-
Although the majority of cerebral infarctions stem from conventional mechanisms such as atherosclerosis or cardioembolism, malignancy is an important yet often underrecognized or overlooked etiology in clinical practice. Recent research has advocated for the recognition of "cancer-related stroke" as a distinct etiologic subtype within stroke classification systems to enhance clinical vigilance and promote targeted investigations[1]. This entity is typically defined by three criteria: The absence of a definite conventional cause, a diagnosis of malignancy within 6 months of the stroke's onset, and no history of anticancer treatment prior to the stroke event[2]. It is estimated that approximately 5%−10% of stroke patients harbor an occult systemic malignancy (OSM)[3], which refers to a noncentral nervous system malignancy first detected during the stroke work-up, lacking clinically evident warning signs like deep vein thrombosis[2,4]. Although the association between stroke and cancer is established, cases where acute multiple cerebral infractions (AMCIs) serve as the initial manifestation of multiple primary malignant neoplasms (MPMNs) are exceedingly rare.
Among solid tumors, gastrointestinal adenocarcinoma carries the highest risk of cancer-associated thrombosis, with hypercoagulability being a key pathophysiological mechanism underlying cryptogenic stroke in cancer patients[5]. Furthermore, MPMN itself—defined as the occurrence of two or more distinct histologically confirmed malignancies in a single patient, either synchronously or metachronously—is an uncommon phenomenon. Its reported incidence varies widely[6−8]. MPMNs frequently arise within cognate systems or paired organs, making the co-occurrence of a gastrointestinal adenocarcinoma and a hematologic malignancy, such as plasmacytoma, an even rarer clinical entity.
Here, we present a singular case of synchronous ileocecal adenocarcinoma and solitary bone plasmacytoma, which initially manifested as AMCI. This case embodies three rare features: Cryptogenic stroke, MPMN, and the coexistence of gastrointestinal and hematologic malignancies. We aim to heighten the clinical awareness that stroke can be the inaugural sign of underlying occult malignancies, and to elucidate the complex pathophysiological interplay, diagnostic challenges, and therapeutic dilemmas present in such cases.
-
A 74-year-old man was admitted to a local hospital following the acute onset of right-sided limb weakness. Brain magnetic resonance imaging (MRI)–diffusion-weighted imaging (DWI) revealed an AMCI involving bilateral anterior and posterior circulations (Fig. 1). Initial laboratory investigations demonstrated an elevated serum carcinoembryonic antigen (CEA) level of 12.68 ng/mL (normal range: 0−5.2 ng/mL) and a significantly reduced hemoglobin level of 88 g/L (normal range: 130−175 g/L), indicative of anemia and potential malignancy.
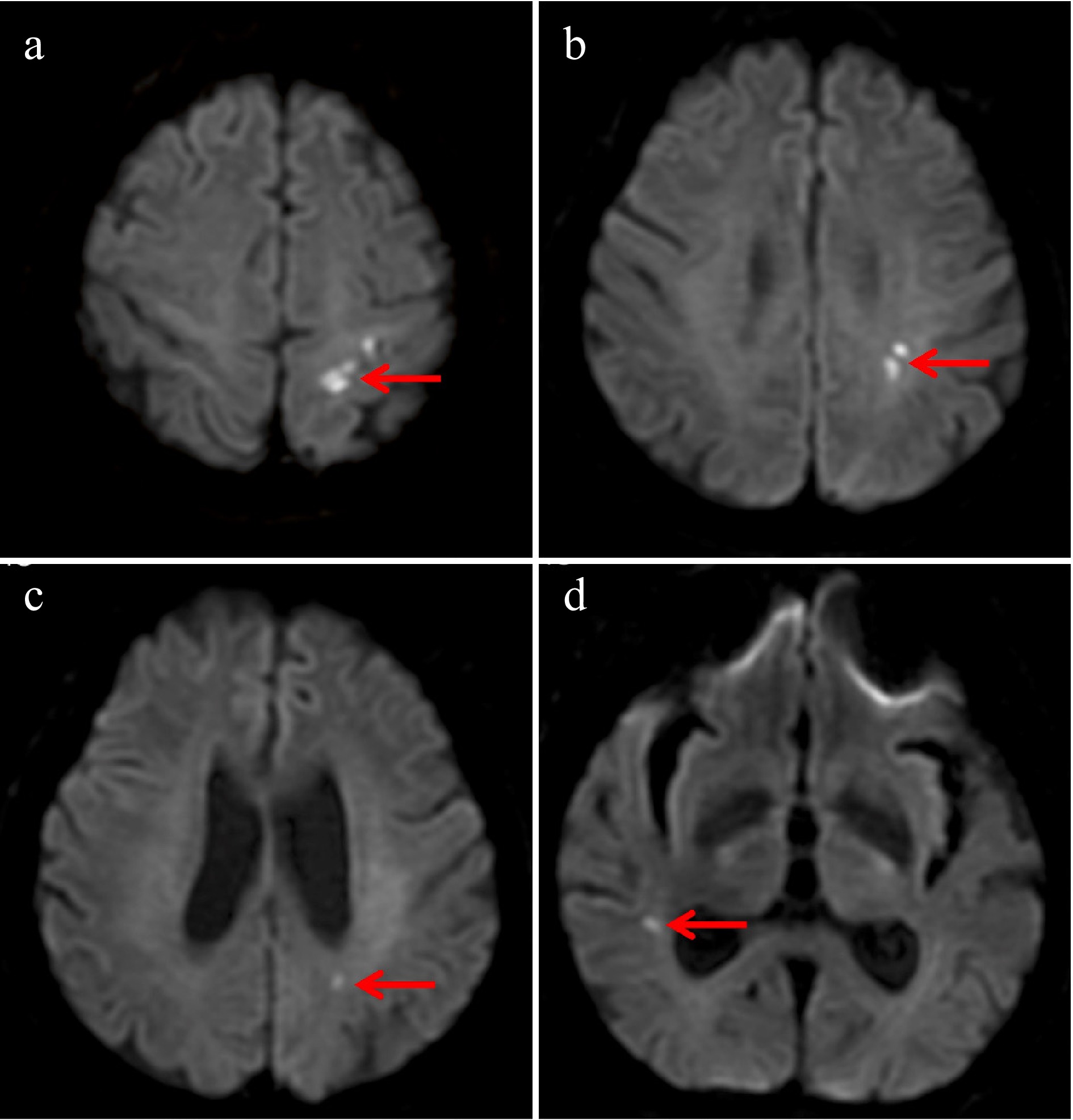
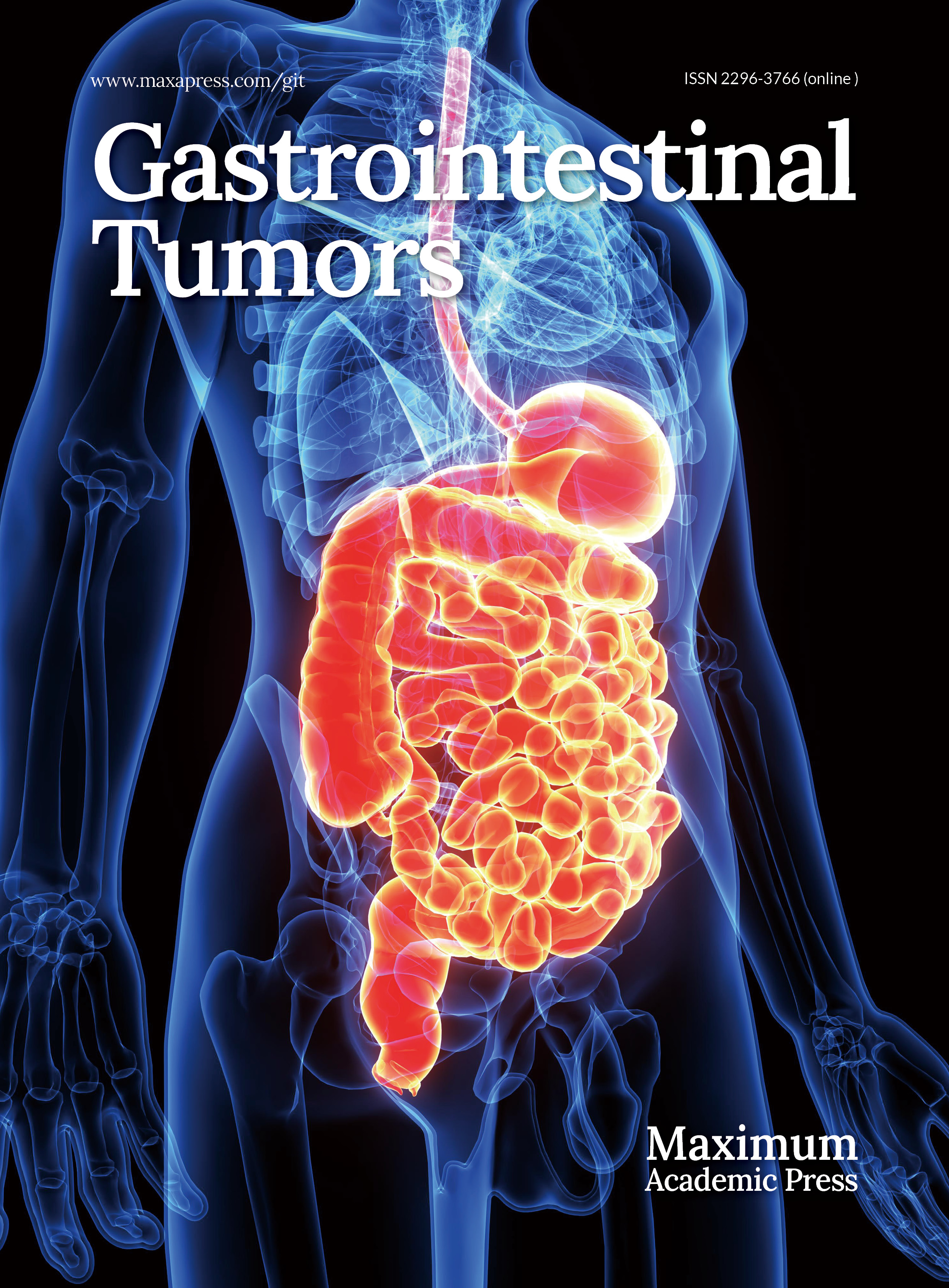
Figure 1.
Brain magnetic resonance imaging–diffusion-weighted imaging (MRI-DWI) demonstrated multiple cerebral infarctions involving both the anterior and posterior circulations. Acute infarction in (a) the left anterior cerebral artery (ACA) region, (b) the left middle cerebral artery (MCA) region, (c) the left posterior cerebral artery (PCA) region, and (d) the right MCA region.
To elucidate the underlying etiology, contrast-enhanced computed tomography (CT) of the chest and abdomen was performed. The scans revealed irregular wall thickening in the ileocecal region (Fig. 2b) and a concomitant destructive mass in the left pleural cavity with adjacent rib erosion (Fig. 3a, b), both highly suggestive of malignant processes.
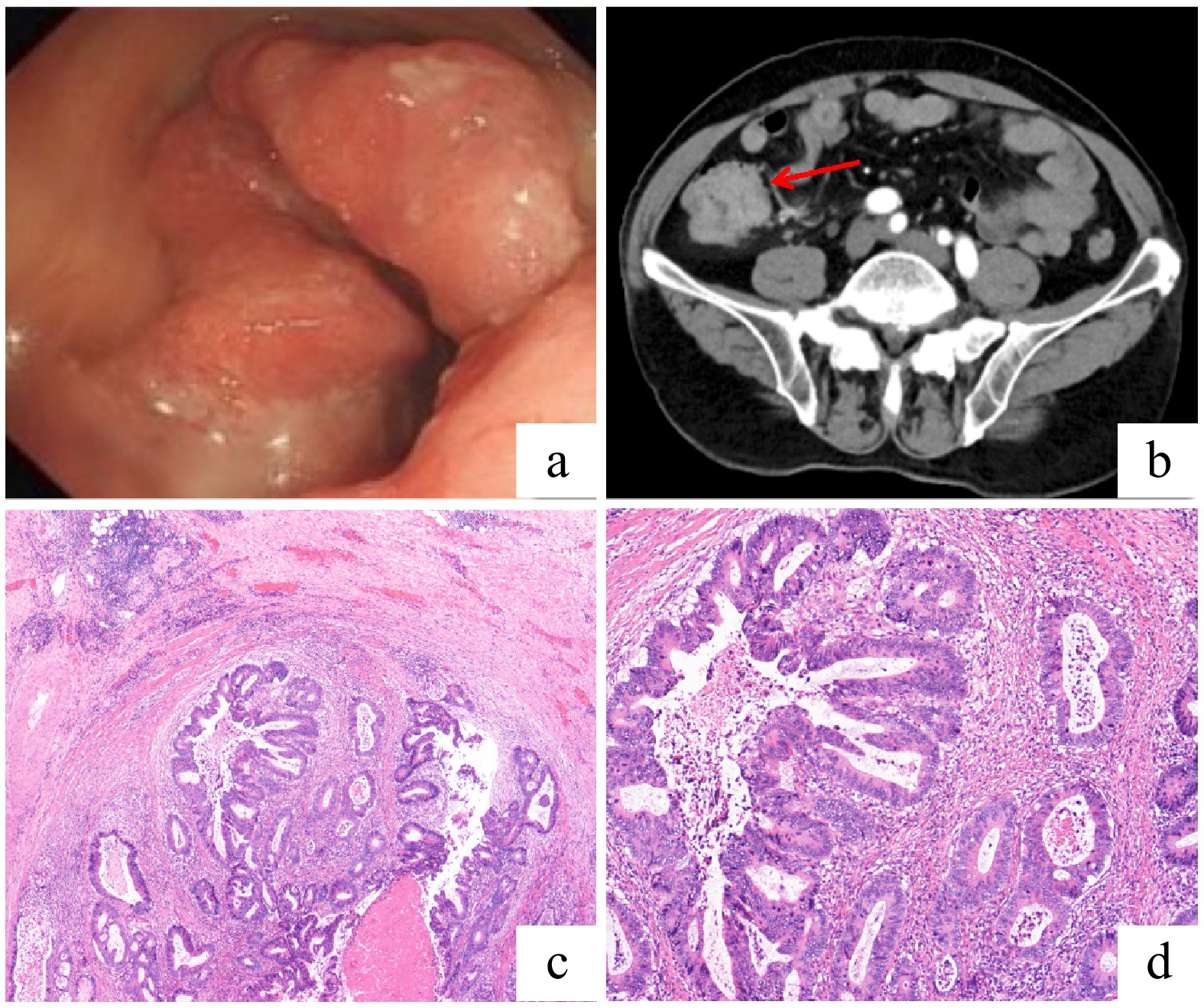
Figure 2.
Diagnostic and histopathological findings of ileocecal adenocarcinoma. (a) Colonoscopic view of a large, irregular mass in the ileocecal region. (b) Contrast-enhanced abdominal CT scan (axial view) showing irregular wall thickening with a marked enhancement in the ascending colon and ileocecal region. (c) Low-power photomicrograph (hematoxylin and eosin [HE] staining) of the resected specimen revealing an ulcerated, moderately differentiated adenocarcinoma with transmural invasion into the muscular layer. (d) High-power photomicrograph (HE staining) demonstrating the neoplastic glands with enlarged, hyperchromatic nuclei and evident mitotic activity. No significant perineural or lymphovascular invasion was identified.
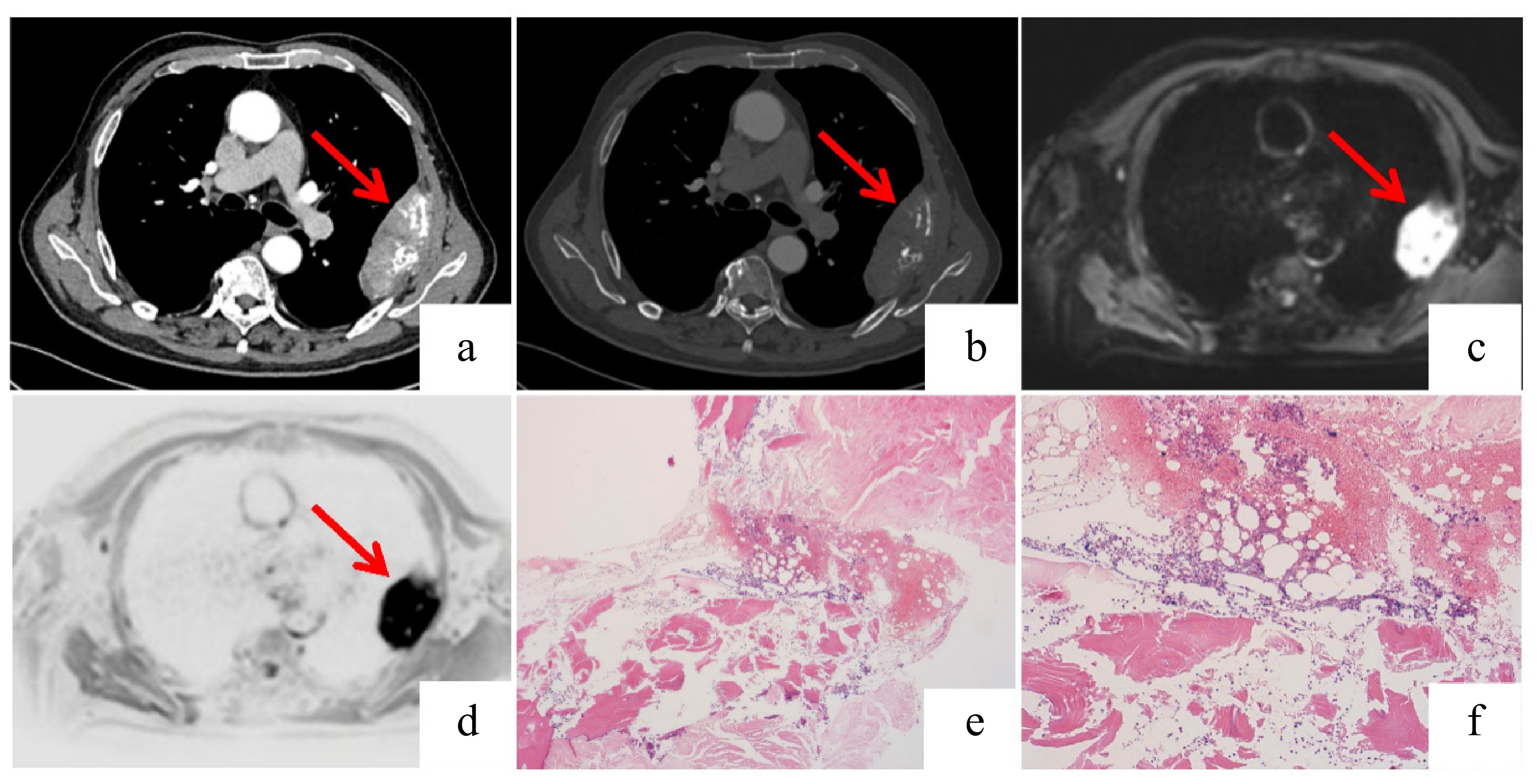
Figure 3.
Imaging and histopathological findings of the plasmacytoma. (a), (b) Axial and coronal contrast-enhanced chest CT scans demonstrating irregular left pleural thickening (measuring approximately 9.8 × 3.1 cm in the maximum cross-section) with associated destruction of the adjacent rib. (c), (d) Fusion positron emission tomography (PET)-MRI images reveal a rounded mass in the left chest wall showing hyperintensity on DWI with corresponding hypointensity on the apparent diffusion coefficient (ADC) map, consistent with restricted diffusion. Increased signal intensity is also noted in the adjacent rib on the MRI sequence. (e), (f) The bone marrow biopsy specimens show scattered plasma cells with lambda light chain restriction indicated by immunohistochemistry: (f) lambda-positive; (e) kappa-negative. This confirmed the presence of monoclonal plasma cells. The overall cellularity and number of plasma cells were not significantly increased.
The patient was subsequently transferred to our institution for comprehensive management. Colonoscopy identified an ulcerative mass in the ileocecal area (Fig. 2a), and a histopathological examination via an endoscopic biopsy confirmed a moderately differentiated ileocecal adenocarcinoma (Fig. 2c, d). Concurrently, a biopsy of the left rib mass was conducted. The histological analysis revealed sheets of monomorphic plasma cells (Fig. 4a, b). Immunohistochemistry (IHC) showed that the neoplastic cells were strongly positive for CD38 and CD138 (Fig. 4c, d) and exhibited lambda light chain restriction (kappa-negative, lambda-positive) (Fig. 4e, f). Although his bone marrow morphology showed only 1% plasma cells, and flow cytometry did not detect a monoclonal population, the presence of clonal lambda-restricted cells supported a diagnosis of plasmacytoma. Whole-body MRI was performed to differentiate between solitary bone plasmacytoma (SBP) and multiple myeloma (MM), revealing no other bony lesions or distant metastases (Fig. 3c, d). A bone marrow biopsy further confirmed minimal involvement (Fig. 3e, f).
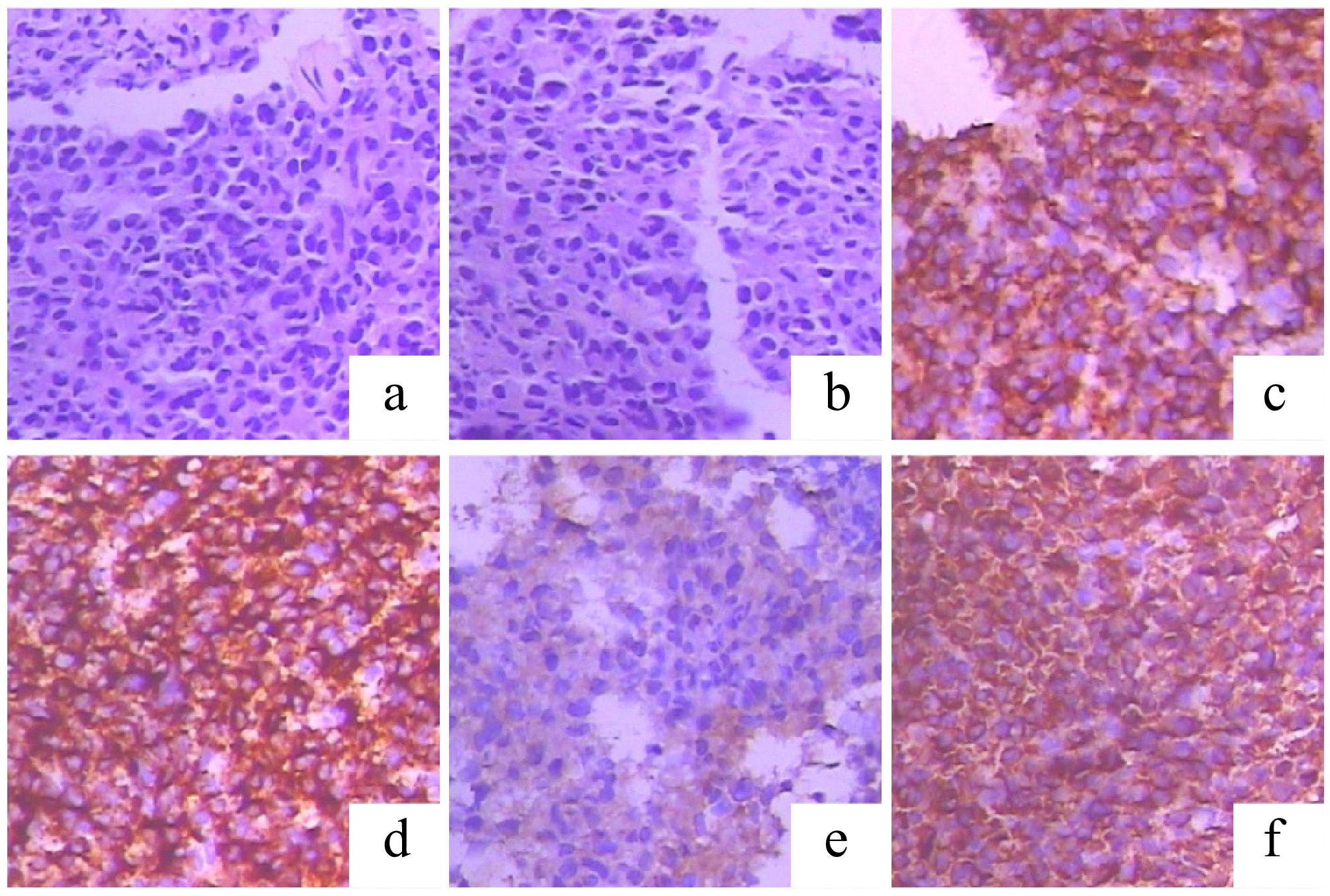
Figure 4.
Histopathological and immunohistochemical (IHC) features of the left rib mass, diagnostic of plasmacytoma. (a), (b) Hematoxylin and eosin (H&E) staining reveals sheets of monomorphic plasma cells. (c), (d) The neoplastic cells show strong and diffuse positivity for the plasma cell markers (c) CD38 and (d) CD138. (e), (f) IHC staining demonstrates lambda light chain restriction, supporting a monoclonal plasma cell proliferation: (f) positive for lambda light chains; (e) negative for kappa light chains.
Following a discussion among the multidisciplinary team (MDT), the patient was definitively diagnosed with MPMN: Synchronous ileocecal adenocarcinoma and SBP with minimal bone marrow involvement, initially presenting as AMCI.
The MDT's consensus prioritized surgical resection of the ileocecal adenocarcinoma because of its more aggressive potential. The patient underwent a radical right hemicolectomy. The final postoperative pathological confirmed a moderately to well-differentiated tubular adenocarcinoma (pT3N1bM0, Stage IIIB). The patient recovered well from surgery. At the 1-month postoperative follow-up, he was in good condition and was transferred to the hematology department for subsequent radiotherapy targeting the SBP. At the 6-month postoperative telephone follow-up, the patient reported no recurrence of cerebral infarction, with good recovery of neurological function and no re-elevation in D-dimer levels. The solitary bone plasmacytoma responded well to local radiotherapy, achieving local control without radiotherapy-related adverse effects, although its long-term efficacy still requires regular monitoring. The patient's intestinal function has also recovered very well, with no signs of recurrence such as anastomotic thickening, and his hemoglobin levels have returned to normal. At the 1-year postoperative telephone follow-up, no long-term treatment-related adverse effects were observed, and the patient's condition remained stable.
-
Patients presenting with cryptogenic stroke constitute a population at an elevated risk for underlying occult malignancy. Epidemiological studies suggest that approximately 20% of strokes with an undetermined etiology may be associated with a cancer diagnosis at presentation[9]. Prospective data from cohorts like the Norwegian Stroke Research Registry (n = 1,646) confirm that 5.0% of ischemic stroke patients harbor a concurrent active cancer, with a significantly heightened risk of cancer diagnosis within the ensuing 6−12 months[3]. Compared with stroke patients without cancer, those with an underlying occult malignancy experience poorer clinical outcomes, higher degrees of disability, and greater healthcare expenditures[10]. Across multiple studies, pulmonary and gastrointestinal adenocarcinomas are the predominant malignancies identified in stroke patients with cancer[10,11]. Although solid tumors are most common, hematologic neoplasms such as non-Hodgkin lymphoma have also been documented in this context[12]. Adenocarcinomas represent the most frequent histological subtype among cancer-related stroke patients[10,12,13], a predilection largely attributed to their mucin-secreting properties[14]. Mucin, a high-molecular-weight molecule, promotes microthrombus formation by interacting with cell adhesion molecules like P- and L-selectins. Nevertheless, the precise mechanisms of cancer-related stroke have not been fully elucidated. The prevailing pathophysiological model centers on cancer-induced perturbations of coagulation and fibrinolysis[15]. Tumor cells release procoagulant factors and inflammatory cytokines that directly or indirectly activate the coagulation cascade, creating a systemic hypercoagulable state. This, in turn, predisposes patients to intracranial microthrombosis and disseminated intravascular coagulation. In patients with plasmacytoma and multiple myeloma, a comprehensive dysregulation of both the coagulation and fibrinolysis, driven by a cytokine network, collectively leads to an increased risk of thrombosis[16]. First, patients with plasmacytoma and multiple myeloma commonly exhibit enhanced endogenous thrombogenic potential. Concurrently, impaired fibrinolytic activity makes the formed thrombi more resistant to lysis. Second, the M-protein secreted by malignant plasma cells, which is the predominant paraprotein, not only increases blood viscosity but also interferes with normal fibrin polymerization, resulting in an abnormal fibrin structure. This abnormal structure impedes the binding of coagulation Factor XIII and plasmin, ultimately leading to the formation of more compact thrombi that are more resistant to fibrinolysis. The aforementioned dysregulation primarily mediated by inflammatory cytokines, particularly interleukin (IL)-6 and vascular endothelial growth factor (VEGF). The cytokine imbalance ultimately increases von Willebrand factor (vWF) antigens, Factor VIII coagulant activity, and fibrinogen levels, which collectively promote the formation of thrombosis[16,17]. Therefore, further mechanistic research is crucial to facilitate earlier detection of occult malignancies in patients presenting with cryptogenic stroke.
When discussing cancer-related stroke, Trousseau's syndrome is a critical concept. First described by French physician Armand Trousseau in 1865, this syndrome represents a spectrum of clinical manifestations, resulting from a cancer-associated hypercoagulable state. It is recognized as a type of paraneoplastic syndrome and represents the second leading cause of death in cancer patients, surpassed only by the malignancy itself[18]. AMCI is a well-documented manifestation of Trousseau's syndrome and can even be its initial presentation[4,19]. Although no universally accepted diagnostic criteria exist, several key features are characteristic. Neuroimaging often reveals multiple small infarcts across multiple vascular territories, with the "three territory sign" (TTS) (involving the bilateral anterior and posterior circulation) holding significant diagnostic value[20]. Furthermore, markedly elevated D-dimer levels are a common laboratory finding[14,21,22]. As a product of fibrin degradation, D-dimer is a sensitive, though nonspecific, biomarker of coagulation activation and thrombosis; its elevation indicates a systemic hypercoagulable state and has been identified as an independent risk factor for cancer-related stroke. Crucially, since acute multiple cerebral infarcts in MRI-DWI are not pathognomonic for Trousseau's syndrome and are also observed in cardiogenic embolism (most frequently atrial fibrillation)[23], a comprehensive work-up including electrocardiography (ECG) and echocardiography is mandatory to exclude cardioembolic sources before attributing the stroke to Trousseau's syndrome.
Applying these diagnostic considerations to our patient, a definitive diagnosis of Trousseau's syndrome remains challenging, despite suggestive features. Although the patient had confirmed active malignancy and the MRI-DWI demonstrated the TTS, several critical factors preclude a conclusive diagnosis. First, the absence of universally accepted diagnostic criteria for Trousseau's syndrome introduces inherent ambiguity. Second, the patient's D-dimer level was only mildly elevated (1.3 mg/L; normal range: 0–0.5 mg/L), contrasting with the markedly elevated levels typically reported in classic cases. Third, although routine ECG and transthoracic echocardiography (TTE) revealed no abnormalities, the inherent limitations of TTE in detecting left atrial appendage thrombi and nonbacterial thrombotic endocarditis (NBTE) compared with transesophageal echocardiography (TEE), coupled with the absence of prolonged Holter monitoring to exclude paroxysmal atrial fibrillation, mean that potential cardioembolic sources cannot be fully eliminated. Finally, the contribution of traditional stroke risk factors (advanced age, hypertension, smoking history) cannot be entirely disentangled from a potential paraneoplastic mechanism. Therefore, although Trousseau's syndrome represents a plausible diagnostic consideration, this case is most accurately classified as cryptogenic stroke representing the initial manifestation of occult systemic malignancies.
This diagnostic dilemma underscores that routine cancer screening is not warranted for all cryptogenic stroke patients because of the cost considerations. However, the presence of specific clinical indicators—including bilateral infarctions or TTS on MRI-DWI, particularly when combined with elevated D-dimer levels and relevant tumor markers such as carcinoembryonic antigen (CEA) and CA125—should prompt targeted investigation for occult malignancy. In such selected cases, timely cancer screening and initiation of appropriate antitumor therapy may potentially improve clinical outcomes.
MPMNs represent a rare but clinically significant phenomenon in oncology. Although numerous MPMN cases have been documented worldwide, the synchronous occurrence of ileocecal adenocarcinoma and SBP described in our case appears to be the first reported instance. The diagnosis of MPMN follows the classical Warren and Gates criteria (1932), requiring: (1) a histological confirmation of malignancy in each tumor; (2) distinct pathological features between tumors; and (3) development in separate anatomical sites with definitive exclusion of metastatic spread or recurrence. The reported incidence of MPMN varies substantially across populations, likely reflecting interethnic genomic differences and variations in the duration of clinical follow-up. This epidemiological variability necessitates cautious interpretation of the prevalence data. Equally important is recognizing the diagnostic challenge posed by MPMN. A kack of clinical suspicion frequently leads to misclassification as metastatic disease, particularly in the context of declining autopsy rates—a procedure that historically has been crucial for a definitive diagnosis. Among MPMN cases, double primary malignant neoplasms (DPMNs) represent the most common presentation, with adenocarcinoma being the predominant histological type. The extreme complexity of MPMN is illustrated by a reported case involving 17 distinct primary malignancies in a single patient[24], highlighting the remarkable potential for multiple oncogenic processes to coexist.
The precise etiology and pathogenesis of MPMNs remain incompletely elucidated. Current evidence indicates that their development involves synergistic interactions among multiple determinants, including genetic susceptibility, somatic mutations, iatrogenic exposure, endocrine influences, environmental factors, and the immunosenescence associated with aging. At the cellular level, carcinogenesis represents a multistep process involving cumulative genetic alterations. With advanced age, a progressive decline in immune surveillance and DNA repair capacity coincides with increased proto-oncogene activation, collectively elevating susceptibility to cancer. Epidemiological studies have revealed that approximately one-third of cancer survivors over the age of 60 develop second primary malignancies[25]. Specific risk factors for MPMN include hereditary cancer syndromes such as Lynch syndrome and familial adenomatous polyposis (FAP)[26,27], with epidermal growth factor receptor (EGFR) mutations representing an established molecular risk factor. Iatrogenic contributions are well-documented, as chemoradiotherapy for primary malignancies can induce second neoplasms[28,29]. Endocrine pathways contribute to MPMNs' pathogenesis, evidenced by the bidirectional risk association between breast and thyroid cancers[30]. Environmental and lifestyle factors account for up to 90% of cancer cases[31], with smoking, obesity, and insulin resistance increasing the risk of secondary cancer[32]. Future research should focus on elucidating the molecular pathogenesis of MPMNs to facilitate effective prevention and early detection strategies.
Early detection and accurate diagnosis are critically important in managing MPMNs. Given the high prevalence of metastatic disease, the emergence of new tumor lesions following an initial cancer diagnosis often leads to a presumptive diagnosis of metastasis, potentially overlooking MPMN. Since the treatment principles for MPMNs (typically with curative intent) differ fundamentally from those for metastatic cancer (palliative), maintaining vigilance when multiple lesions are identified is essential, with thorough investigations conducted to confirm the diagnosis. In the present case, after confirming ileocecal adenocarcinoma, the biopsy of the suspicious left chest wall lesion revealed plasmacytoma, establishing the diagnosis of synchronous MPMNs. This accurate diagnosis provided the crucial foundation for the subsequent MDT discussion, leading to the decision to prioritize radical surgery for the ileocecal adenocarcinoma and ensuring an evidence-based therapeutic pathway.
The management of cancer-related stroke is complex, and the current guidelines lack definitive recommendations on the optimal antithrombotic strategies to prevent recurrence. Antiplatelet therapy is often the pragmatic choice because of its favorable safety profile and potential antitumor effects via inhibiting platelet-mediated oncogenesis, but it may be insufficient to counteract the systemic hypercoagulable state[33]. Anticoagulation, particularly with low-molecular-weight heparin (LMWH), is the guideline-endorsed first-line intervention, though its efficacy for arterial stroke prevention remains inconclusive, as evidence primarily derives from venous thromboembolism studies. The significant bleeding risk in cancer patients, with an annual incidence of major hemorrhage reaching 20%[15], must be carefully weighed against the thrombotic risk. For this patient, the MDT implemented a perioperative LMWH protocol, discontinuing subcutaneous administration 24 hours preoperatively and reinstating it as early as feasible postoperatively, with vigilant monitoring.
No established evidence-based guidelines exist for managing MPMNs. Therapeutic decisions require personalized strategies through the MDT's evaluation of factors including age, life expectancy, tumor types, pathological staging, and nutritional status. In this case, the MDT prioritized managing the more aggressive ileocecal adenocarcinoma with radical surgery. Postoperative pathology confirming Stage IIIB disease led to a further MDT review, which concluded that although adjuvant CAPEOX chemotherapy would typically be indicated, priority should be given to treating the coexisting plasmacytoma. This case underscores the pivotal importance of the MDT's approach in managing complex MPMN presentations.
-
The co-occurrence of ileocecal adenocarcinoma and SBP presenting as cryptogenic stroke is exceptionally rare and carries significant clinical implications. On the basis of existing evidence demonstrating that cryptogenic stroke in cancer patients typically shows elevated D-dimer levels and multi-territorial cerebral infarcts[21], we propose that patients exhibiting these characteristics, along with no evidence of cardiogenic embolism, should raise a strong suspicion of underlying occult systemic malignancy.
The management and prognosis of MPMNs differ fundamentally from those of metastatic cancer. Enhancing clinical research to reduce misdiagnosis is therefore crucial for optimizing patient outcomes. The MDT approach has become essential in oncology care, proving particularly valuable in complex cases such as MPMNs accompanied by cerebral infarction. Through a comprehensive risk–benefit analysis, the MDT model enables the formulation of precise treatment strategies that can improve overall survival and quality of life for these patients.
-
The study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from the patient for publication of this case report and any accompanying images. All identifiable information has been removed to protect the patient's privacy. The article is exempt from Ethical Committee approval, as it is a case report that does not include data collection and analysis.
-
The authors confirm their contributions to the paper as follows: conceptualization: Zheng C; writing − original draft, investigation: Meng Z; supervision: Zheng C, Ye L; project administration: Gao H, Zhang K; data curation: Gao H, Li P; writing − review and editing: Ye L, Chen J. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed during this study are included in this article. Further inquiries can be directed to the corresponding author.
-
We appreciate the help of Li L (Department of Radiology) from Shandong Provincial Hospital Affiliated to Shandong First Medical University, and Cui X (Department of Pathology) from The Second Hospital of Shandong University.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Meng Z, Gao H, Zhang K, Li P, Chen J, et al. 2026. A case of synchronous ileocecal adenocarcinoma and plasmacytoma initially presenting with multiple cerebral infarctions. Gastrointestinal Tumors 13: e002 doi: 10.48130/git-0026-0004
A case of synchronous ileocecal adenocarcinoma and plasmacytoma initially presenting with multiple cerebral infarctions
- Received: 10 November 2025
- Revised: 19 January 2026
- Accepted: 26 January 2026
- Published online: 26 February 2026
Abstract: Acute multiple cerebral infarctions as the initial manifestation of multiple primary malignant neoplasms are very rare. Studies have shown that it may even be the initial manifestation of occult systemic malignancy in up to 3% of patients. We retrospectively reported a 74-year-old male who initially presented with right hemiparesis. Magnetic resonance imaging and diffusion-weighted imaging of the brain showed acute small infarcts in the left parietal and right temporal lobes, and acute multiple cerebral infarctions were diagnosed. At the same time, computed tomography scans of the chest and abdomen revealed the concurrent ileocecal wall thickening and a left pleural mass with associated adjacent rib destruction. Subsequently, colonoscopy and biopsy of the rib lesion confirmed that the patient was diagnosed with synchronous ileocecal adenocarcinoma and plasmacytoma. Given that cerebral infarction may be the first sign of multiple primary malignant neoplasms, a precise etiological diagnosis is crucial to guiding treatment strategies and optimizing clinical prognosis.