-
Gastric cancer (GC) is one of the most prevalent malignant tumors that poses a significant challenge to global public health. According to the 2022 Global Cancer Statistics report published by the International Agency for Research on Cancer (IARC), GC ranks among the top five cancers worldwide in both incidence and mortality, with its mortality rate standing at third in China[1].
In recent years, with the advancement of minimally invasive surgical techniques, laparoscopic gastrectomy has become a widely accepted standard treatment in many centers around the world[2,3]. Compared to traditional open gastrectomy, this approach offers several advantages, including reduced surgical trauma, decreased intraoperative bleeding, diminished postoperative pain, lower rates of postoperative complications, and faster recovery[4−6].
However, laparoscopic surgery is a complex and challenging procedure, resulting in a relatively high incidence of postoperative complications, ranging from 10% to 25%[3,7]. The most common complications are pneumonia and paralytic ileus, etc. Gastric cancer surgery often involves reconstructing the digestive tract to varying degrees, which can lead to abdominal adhesions and inhibition of intestinal function, affecting gastrointestinal function[8]. Laparoscopic surgery may still cause postoperative complications due to the use of anesthetic drugs during the operation and the injection of carbon dioxide. Such complications may include prolonged ileus, constipation, vomiting, and nausea[9]. Therefore, effective nursing measures are essential to promote gastrointestinal function recovery, accelerate postoperative rehabilitation, and reduce complications in patients undergoing gastric cancer surgery.
The Enhanced Recovery after Surgery (ERAS) concept aims to alleviate perioperative stress responses, reduce postoperative complications, accelerate patient recovery, and improve postoperative quality of life[10,11]. Early mobilization (EM) is a crucial component of ERAS, which includes activities such as bed mobility, range-of-motion exercises, sitting at the edge of the bed, standing, transfers, and gait training[12]. It contributes to reducing the occurrence of complications such as pneumonia, pressure injuries, and deep vein thrombosis. Moreover, it can promote the recovery of intestinal function, reduce the risk of ileus, accelerate the patient's recovery, shorten the hospital stay, and lower the re-admission rate, and mortality rate[13−15].
Various guidelines[16,17] strongly recommend that patients with gastric cancer should start early activities 24−48 h after surgery, unless there are contraindications. But they do not offer detailed explanations regarding the frequency, type, and extent of the activities, which may not fully meet the unique requirements of early postoperative activities after laparoscopic gastric cancer surgery. This has led to a lack of detailed guidance in clinical nursing practices, and practice compliance remains low[18]. There is literature suggesting that the rate of EM completion for patients with gastric cancer ranges from 36% to 50%[18,19].
Therefore, this study conducted a comprehensive review of the relevant literature on EM after laparoscopic gastrectomy for gastric cancer, synthesizing the available scientific evidence. The goal was to provide healthcare professionals with the most reliable evidence regarding EM following laparoscopic gastrectomy for gastric cancer in clinical practice, thereby promoting the early rehabilitation of gastric cancer patients.
-
This study adopted the reporting specifications of the evidence summary report from the Fudan University Center for Evidence-based Nursing[20]. The registration number is 'ES20233813'.
Problem establishment
-
The PIPOST model was utilized to identify the evidence-based problem in this study[21]. The target population (P) consisted of patients aged 18 years or older who underwent laparoscopic gastrectomy for gastric cancer; intervention (I) was early postoperative mobilization; professionals (P) involved in applying evidence were clinical medical staff specializing in gastrointestinal surgery; outcome (O) was the implementation rate of EM by clinical medical staff, the time of first ambulation and first exhaust, as well as pain score; setting (S) was gastrointestinal surgical wards; the types of evidence (T) included guidelines, expert consensuses, clinical decision-making resources, systematic reviews, evidence summaries, and original research.
Literature retrieval
-
According to the pyramid '6S' evidence model[22], a comprehensive search was conducted across various databases such as UpToDate, National Guideline Clearinghouse (NGC), BMJ Best Practice, UK National Institute for Health and Clinical Excellence (NICE), Scottish Intercollegiate Guidelines Network (SIGN), Registered Nurses' Association of Ontario (RNAO), Joanna Briggs Institute Library, Chinese Clinical Guideline Database, Yi Maitong Guidelines Network, Cochrane Library, PubMed, Embase, Chinese Journal Full-text Database (CNKI), WanFang Database, and VIP Database. The keywords utilized in the search included terms such as: gastric cancer, stomach cancer, gastric carcinoma, stomach neoplasms, early motivation, early ambulation, early exercise, early mobilization, early mobility, accelerated ambulation, early rehabilitation, early activity, enhanced recovery after surgery, ERAS, and fast track surgery. The retrieval period was from the inception of the database up to August 4, 2025. Taking PubMed and CNKI as examples, the search strategies are shown in the Supplementary Tables S1 and S2.
Inclusion and exclusion criteria of literature
-
The inclusion criteria were as follows: (1) Participants were patients aged 18 years or older, who had undergone laparoscopic gastrectomy for gastric cancer; (2) The study focused on topics related to early postoperative mobilization; (3) The research type included guidelines, expert consensus, clinical decision-making, evidence summary, systematic review, and original research from both domestic and international sources; (4) The literature was written in either Chinese or English.
The exclusion criteria included: (1) Inability to access the full text of the study; (2) Duplicate publications; (3) Conference papers, abstracts, or interpretive literature guides; (4) Literature that did not meet established quality evaluation standards.
Literature screening
-
The literature was imported into Note Express for the purpose of identifying and removing any duplicate sources. Two researchers (Wei & Lyu), both trained in evidence-based practices, conducted individual assessments of the literature's quality. In cases of disagreement, a third researcher (Ni) was consulted to reach a consensus. Initial screening involved reviewing the title and abstract of the literature, followed by a more thorough examination of the full text to eliminate irrelevant sources. The quality of the literature that passed the screening process was subsequently assessed.
Quality evaluation of the literature
-
The Appraisal of Guidelines for Research and Evaluation (AGREE II) was utilized to evaluate the quality of the guidelines, encompassing six domains and a total of 23 items[23]. Guidelines with scores of six fields ≥ 60% were classified as A-level recommendations, while those with scores of 30% to 60% in three or more fields were categorized as B-level. A score of less than 30% in three or more fields was considered C-level. This study focused on guidelines classified as A- and B-level recommendations.
Expert consensus used The Australian JBI Evidence-Based Health Care Center for text and opinion papers for quality evaluation[24]. The evaluation of systematic reviews and original research was conducted using the assessment tools provided by the JBI Evidence-based Health Care Center in Australia[25].
Evidence extraction and summary
-
Two researchers (Wei & Lyu) trained in evidence-based medicine collected and summarized evidence according to the following principles of evidence integration: (1) Prioritizing professional, concise, and clear evidence when the content was similar; (2) Merging complementary evidence based on logical relationships when evidence from multiple sources was similar; (3) Resolving conflicting evidence by giving precedence to the most recent, authoritative and high-quality sources. The evidence-based team participated in the evidence summary process, and the evidence was assessed using the JBI Evidence Pre-Grading and Evidence Recommendation System (2014 version)[26]. This system classifies evidence into five levels based on the type of research design, ranging from level 1 (highest quality) to 5 (lowest quality). According to the FAME framework of JBI, the feasibility, appropriateness, meaningfulness, and effectiveness of the evidence was evaluated, and the evidence was categorized into grade A (strongly recommended), and grade B (weakly recommended).
-
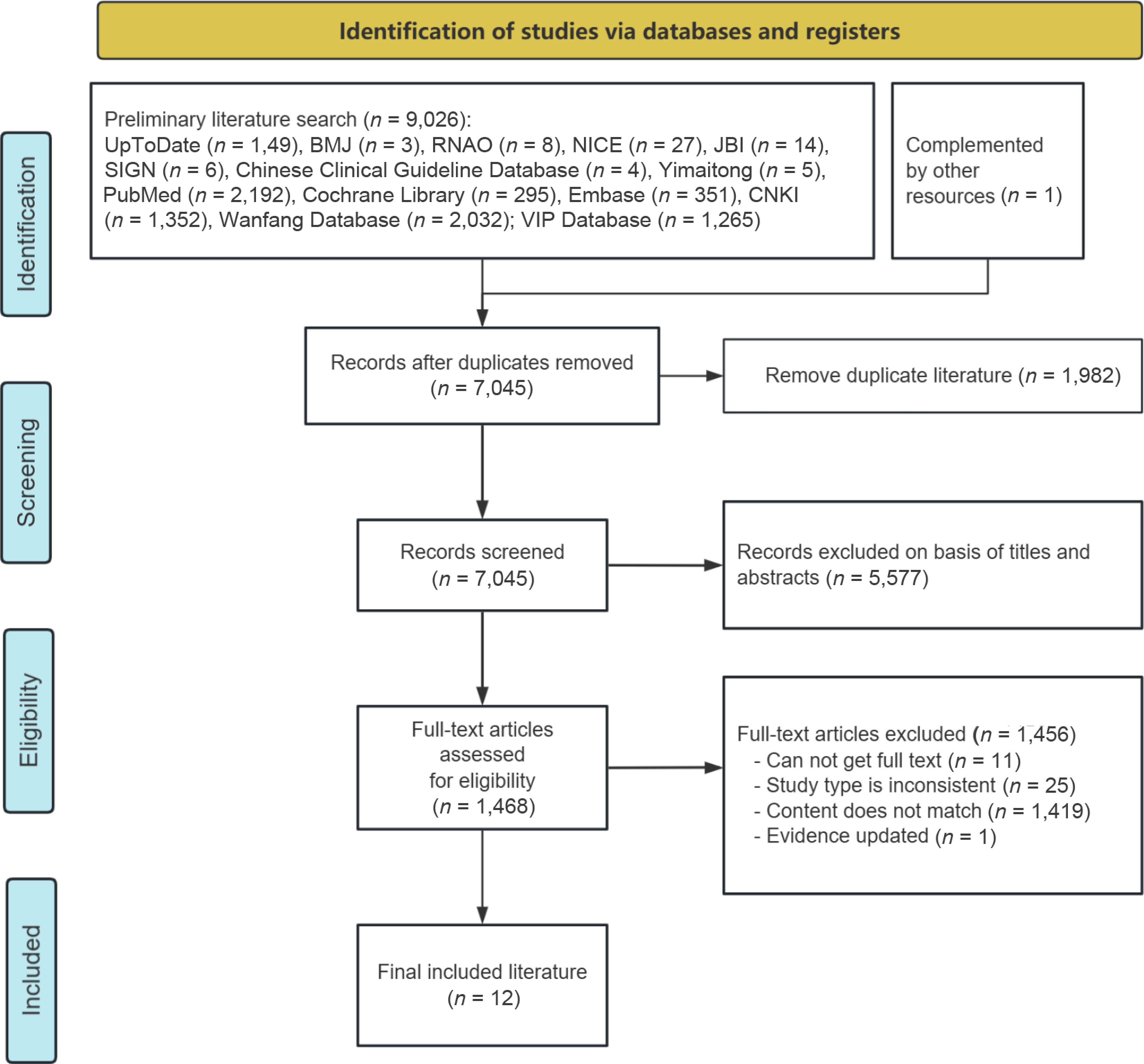
A total of 9,026 articles were retrieved in the study, with one article supplemented by additional resources. After excluding 1,982 duplicates and 5,577 with irrelevant titles and abstracts, 1,468 studies were selected for full-text screening. Finally, a total of 12 articles met the inclusion criteria, including three guidelines[16,27,28], five randomized controlled trials[29−33], two expert consensuses[34,35], one systematic review[36], and one quasi-experimental study[37]. The process of literature retrieval and screening is illustrated in Fig. 1, while the general characteristics of the included literature are detailed in Table 1.
Table 1. General characteristics of the included articles (n = 12).
Author Year Country Article type Article source Research theme Mortensen et al.[16] 2014 Norway, Sweden, France, Switzerland, Italy, Canada, UK Guideline Yimaitong Consensus guidelines for enhanced recovery after gastrectomy Irani et al.[27] 2023 The United States Guideline PubMed Clinical practice guidelines for enhanced recovery after colon and rectal surgery from the American society of colon and rectal surgeons and the society of American gastrointestinal and endoscopic surgeons Chen et al.[28] 2018 China Guideline Yimaitong Chinese consensus and clinical guidelines for enhanced recovery after surgery (2018 edition) Liu et al.[29] 2018 China RCT CNKI Implementation of quantitative ambulation schemes in enhanced recovery after surgery for gastric cancer patients Ni et al.[30] 2023 China RCT CNKI The application of enhanced recovery after surgery nursing pathway in laparoscopic radical gastrectomy for gastric cancer Lee et al.[31] 2025 Korea RCT PubMed Effectiveness of ERAS program on postoperative recovery after gastric cancer surgery: a randomized clinical trial He et al.[32] 2022 China RCT Cochrane Library Fast-track surgery nursing intervention in CRC patients with laparotomy and laparoscopic surgery Gao et al.[33] 2020 China RCT Cochrane Library Effect of fast-track surgery nursing on postoperative rehabilitation of patients undergoing laparoscopic radical gastrectomy Association[34] 2017 China Expert consensus Yimaitong Expert consensus on enhanced recovery after gastrectomy for gastric cancer (2016 edition) Surgery[35] 2016 China Expert consensus Yimaitong Chinese expert consensus on enhanced recovery after surgery in perioperative management (2016 edition) Tian et al.[36] 2024 China Systematic review PubMed The efficacy and safety of enhanced recovery after surgery (ERAS) Program in laparoscopic distal gastrectomy: a systematic review and meta-analysis of randomized controlled trials Hu et al.[37] 2021 China Quasi-experimental study CNKI Application of best evidence for early postoperative ambulation in gastric cancer patients Note: RCT, randomized controlled trial. Quality evaluation results of the included literature
-
The quality evaluation results of the 12 articles included are shown in Tables 2−6. Given the overall high quality of the 12 articles, they were all included in the summary.
Table 2. Quality evaluation results of clinical guidelines (n = 3).
Guideline Standardized scores in various domains (%) ≥ 60% ≥ 30% Quality evaluation Scope and purpose Stakeholder involvement Rigour of development Clarity of presentation Applicability Editorial independence Mortensen et al.[16] 86.08% 83.33% 72.92% 88.83% 75% 66.67% 6 6 A Irani et al.[27] 91.67% 61.17% 93.79% 88.89% 41.67% 75% 5 6 B Chen[28] 91.67% 88.89% 90.63% 91.67% 33.33% 66.67% 5 6 B Table 3. Quality evaluation results of expert consensus (n = 2).
Items Association[34] Surgery[35] 1. Is the source of the opinion clearly identified? Yes Yes 2. Does the source of opinion have standing in the field of expertise? Yes Yes 3. Are the interests of the relevant population the central focus of the opinion? Yes Yes 4. Is the stated position the result of an analytical process, and is there logic in the opinion expressed? Yes Yes 5. Is there reference to the extant literature? Yes Yes 6. Is any incongruence with the literature/sources logically defended? Yes Yes Table 4. Quality evaluation results of randomized controlled trials (n = 5).
Items Liu et al.[29] Ni et al.[30] Lee et al.[31] He et al.[32] Gao et al.[33] 1. Was true randomization used for assignment of participants to treatment groups? Yes Yes Yes Yes Yes 2. Was allocation to treatment groups concealed? Unclear Unclear Yes Unclear Unclear 3. Were treatment groups similar at the baseline? Yes Yes Yes Yes Yes 4. Were participants blind to treatment assignment? Unclear Unclear Unclear Unclear Unclear 5. Were those delivering the treatment blind to treatment assignment? Unclear Unclear No Unclear Unclear 6. Were treatment groups treated identically other than the intervention of interest? Yes Yes Yes Yes Unclear 7. Were outcome assessors blind to treatment assignment? Unclear Unclear Yes Unclear Unclear 8. Were outcomes measured in the same way for treatment groups? Yes Yes Yes Yes Yes 9. Were outcomes measured in a reliable way? Yes Yes Yes Yes Yes 10. Was follow-up complete and, if not, were differences between groups in terms of their follow-up adequately described and analyzed? Yes Yes Yes Yes Yes 11. Were participants analyzed in the groups to which they were randomized? Yes Yes Yes Yes Yes 12. Was appropriate statistical analysis used? Yes Yes Yes Yes Yes 13. Was the trial design appropriate and any deviations from the standard RCT design (individual randomization, parallel groups) accounted for in the conduct and analysis of the trial? Yes Yes Yes Yes Yes Table 5. Quality evaluation results of systematic review (n = 1).
Items Tian et al.[36] 1. Is the review question clearly and explicitly stated? Yes 2. Were the inclusion criteria appropriate for the review question? Yes 3. Was the search strategy appropriate? Yes 4. Were the sources and resources used to search for studies adequate? Yes 5. Were the criteria for appraising studies appropriate? Yes 6. Was critical appraisal conducted by two or more reviewers independently? Yes 7. Were there methods to minimize errors in data extraction? Yes 8. Were the methods used to combine studies appropriate? Yes 9. Was the likelihood of publication bias assessed? Yes 10. Were recommendations for policy and/or practice supported by the reported data? Yes 11. Were the specific directives for new research appropriate? Yes Table 6. Quality evaluation results of quasi-experimental study (n = 1).
Items Hu et al.[37] 1. Is it clear in the study what is the 'cause' and what is the 'effect' (i.e., there is no confusion about which variable comes first)? Yes 2. Were the participants included in any comparisons similar? Yes 3. Were the participants included in any comparisons receiving similar treatment/care, other than the exposure or intervention of interest? Yes 4. Was there a control group? Yes 5. Were there multiple measurements of the outcome both pre, and post the intervention/exposure? Yes 6. Was follow up complete and if not, were differences between groups in terms of their follow up adequately described and analyzed? Yes 7. Were the outcomes of participants included in any comparisons measured in the same way? Yes 8. Were outcomes measured in a reliable way? Yes 9. Was appropriate statistical analysis used? Yes Summary and description of evidence
-
Two researchers extracted and synthesized the gathered evidence, resulting in the identification of 28 pieces of the best evidence across seven aspects: preoperative education, postoperative pain management, postoperative drainage tube management, pre-mobilization evaluation, early postoperative bed activity, early postoperative off-bed mobilization, and discharge criteria, as shown in Table 7.
Table 7. Evidence summary for EM after laparoscopic gastrectomy of gastric cancer.
Category Content of evidence Evidence Level Recommendation grade Preoperative education 1. Nurses took the initiative to communicate with patients enthusiastically upon the patients' admission, and informed patients of relevant precautions through health manuals, videos, posters, and words, so as to make patients ready for the operation psychologically, with optimism and good mentality[33]. 1 A 2. For different patients, cards, multimedia, exhibition boards, and other forms were used to introduce the perioperative process of anesthesia, surgery, and postoperative treatment, so as to relieve their anxiety, fear, and tension, making patients aware of their important role in this plan, and obtain the understanding and cooperation of patients and their families, including early postoperative feeding and early ambulation[28]. 5 A 3. Health care workers should introduce the anesthesia and operation process to patients and their families in detail by oral or written form before the operation. Inform the purpose and main items of the ERAS program to relieve patients ' tension and anxiety. Strive for the understanding and cooperation of patients and their families to promote rapid postoperative recovery[34,35]. 5 A Postoperative pain management 4. The postoperative pain intensity in patients was evaluated using either the Visual Analog Scale (VAS) or the Numerical Rating Scale (NRS)[31,33]. 1 A 5. If the pain score was less than 3, distract the patient's attention by chatting, listening to music, cold compress, hot compress, and other forms to relieve the pain. If the pain score was greater than 3, followed the doctor's advice to give analgesic treatment. For patients with a pain score of 7 or higher, 50 µg of IV fentanyl was administered as the first-line rescue analgesic[31,33]. 1 B 6. Multimodal analgesia is recommended. The important principle of analgesia is that non-steroidal anti-inflammatory drugs (NSAIDs) are the basic drugs for postoperative analgesia, and the application of opioids should be minimized[34,35]. 5 B 7. For patients undergoing laparoscopic gastric surgery, transversus abdominis plane nerve block and local infiltration of incision can be selected for analgesia[28]. 5 B Postoperative drainage tube management 8. No routine use of nasogastric tube during operation. If necessary, it should be retained during operation and removed within 24 h after operation[28,34,35]. 5 B 9. No routine indwelling intra-abdominal drain is required. After total gastrectomy and proximal gastrectomy, intra-abdominal drains can be retained while in other cases they should be removed 2−3 d after the operation[28]. 1 B 10. No special case, the urinary catheter should be removed 1−2 d after gastric surgery[16,28,31,34−36]. 5 B Pre-mobilization evaluation 11. If the vital signs are stable, the patients should get out of bed within 24 h after laparoscopic surgery[30,32]. 1 A 12.In the absence of nausea, dizziness, active bleeding, or unbearable incisional pain, the patient may be assisted to stand beside the bed for 5−10 min. Once no symptoms of orthostatic intolerance (including dizziness, palpitations, and blurred vision) are present, the patient may ambulate alongside the bed with assistance from a nurse or family member[29,33]. 1 A 13. If the pain score was less than 3 points, guide the patient to get out of bed correctly to protect the wound[30,37]. 1 A Early postoperative bed activity 14. After regaining consciousness postoperatively, patients may assume a semi-recumbent position or engage in appropriate in-bed activity; supine positioning without a pillow for 6 h is not required[28,34]. 5 A 15. After regaining consciousness from anesthesia, the patient is encouraged to mobilize in bed, and reposition every 2 h[30,32]. 1 A 16. Within 2 h after the operation, the abdominal belt was applied and the heel was raised by 15~20° with a small pillow. After the anesthesia, fully awake, and with normal vital signs, the head of the bed was raised by 30~45°. Assist the patient to carry out passive activities of both lower limbs. The massage included lower limb muscle massage, 3~5 min on each side, from the distal to the proximal massage, 3 times a day. Passive flexion and extension, internal and external rotation of ankle joint were performed, 15−20 times in each group, 3 groups per day[29]. 1 A 17. Two to 4 h after operation, patients were guided and assisted in abdominal breathing and lip contraction breathing[29]. 1 A 18. Four to 6 h after the operation, the head of the bed was raised by 45~60°, and the patient was assisted to turn over twice. Guide the patient to perform active muscle contraction and joint activities on the bed[29]. 1 B 19. Six to 12 h after operation, no fatigue or mild fatigue, and no active bleeding, the head of bed was raised to 90° sitting position for 3 min. If there is no obvious discomfort, assist to complete the bed-to-sit-up, three times. The sitting time was 10, 15, and 20 min respectively[29,30]. 1 B Early postoperative off-bed mobilization 20. On the day of surgery, the patients were allowed to sit on the edge of the bed and move if they desired[31]. 1 A 21. One day after operation, the patients began to get out of bed. Early and scheduled mobilization[16,28,34−36]. 1 A 22. One day after operation, the patient was allowed to get out of bed and move around for 1 to 2 h. Subsequently, they were allowed to get out of bed and move around for 4 to 6 h each day[28,30]. 1 B 23. The sequence of activities is: sitting up beside the bed for 3 min, standing beside the bed for 3 min, walking with support of the bed, and then walking away from the bed[30,37]. 1 A Early postoperative off-bed mobilization 24. Two days after operation, the patients were assisted to move in the corridor area of the ward[29,37]. 1 B 25. The walking distance was 25 to 50 m on the first day after operation, and gradually increased to 50 to 100 m when discharged[28,37]. 2 A 26. Daily check the patient 's activity completion. Establish a patient 's activity diary, and those who have not completed are urged to continue to complete[29]. 1 A Discharge criteria 27. (1) Tolerance of soft blended diet for 24 h, (2) safe ambulation until 600 m without assistance, (3) adequate pain control (NRS ≤ 3) with oral non-opioid analgesics, (4) the wound healed well, no infection, and (5) no abnormal physical findings or laboratory tests[27,28,31,34]. 1 B 28. There were no postoperative complications (e.g., anastomotic leak, bleeding, infection) and other basic diseases that needed to be treated. The patient could be discharged about 7 d after gastric surgery[28]. 5 B -
Early mobilization is a crucial aspect of the ERAS protocol concept, with significant implications for preserving muscle function, promoting gastrointestinal function recovery, and preventing complications. Although there is extensive research supporting the benefits of EM, limited studies have focused on the initiation time and management of the mobilization process[38]. The ERAS guidelines recommended that the patient should get out of bed for 1 to 2 h on the first day after surgery, and 4 to 6 h a day until discharge[27,28], but this does not include standard monitoring and assessment. There is still a lack of structured and universally accepted EM protocols. This study integrates detailed evidence-based guidance on pre-mobilization assessment, structured in-bed and off-bed activity protocols. To enhance adherence to EM practices following laparoscopic gastrectomy for gastric cancer, this study has extracted and summarized the most compelling evidence across seven key aspects. It advocates for healthcare professionals to enhance patient health education, foster interdisciplinary cooperation, and effectively manage early postoperative activities and discharge procedures to improve the quality of clinical care, and promote the rehabilitation of patients.
Preoperative education is the core of enhancing patients' adherence to early mobilization
-
Preoperative education is a systematic process that leads to changes in patients' knowledge, attitudes, and behaviors[39]. Evidence 1−3 summarizes the methods and content of preoperative education for laparoscopic gastric cancer. The complexity of the procedure, patients' unfamiliarity with the operating room, fear of the operation, and initial resistance can lead to a strong psychological and physiological stress response in patients. This stress response can impact the safety of the surgery and the prognosis of the patients. Effective preoperative education plays a significant role in helping patients understand the upcoming surgical procedure, reducing psychological pressure, and fostering active cooperation with surgical and nursing teams. Utilizing videos, pictures, and texts to introduce patients to the surgical plan, treatment process, and perioperative care can help alleviate patient anxiety[40]. Furthermore, preoperative patient education can provide patients with health information and opportunities to learn certain skills before surgery[41]. Providing patients with preoperative guidance on how to participate in early postoperative activities can significantly improve their adherence to postoperative activity protocols.
Currently, clinical preoperative education materials are developed separately for each department. The information is fragmented, one-sided, and lacks strong coherence. Most of it is available in oral and paper formats, making the educational process non-intuitive and dull. It is recommended to transition towards a multimedia educational approach that incorporates animation, sound, and text to emphasize visual and auditory effects. This approach can vividly explain important points of knowledge and preoperative preparations, enabling patients to better comprehend and engage with the educational content. The hospitalization of gastric cancer patients involves various interdisciplinary fields, including surgery, nursing, anesthesiology, and nutrition. Therefore, there is a necessity to enhance interdisciplinary communication to develop a more comprehensive and multidisciplinary preoperative education strategy.
Effective pain management is a crucial prerequisite for early postoperative mobilization
-
The surgery can cause varying degrees of acute pain, which may adversely affect a patient's comfort, health, and healing. Effective pain management is essential to alleviate discomfort, promote EM, facilitate rehabilitation, and prevent complications associated with inadequate pain control[42]. The management of pain following laparoscopic gastric cancer surgery was discussed in Evidence 4−7. The initial step in pain management involves assessing the pain, which is essential for evaluating the effectiveness of analgesic interventions. Both Visual Analog Scale (VAS), and Numeric Rating Scale (NRS) are widely used tools at national and international levels due to their simplicity, convenience, and accuracy. They have been recommended as assessment tools in guidelines for managing postoperative pain[43,44]. These tools are suitable for adult patients who are conscious and able to effectively communicate. Patients with a score greater than three were provided with analgesic treatment as recommended by the physician.
Implementing postoperative strategies to alleviate pain can promote EM and gastrointestinal function recovery, reduce complications, and improve patient satisfaction[45]. Multimodal analgesia is anticipated to improve the analgesic effect and minimize adverse effects, leading to lower pain scores and decreased opioid consumption[46,47]. Research indicates that patient-controlled intravenous analgesia (PCIA) as a standalone method following laparoscopic gastrectomy can effectively address pain and facilitate the restoration of gastrointestinal function[45]. Acetaminophen (Ace) has been commonly utilized for postoperative pain management, and its combination with PCIA has shown effectiveness in reducing postoperative pain, promoting gastrointestinal function recovery, and decreasing postoperative nausea and vomiting incidents[48−50]. The combination of PCIA with Ace is recognized as a promising emerging approach for pain management following laparoscopic surgery.
Reasonable placement of the drainage tube is the basis for promoting early mobilization
-
The retention of catheters is one of the main factors hindering early mobilization[51]. It may increase the risk of EM and the level of fear among patients. The synthesis of Evidence 8−10 focuses on the retention of drainage tubes following laparoscopic gastric cancer surgery. Evidence 8 is: 'No routine use of nasogastric tube during operation. If necessary, it should be retained during operation and removed within 24 h after operation'. A meta-analysis of patients undergoing colorectal surgery revealed that avoiding gastrointestinal decompression after surgery could decrease the incidence of pharyngitis and respiratory tract infections, although it might lead to vomiting[52]. Another study illustrated that refraining from nasogastric tube insertion after abdominal surgery could accelerate the recovery of gastrointestinal function and reduce pulmonary complications[53]. In clinical practice, the decision to insert a nasogastric tube should be based on individual patient circumstances. Evidence 9 states: 'No routine indwelling intra-abdominal drain is required. After total gastrectomy and proximal gastrectomy, intra-abdominal drains can be retained, while in other cases they should be removed 2−3 d after the operation'. Research has indicated that the placement of abdominal drainage tubes does not lower postoperative complications, mortality, or leaks[27]. However, in cases where patients are at risk of anastomotic leakage due to various factors, such as blood supply, tension, infection, or unsatisfactory anastomosis, it is advisable to insert an abdominal drainage tube[54]. The decision to place an abdominal drainage tube should be made by the operating surgeons based on the patient's clinical condition. Evidence 10 states: 'No special case, the urinary catheter should be removed 1−2 d after gastric surgery'. Prolonged catheter retention is associated with an increased risk of urinary tract infection, catheter-related trauma, and urinary retention[55], which can delay EM. It has been confirmed that early removal of the catheter in patients undergoing thoracic and abdominal surgery can reduce the risk of urinary tract infection[56]. Similarly, in patients undergoing laparoscopic gastric cancer surgery, it is deemed safe to remove the catheter within 24 h post-surgery[57]. Indwelling a catheter does not reduce complications and may even be harmful. Therefore, it is advisable to remove the catheter as soon as possible after operation.
Gradually implement the practice of early postoperative mobilization
-
Evidence 11−26 summarizes the contents of early pre-mobilization evaluation, in-bed activity, and off-bed mobilization following laparoscopic gastric cancer surgery. A more comprehensive standard for assessing getting-out-of-bed can improve patient adherence with EM post-operation. Given the complexity of gastric surgery and the individual variabilities among patients, it is crucial to conduct a thorough evaluation of each patient's specific circumstances when implementing accelerated rehabilitation. This approach should prioritize patient safety as the primary goal[17]. Patients undergoing surgery are advised to engage in bed activities once they have recovered from anesthesia and should change positions every 2 h[32]. Early bed exercises include breathing exercises, ankle exercises, muscle contraction training, massage, and sitting up in bed. These activities are beneficial in reducing the risk of pulmonary complications and deep vein thrombosis, as well as in promoting gastrointestinal function recovery[40]. Evidence 21 states: 'One day after the operation, the patients began to get out of bed. Early and scheduled mobilization'. EM is an essential component of ERAS protocols, as it can effectively promote the recovery of intestinal paralysis, enhance gastrointestinal motility, and prevent intestinal obstruction. Studies have shown that walking within 24 h after surgery significantly reduces the incidence of severe postoperative complications, shortens hospital stays, and improves prognosis[58]. According to the patient's tolerance, off-bed mobilization should be conducted, and excessive activity should be avoided[33]. Daily activities should be planned and implemented based on the patient's specific condition, with the establishment of a diary to monitor their progress. EM practices should be systematically implemented across three key areas: pre-activity evaluation, early bed activity, and off-bed mobilization.
Clear discharge criteria are important indicators for evaluating recovery outcomes
-
The discharge criteria following laparoscopic gastric cancer surgery are outlined in Evidence 27 and 28. It is imperative to establish precise discharge standards and daily activity goals. Establishing distinct discharge goals is a crucial indicator for evaluating the effectiveness of EM and patient rehabilitation. Guidelines recommend that discharge criteria should be measurable and actionable, with a primary focus on ensuring patient safety[28]. The discharge criteria included the absence of intravenous therapy, lack of postoperative complications, oral intake tolerance, oral analgesics use, satisfactory wound healing, independent ambulation, and patient consent. Patients become eligible for discharge upon meeting these criteria. Subsequent to hospital discharge, active patient follow-up is essential.
Scientific implementation of early mobilization protocol
-
The implementation of EM remains uncommon in clinical settings due to various barriers. Some of these barriers encompass patient refusal, pain, catheter insertion, delayed screening for eligibility, absence of EM protocol, and no eligibility criteria[59,19]. Identifying the barriers to EM and formulating effective strategies to address them can facilitate the integration of EM into routine clinical practice by healthcare professionals. This study systematically summarized an EM protocol covering seven aspects: preoperative education, postoperative pain management, postoperative drainage tube management, pre-mobilization evaluation, early postoperative bed activity, early postoperative off-bed mobilization, and discharge criteria. The failure to meet the EM targets might mainly be due to a lack of awareness and education about the benefits of EM. Early mobilization through patient education and encouragement is an important component of ERAS programs[60]. Relatively simple intervention related to education can significantly enhance the compliance with EM[61]. Early postoperative mobilization is the core principle of ERAS, and some care elements are also auxiliary measures to encourage EM, including measures related to optimal postoperative pain management and early removal of urinary catheters[62]. Furthermore, the present EM protocol provides safety criteria for inside and outside bed mobilization, various physiological considerations, as well as termination criteria, which can be applied in safe practices. Based on the pre-mobilization evaluation criteria, a suitable EM plan and the correct dosage was adopted for all eligible patients.
Limitations
-
This study has several limitations. The search was restricted to Chinese and English databases, excluding literature in other languages. Additionally, some randomized controlled trials (RCTs) included in this review did not clearly report allocation concealment or blinding procedures, which may introduce potential bias. Future high-quality trials with rigorous methodological reporting are needed to strengthen the evidence base. The literature reviewed in this study only represents a portion of the evidence concerning colorectal and gastric cancer surgeries. Future studies should prioritize patients undergoing laparoscopic procedures to generate optimal evidence for EM. Moreover, this study primarily focused on clinical outcomes and did not systematically incorporate patient-reported outcome measures (PROMs), such as quality of life, satisfaction, or psychological status. Future evidence summaries and clinical studies should include PROMs to provide a more patient-centered evaluation of EM interventions. Furthermore, the majority of included studies were conducted in China, with a smaller proportion from international settings. Differences in healthcare systems, resources, and cultural attitudes toward mobilization may affect the transferability of these findings. Clinicians should adapt the recommendations to local contexts, particularly when applying ERAS principles across diverse geographic and resource settings.
-
In accordance with the evidence summary framework developed by the Fudan University Center for Evidence-based Nursing, this study summarized 28 pieces of the best evidence of EM in patients with laparoscopic gastric cancer from seven aspects: preoperative education, postoperative pain management, postoperative drainage tube management, pre-mobilization evaluation, early postoperative bed activity, early postoperative off-bed mobilization, and discharge criteria. The findings of this study provide a foundational framework for healthcare practitioners involved in the care of patients undergoing laparoscopic radical gastrectomy. However, due to the diverse origins of the evidence, it is essential to assess its applicability within specific clinical contexts before implementation. Future research should focus on conducting RCTs to validate the clinical applicability and generalizability of this evidence-based protocol across different healthcare settings and patient populations. Additionally, it is important to identify the facilitators and barriers to its implementation and to promote adherence to clinical guidelines.
-
This study is a review of publicly available literature and does not involve human or animal participants; therefore, ethical approval was not required. No AI tools were used during the preparation of this work.
-
The authors confirm their contributions to the paper as follows: conceptualization, methodology, data curation, formal analysis, writing − original draft: Wei R; methodology, data curation, formal analysis, writing − review and editing: Lyu M; methodology, writing − original draft preparation: Chen M; writing − original draft preparation: Ruan C; conceptualization, writing − review and editing, supervision, project administration: Ji M; conceptualization, data curation, validation, writing − review and editing, supervision, project administration: Ni J. All authors have read and approved the final manuscript.
-
All data generated or analysed during this study is included in this published article.
-
This work was supported by the Clinical Project of Nursing Department of Shanghai Jiao Tong University School of Medicine Affiliated Renji Hospital (Grant No. hlbynkt2023-003); 2022 Minhang District Natural Science Research Project (Grant No. 2022MHZ061). The funders had no role in the study design, collection, analysis, interpretation of data, writing of the report, or decision to submit the article for publication.
-
The authors declare that they have no conflict of interest.
-
# Authors contributed equally: Rong Wei, Mengmeng Lyu
- Supplementary Table S1 PubMed search strategy.
- Supplementary Table S2 CNKI search strategy.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Wei R, Lyu M, Chen M, Ruan C, Ji M, et al. 2026. Best evidence summary of early mobilization after laparoscopic gastrectomy for gastric cancer. Gastrointestinal Tumors 13: e006 doi: 10.48130/git-0026-0002
Best evidence summary of early mobilization after laparoscopic gastrectomy for gastric cancer
- Received: 29 August 2025
- Revised: 04 December 2025
- Accepted: 02 February 2026
- Published online: 31 March 2026
Abstract: Early mobilization is beneficial for the rehabilitation of patients undergoing laparoscopic gastric cancer surgery, but the clinical practice remains poor. This study aims to summarize the best evidence for early mobilization of patients following laparoscopic gastrectomy and to develop a theoretical framework to direct clinical practice. The available literature concerning early mobilization following laparoscopic gastric cancer surgery was systematically searched using 15 databases. The literature encompassed guidelines, expert consensuses, evidence summaries, best clinical practices, clinical decision-making resources, systematic reviews, and high-quality original research, from inception to August 4, 2025. Two researchers conducted the quality assessment and evidence extraction process. Twelve articles were included, comprising three guidelines, five randomized controlled trials, two expert consensuses, one systematic review, and one quasi-experimental study. A total of 28 pieces of evidence were summarized across seven aspects: preoperative education, postoperative pain management, postoperative drainage tube management, pre-mobilization evaluation, early postoperative bed activity, early postoperative off-bed mobilization, and discharge criteria. This evidence summary highlights the effective strategies for managing early mobilization following laparoscopic gastric cancer surgery, which could provide guidance for clinical practice. The framework offers a comprehensive implementation strategy for healthcare professionals and has the potential to improve early postoperative rehabilitation outcomes through its effective clinical application.
-
Key words:
- Gastric cancer /
- Laparoscopic gastrectomy /
- Early mobilization /
- Evidence summary