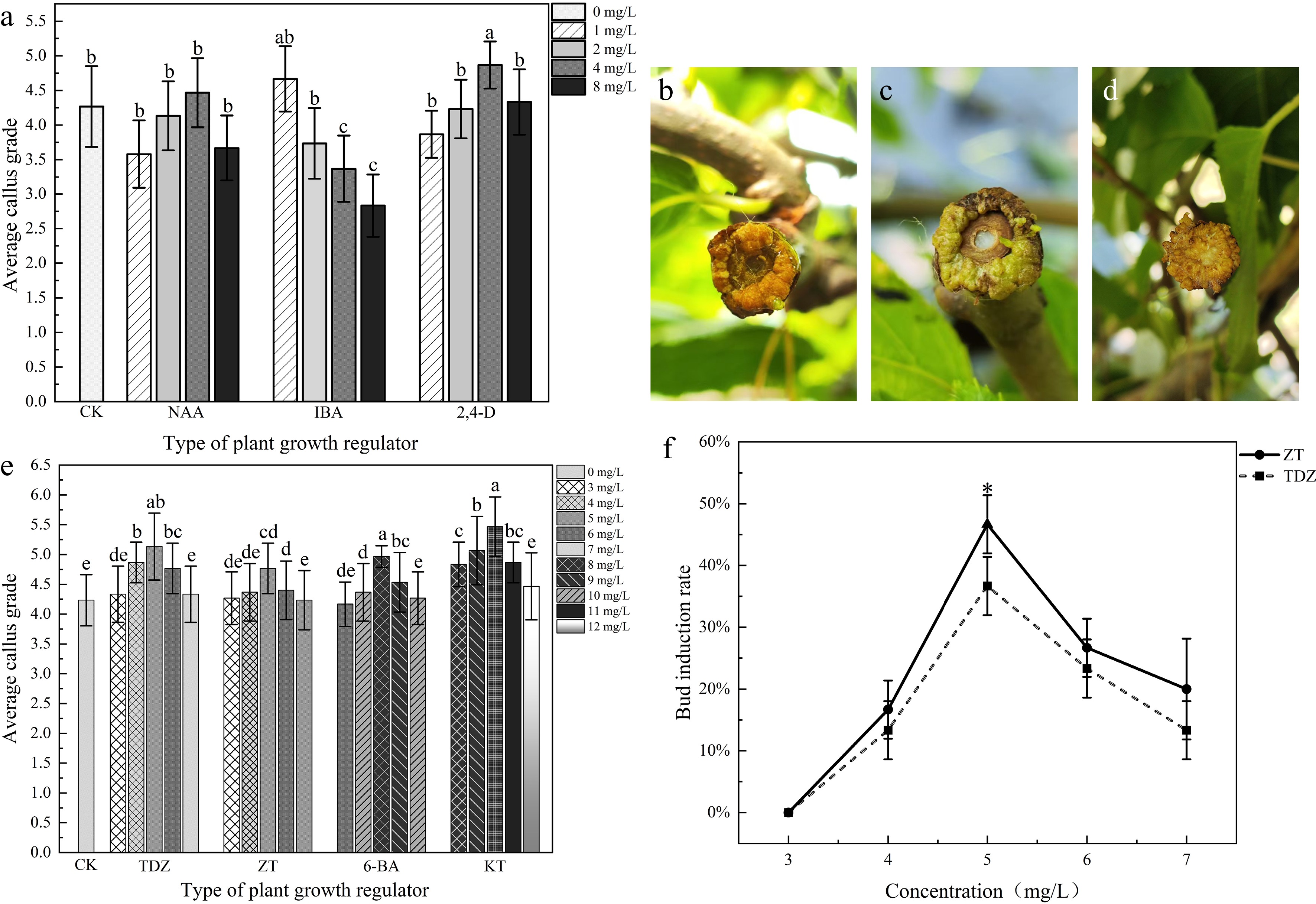
Figure 4.
The effects of different plant growth regulators on callus induction and bud regeneration. (a) Effects of auxin analogues on callus growth grade. (b) Adventitious bud regeneration after 52 d of treatment with 4.0 mg/L 2,4-D. (c) Adventitious bud regeneration after 58 d of treatment with 1.0 mg/L IBA. (d) Adventitious bud regeneration after 75 d of treatment with 1.0 mg/L IBA. (e) Effects of different cytokinin concentrations on callus growth grade. (f) Effects of ZT and TDZ concentrations on bud induction rate. Note: different letters or asterisks indicate significant differences between ZT and TDZ (p < 0.05).
Effect of different cytokinin concentrations on callus induction and adventitious bud regeneration
-
Cytokinins can maintain continuous division of callus tissue and facilitate bud regeneration. Firstly, the experiment was conducted using concentration gradients of 1.0, 2.0, 4.0, 8.0, and 10 mg/L to determine the suitable concentration range (Supplementary Table S2). Secondly, optimization was performed based on these results (Fig. 4e). The results demonstrated that four cytokinins achieved a 100% callus induction rate, and treatments with 8.0 mg/L 6-BA, 10.0 mg/L KT, 5.0 mg/L TDZ, and 5.0 mg/L ZT exhibited a relatively higher callus induction grade (Fig. 4e).
Nevertheless, the corresponding bud regeneration rate remained relatively low using concentration gradients of 1.0, 2.0, 4.0, 8.0, and 10 mg/L (Supplementary Table S2). Under 6-BA, TDZ, and ZT treatments, callus quality initially improved with increasing concentrations, reaching a maximum grade at 8.0 mg/L, 4.0 mg/L, and 4.0 mg/L, which was accompanied by a bud regeneration rate of 13.3%, 13.3%, and 16.7% (Supplementary Table S2). With further concentration increases of 6-BA, TDZ, and ZT, bud regeneration capacity exhibited a continuous decline and ultimately diminished to zero. For KT treatments at concentrations ranging from 8.0 to 12.0 mg/L, the bud regeneration rate was zero (Supplementary Table S2).
For TDZ and ZT treatments, we further optimized the concentrations for bud regeneration. The bud regeneration rates exhibited an initial increase, followed by a decrease with concentration increase; specifically, the maximum bud regeneration rates of 36.7% and 46.7% were achieved at 5.0 mg/L, respectively (Fig. 4f).
Effect of AgNO3 on callus induction and bud regeneration
-
During the callus induction process, browning is usually observed. Recent studies have demonstrated that AgNO3 effectively inhibits the physiological activity of ethylene. AgNO3 can significantly enhance the differentiation rate of bud regeneration and reduce browning in plant tissues. Based on these findings, different concentrations of AgNO3 (ranging from 1.0 to 6.0 mg/L) were applied together with 5.0 mg/L zeatin (ZT).
These results showed that the callus growth grade and bud regeneration rate initially increased and then decreased as the concentration of AgNO3 increased. Specifically, treatment with 4.0 mg/L AgNO3 resulted in a significantly higher callus growth grade compared to other treatments (Fig. 5a), with a bud regeneration rate of 56.7% (Fig. 5b). However, higher AgNO3 concentrations imposed a pronounced inhibition on bud regeneration from callus. At 6.0 mg/L, adventitious bud regeneration was completely lost (0% bud regeneration). Induced embryogenic callus and initial differentiation were detected on callus surfaces at 43, 45, and 48 d under the treatments of 2, 3, and 5 mg/L AgNO3, respectively (Fig. 5c–e). However, the 4 mg/L treatment outperformed other treatments by producing embryogenic callus on the callus surface at 40 d with a reduced induction time (Fig. 5f).
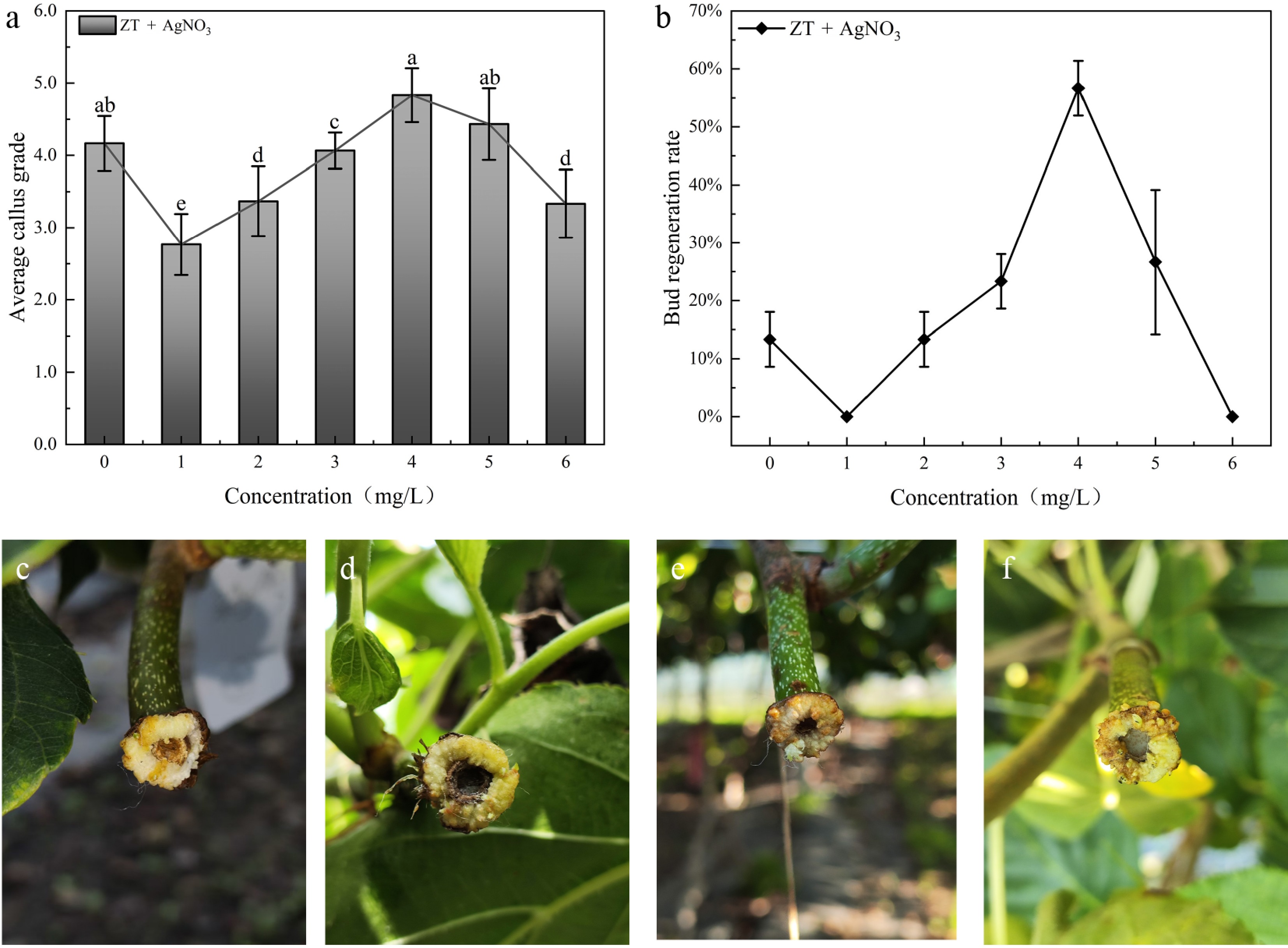
Figure 5.
The effect of ZT combined with different concentrations of AgNO3 (1.0–6.0 mg/L) on callus and bud regeneration. (a) Investigation on callus growth grade. CK: without AgNO3 addition. (b) Investigation on bud regeneration rate of callus. (c) 45 d, ZT 5.0 + 2.0 mg/L AgNO3. (d) 43 d, ZT 5.0 + 3.0 mg/L AgNO3. (e) 40 d, ZT 5.0 + 4.0 mg/L AgNO3. (f) 48 d, ZT 5.0 + 5.0 mg/L AgNO3. Note: different letters indicate significant differences (p < 0.05).
The effects of different dark culturing durations on callus formation and bud regeneration
-
To optimize dark culturing duration for both callus induction and bud regeneration, the cut surface of the branch was initially covered with absorbent cotton, followed by the application of 5.0 mg/L zeatin (ZT), and 4.0 mg/L AgNO3 (Table 2). After dark culturing duration, all treatments could produce callus, but with different grades. These results indicated that different dark culturing times obviously influenced callus induction and bud regeneration. Callus grade was the highest at 10 d and 15 d of dark incubation, but decreased significantly at 20 d and 25 d. Ten-day dark incubation maximized the adventitious bud differentiation rate, whereas longer periods induced a significant decline.
Table 2. Effect of different dark culture time on callus formation and bud regeneration.
Concentration
(mg/L)Dark culture
durationAverage diameter
of branches (cm)Callus induction
rate (%)Average callus
gradeAdventitious bud
regenerationBud regeneration
rate (%)ZT5.0 + 4.0 AgNO3 0 d 0.64 a 93.3 b 0.73 ± 0.87 d − 0 5 d 0.67 a 100 a 1.97 ± 0.55 c − 0 10 d 0.73 a 100 a 4.67 ± 0.48 a 40 d 76.7 a 15 d 0.71 a 100 a 4.73 ± 0.44 a 49 d 53.3 b 20 d 0.62 a 100 a 4.13 ± 0.62 b 61 d 36.7 c 25 d 0.69 a 100 a 4.23 ± 0.62 b 67 d 13.3 d 30 d 0.78 a 100 a 4.37 ± 0.55 ab 70 d 6.7 e Morphological differentiation process of adventitious bud regeneration from callus of Actinidia arguta
-
The current year's new shoots were cut and subsequently treated with ZT (5.0 mg/L) in combination with AgNO3 (4.0 mg/L) to promote bud regeneration (Fig. 6a). After 10 d of culturing duration, callus tissue was observed on the cut surface covering approximately 50% of the cut surface (Fig. 6a). After 20 d (Figs. 6b, 7b), the callus tissue exhibited continued swelling and vigorous growth. After 25 d, the callus transformed into yellowish-white granules, with translucent callus, and a light yellowish-white bud primordium became evident (Figs. 6c, 7c). After 30 d, adventitious buds were differentiated from the callus tissue, and lignification was initiated in the callus tissue (Fig. 6d). By approximately 40 d, the shoots had reached a height of about 2–3 cm (Fig. 6e). It was observed that 72 d after treatment, the plants had developed normally, and had become completely lignified (Fig. 6f).

Figure 6.
Callus development and adventitious bud regeneration from branches of A. arguta under optimal treatment. (a) 0 d, freshly cut surface. (b) 20 d, visible callus proliferation. (c) 25 d, callus expansion. (d) 30 d, continued callus growth. (e) 40 d, well-formed adventitious buds. (f) 72 d, the plant has been completely lignified.
-
The system of in vivo callus induction and shoot regeneration is much simpler, less costly, and more convenient[26−30]. Previous work on A. arguta had observed that adventitious buds could be induced from callus that generated from the graft cut surface under field conditions (Supplementary Fig. S1), although the frequency remains low. This mechanism likely involves impaired polar auxin transport at the wound site, resulting in localized elevation of the cytokinin/auxin ratio, and subsequent cell fate determination toward organogenesis[31,32]. The processes are termed as de novo organogenesis that is among the most energy-demanding and failure-prone regenerative events in plants. However, wound healing and defence reactions are the top priority survival strategies[33,34]. Thus, A. arguta offers an attractive, largely untapped model for exploiting in situ bud regeneration in fruit-tree improvement.
Effects of different hormones on callus and bud regeneration
-
Some studies have demonstrated that under in vitro conditions, although the synergistic interaction between auxins and cytokinins is essential for callus induction and adventitious bud differentiation in A. arguta, the optimal hormone combinations remain controversial and exhibit significant genotype-dependent variation[35−37]. Wang[38], and Zheng[39] reported that 6-BA exhibited superior effects on callus induction compared to KT when applied within optimal concentration ranges. Furthermore, ZT was gradually replacing traditional 6-BA as the preferred cytokinin due to its superior performance in both adventitious bud induction rate and quality[40]. In the present study, ZT exhibited more consistent induction capacity than 6-BA and TDZ, consistent with previous reports. However, no significant difference was detected between 6-BA and KT in callus induction efficacy.
In vivo systems of jujube, grape, and pomegranate, the application of appropriate cytokinin and auxin concentrations on the wound site has proven to be a critical strategy for promoting callus formation and adventitious bud differentiation[11,16,17]. In Chinese jujube, IBA alone failed to induce callus formation, whereas either TDZ alone, or TDZ combined with IBA could have had a better induction effect. By contrast, our experimental results demonstrated that IBA alone was capable of inducing callus formation in A. arguta, albeit with a lower bud regeneration rate. Furthermore, TDZ alone in this study significantly outperformed the TDZ + IBA combination. These differences between Chinese jujube and A. arguta may be attributed to interspecific variation. However, the present study revealed that exogenous application of cytokinin combined with auxin to the wound sites of A. arguta resulted in extremely low bud regeneration rates (Supplementary Table S1, Supplementary Fig. S3). Specifically, the combination of ZT with either IBA or NAA resulted in inhibited callus growth, likely attributable to auxin overdose-induced hyper-proliferation that compromised subsequent callus tissue differentiation[41−44]. In contrast, treatment with 5 mg/L ZT alone for 40 d yielded the highest bud regeneration rate of 46.7%, significantly surpassing all other treatments. Importantly, this optimal concentration (5 mg/L) substantially exceeds the 2.0 mg/L commonly used in in vitro culture systems, indicating an order-of-magnitude difference in hormonal dosage requirements. This discrepancy may be attributable to differences in tissue absorption barriers or metabolic degradation rates under field conditions[45,46].
Effect of AgNO3 on callus induction and bud regeneration
-
After wounding stress, the cells surrounding the wound rapidly produce large amounts of ethylene and phenolic compounds[47,48], leading to browning and hardening of the callus tissue. This process partially inhibits cell differentiation and the formation of new organs[49]. Numerous studies have shown that Ag+ can inhibit effective ethylene activity[50,51]. AgNO3 as an effective inhibitor of ethylene action can mitigate callus tissue browning and indirectly promote callus formation and growth by delaying ethylene-induced senescence[52,53]. This effect has been confirmed in coffee, tomato, pepper, potato, strawberry, and other species[54−59]. Our study demonstrated that applying 4.0 mg/L AgNO3 increased the bud regeneration rate to 56.7%, approximately 20% higher than using 6 mg/L ZT alone, thereby AgNO3 further promoting bud regeneration of A. arguta.
Although the application of AgNO3 may increase material costs compared to hormone-only treatments, the improvement in bud induction rate and the shortened regeneration cycle will offset the additional expenses. Furthermore, the recommended concentration (4.0 mg/L) is relatively low, and the volume of solution applied per plant is minimal. In addition, it should also be noted that the field-based approach eliminates expenses associated with maintaining sterile culture conditions. We will explore lower-cost alternatives or optimize application protocols to further reduce costs.
Effects of dark culturing duration on callus formation and bud regeneration
-
Previous studies have demonstrated that dark incubation is a critical stage for inducing dedifferentiation and cell proliferation; darkness significantly promotes callus formation in woody plants by suppressing photooxidation and maintaining endogenous hormone homeostasis[60,61]. Preliminary observations in this study revealed that single-layer covering materials exhibited insufficient moisture retention under field high-temperature conditions (complete desiccation of absorbent cotton within 2–3 d), whereas double-layer covering extended the moisture maintenance period to 5–8 d. Consequently, all treatments utilized the double-layer plastic covering system to ensure uniform microclimatic conditions across groups. Given that callus induction requires darkness, and field conditions cannot achieve the absolute darkness of culture rooms, this study employed the BP + WP covering treatment. This treatment exhibited superior performance compared to other covering approaches in both callus initiation rate and growth velocity, producing healthy callus morphology that established a robust foundation for subsequent differentiation.
Concurrently, the duration of dark incubation represents a critical determinant of callus competence for organogenesis. Specifically, prolonged dark incubation typically leads to excessive callus proliferation and reduces the potential for differentiation into adventitious buds, highlighting the importance of optimizing the duration of darkness treatment.[62]. Furthermore, prolonged incubation may cause carbon source depletion and ethylene accumulation, ultimately driving callus tissues into senescence or metabolic disorders[63]. Therefore, timely transfer to light conditions is essential for triggering redifferentiation. This study established dark incubation periods at 5-d intervals. Results demonstrated that although callus growth grades did not differ significantly between the 10-d and the 15-d treatments, the 10-d treatment achieved a significantly higher adventitious bud regeneration rate (76.7%) compared to the 15-d treatment, while substantially reducing the overall culture duration. Based on a comprehensive evaluation of regeneration efficiency and time cost, the 10-d dark incubation period was established as optimal.
Future perspectives
-
The present study establishes an efficient in vivo regeneration protocol based on phenotypic optimization. However, several mechanistic aspects need further investigation: (1) the direct effect of dark treatment on PPO activity and phenolic metabolism remains to be validated by enzymatic assays and metabolite profiling; (2) the precise mode of Ag+ action—whether through ethylene biosynthesis inhibition or signaling interference—was not elucidated through quantitative hormone analysis or gene expression studies; (3) the interaction between light and Ag+ treatment at the molecular level requires transcriptomic or proteomic approaches. Furthermore, this in vivo regeneration system will offer a promising foundation for advancing genetic transformation and mutagenesis breeding applications.
-
This study established an efficient in vivo callus induction and bud regeneration system for A. arguta. The optimal protocol comprised double-layer covering (WP inner, BP outer), optimal hormone concentration (5.0 mg/L ZT, 4.0 mg/L AgNO3), and optimal dark culture period (10 d), resulting in 76.7% bud regeneration. This method will offer a technically simpler and faster alternative to in vitro culture, addressing the current lack of in vivo regeneration systems for this species.
-
The authors confirm their contributions to the paper as follows: conceptualization, methodology: Ai J, Wang ZX; formal analysis: Wang DL, Xu ZX; data curation: Sun D, Shi GL, Wang DL, Li JX; writing − review and editing: Wang DL, Wang ZX; visualization: Zhao SY, Zhou X, Jin XM, Wang YX; funding acquisition: Wang ZX. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed in this study are included in this published article and supplementary information files.
-
We thank Professor Da-yong Fan and Yang Yu from the Beijing Forestry University and Beijing Academy of Agriculture and Forestry Sciences, for providing digitized data collection and methods. This study was supported by the Science and Technology Department of Jilin Province (20260204027YY), the Natural Science Foundation of China (to ZXW, GLS, and JA, 31870673), and the Province Development and Reform Commission (2022C037-1). This work complies with Chinese law.
-
The authors declare that they have no conflict of interest.
-
accompanies this paper online at: https://doi.org/10.48130/frures-0026-0007.
-
# Authors contributed equally: De-Li Wang, Zi-Xun Xu
- Supplementary Table S1 Effect of different combinations of growth regulators on callus formation and bud regeneration.
- Supplementary Table S2 The Effect of different cytokinin concentrations on callus induction and adventitious bud regeneration.
- Supplementary Fig. S1 The operation flow chart.
- Supplementary Fig. S2 Callus and adventitious bud formation at the graft union.
- Supplementary Fig. S3 Morphology of callus cultured under different growth regulators combinations, imaged at 20 days after treatment.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Wang DL, Xu ZX, Li JX, Sun D, Shi GL, et al. 2026. Establishment of an in vivo callus induction and bud regeneration system for Actinidia arguta . Fruit Research 6: e012 doi: 10.48130/frures-0026-0007
Establishment of an in vivo callus induction and bud regeneration system for Actinidia arguta
- Received: 04 February 2026
- Revised: 26 February 2026
- Accepted: 09 March 2026
- Published online: 31 March 2026
Abstract: Conventional in vitro tissue culture methods for fruit trees are often limited by the long duration from plantlet induction to field transplantation, the need to maintain sterile conditions, and high operational costs. To overcome these limitations, we optimized key factors—including covering materials, dark incubation duration, concentrations of plant growth regulators, and silver nitrate (AgNO3) levels—to establish a highly efficient in vivo system for callus induction and bud regeneration in Actinidia arguta. The optimized method involved applying 5.0 mg/L zeatin (ZT) and 4.0 mg/L AgNO3 to the cut surface, followed by covering it with a double-layer plastic bag (white inner layer and black outer layer) during a 10-d dark incubation. Subsequently, the double-layer covering was replaced with a white plastic bag for continued cultivation under light conditions, which effectively induced callus formation and adventitious bud regeneration. These results demonstrated that the system is simpler, lower cost, and more convenient. This method will provide a foundation for genetic transformation and polyploid breeding of Actinidia arguta.
-
Key words:
- In vivo /
- Callus induction /
- Bud regeneration /
- Actinidia arguta