-
Kombucha is a beverage obtained by fermenting sweet tea under a symbiotic culture of bacteria and yeast (SCOBY)[1]. The fermentation results in a beverage with a diverse chemical composition, including organic acids (acetic, gluconic, glucuronic, citric, L-lactic, etc.), sugars (sucrose, glucose, and fructose), vitamins (B1, B2, B6, B12, and C), polyphenols, amino acids, and other compounds[2]. Black tea-based kombucha has good antioxidant activity due to the content of natural minor bioactive substances such as various groups of flavonoids (catechins, catechin gallates, etc.), purine series alkaloids (caffeine, theobromine, theophylline, etc.)[3,4]. Meanwhile, the final chemical composition of the fermented beverage depends on the SCOBY composition, sugar and tea concentration, fermentation duration, and temperature[5]. Due to its diverse chemical composition, there is a strong demand for this category of beverage[6], with active research on the development of food products containing kombucha, such as cupcakes[7], bread[8−11], cheese[12], and dairy products[13]. Such interest in the development of food products containing kombucha seems to be related not only to the possibility of obtaining fortified products due to their diverse chemical composition but also to the possibility of regulating the processes occurring within complex food matrices (food products) to obtain products with improved characteristics.
The demand for structured and thickened food products (beverages, gels, liquid candies, jelly desserts, sauces, dressings, etc.) is increasing in the market. At present, products containing gelling agents (hydrocolloids) have become increasingly popular among diverse consumer demographics. This is primarily due to their high water content, low-calorie content, and satiety-enhancing properties[14]. The necessity to perpetually augment the array of food products is attributable to the emergent demand for specific items. The food-grade hydrocolloids market is expected to grow in value by at least 50% over the next ten years[15]. This is due to their ease of use, variety of textures, and the possibility of their modification for different needs (sports gels, baby food, and specialty products), high potential for fortification with biologically active substances, and adaptation to different consumer groups. The key demand drivers are: (1) the trend towards healthy eating (high-protein puddings, vitaminized mousses, products with reduced sugar and fat); (2) convenience and practicality when eating on the go (energy gels, isotonic gels, yogurts, etc.)[16]; (3) innovativeness (the product is new and interesting to the consumer: product spherification, gelling, coating); (4) high popularity of the confectionery industry, where various jelly desserts (marmalade, both in the usual form and more modern) and various toppings and fillings, which often need to be thickened; (5) clean labeling and trends towards GMO-free products[17].
Depending on the purpose and desired texture of the final product, starches (including modified starches), xanthan gum, konjac gum, carob gum, carrageenan, gelatin, etc., can be used as gelling agents[14,18]. When analyzing the market segment of food products containing gel-forming agents, it was found that the leaders in this sector are various types of marmalade and jelly, which have a strong gel-like consistency. But nowadays, food gels structured with various hydrocolloids and having a less strong consistency than marmalade and jelly are very popular. Examples of such products are 'liquid marmalade', 'liquid candy', sports nutrition gels, products for people with swallowing difficulties, etc. Starch, xanthan gum, and carrageenans are frequently used as structuring agents in these products.
Xanthan gum (XG) is a natural polysaccharide that is derived from Xanthomonas campestris. XG demonstrates good solubility in both cold and hot water[19]. The elevated characteristic viscosity of XG solutions at low concentrations, in conjunction with their resistance to pH and salts, facilitates their extensive utilization as a thickening, stabilizing, and suspending agent in various industrial applications[20]. The effectiveness of XG is related to its macromolecular conformation and association in solution and at the interface[19].
Carrageenan (CAR), a polysaccharide derived from the red algae Chondrus crispus and Eucheuma denticulatum, with an average molecular weight of 200 to 800 kDa, is widely used in various food products as a gelling, thickening, moisture-retention, and stabilizing agent[21,22]. In the food industry, ι-, κ-, and λ-carrageenan are most commonly used. The solubility of carrageenan is found to be contingent upon its chemical structure, exhibiting high levels of solubility in water at temperatures of 60 °C and above, regardless of the class of carrageenan. CAR demonstrates stability at neutral and alkaline pH, except for acid hydrolysis, which occurs at pH 3.0 and below[21].
The objective of this study was to develop a technology for obtaining a kombucha structured with hydrocolloids (xanthan gum or carrageenan). The results of the study of hydrocolloid-structured kombucha are presented for the first time. The obtained data can be used in the development of fortified food products with modified structures.
-
For the production of kombucha, the following were used: black large-leaf tea (Moscow, Russia), sugar (Moscow, Russia), symbiotic culture of bacteria and yeast (Moscow, Russia), and drinking water. Hydrocolloids: xanthan gum (Neimenggu Fufeng Biotechnologies Co. Ltd., China) and carrageenan CEAMVIS 3383 (CEAMSA, Spain) were used as gelling agents. Vitamins: vitamin premix RUS 28.174 − composition: (thiamine (vitamin B1) − 48.75 mg/kg, riboflavin (vitamin B2) − 44.0 mg/kg, pyridoxine (vitamin B6) − 57.0 mg/kg, nicotinamide − 479.998 mg/kg, folic acid − 7,500 mg/kg) (DSM Nutritional Products Europe Ltd., Basel, Switzerland), ascorbic acid (Dia-m, Russia). Sodium hydroxide (LenReactiv, Russia), Folin–Ciocalteu's phenol reagent (Sigma-Aldrich, USA), gallic acid standard (98%) (Dia-m, Russia), sodium carbonate (LenReactiv, Russia), 2,2-diphenyl-1-picrylhydrazyl (DPPH) (ABCR GmbH and Co. KG, Karlsruhe, Germany) were used for analytical studies. Analytical grade reagents (Sigma-Aldrich, St. Louis, MO, USA) were used for chromatography. Flavorings: water-soluble natural identical flavorings (raspberry, apple, blackcurrant), consisting of a mixture of aromatic substances, carrier E1520, propylene glycol (Edaprof, Russia).
Technology of kombucha production
-
Kombucha was produced according to the technology described in a previously published article[23]. Briefly, black tea was taken in an amount of 0.5 wt.% added to boiling water (94 ± 2 °C) and infused for 10 min with periodic stirring. Sugar was then added in an amount of 5.0 wt.% and stirred for 5 min until dissolved, followed by filtration. The fermentation process was stopped when pH 3.3 ± 0.2 was reached. The duration of the fermentation process was from 10 to 14 d at 24−26 °C. The resulting kombucha was filtered using a 70 µm mesh filter, pasteurized in a hermetically sealed glass container at 65 °C in a Binder FED 53 chamber (Germany) for 45 min, and stored at 4 °C until use.
The fermentation process was monitored by changes in pH, acidity, and total soluble solids.
Methods of kombucha analysis
Determination of pH
-
The pH was measured using a Mettler Toledo S20_K electronic pH meter (Greifensee, Switzerland)[24].
Determination of titratable acidity
-
The acidity value was determined by the titrimetric method according to Loncar et al.[25]. Before the study, the samples were freed from carbon dioxide formed during fermentation by boiling.
Determination of dry matter content
-
The total soluble solids content was determined using an Atago refractometer (Tokyo, Japan) and expressed in degrees Brix (°Brix) according to Gamboa-Gómez et al.[26].
Study of color characteristics
-
The study was conducted using a portable colorimeter BS 7016 (3NH, China). The color was assessed using the system (L*, a*, b*), calculated indicators: whiteness index (WI) and yellowness index (YI), chroma (Cab), and total color difference (ΔE*). The values of L*, a*, and b* were automatically calculated using the device software. Measurements were taken at four different points of the studied object. Based on the results obtained, the whiteness and yellowness indices, as well as Cab and ΔE*, were calculated. The whiteness index (WI) and yellowness index (YI) were calculated according to the study[27] using Eqs. (1) and (2), respectively:
$ \mathrm{WI}=\sqrt{(100-L^*)^2+a^{*^2}+b^{*^2}} $ (1) $ \mathrm{YI}=142.86\dfrac{b^*}{L^*} $ (2) Chroma (Cab), total color difference (ΔE*) were determined according to Huang et al.[28] using Eqs. (3) and (4), respectively:
$ \mathrm{Cab}=\sqrt{(a^{*^2}+b^{*^2})} $ (3) $ \Delta E* = \sqrt{{\Delta L}^{{*}^{2}}+{\Delta a}^{{*}^{2}}+{\Delta b}^{{*}^{2}}} $ (4) Technology of kombucha production structured with hydrocolloids
-
Calculated amounts of each of the hydrocolloids (0.3 wt.%, 0.6 wt.%, 0.9 wt.%) were dissolved in water or kombucha solution (water : kombucha [1:1]) under constant stirring (150 rpm) and heating to 90 °C until complete dissolution (Supplementary Fig. S1). The obtained solutions were thermostated at 23 ± 1 °C for 12 h in a climatic chamber KK 240 (Pol-Eko-Aparatura, Wodzisław Śląski, Poland) before the studies.
Research methods of kombucha structured with hydrocolloids
Determination of pH
-
pH of the tested samples was determined accordance with the method described previously.
Investigation of color characteristics
-
The analyses were performed with the method described previously.
Viscosity analysis
-
The viscosity of the obtained samples was determined using a Fungilab Expert rotary viscometer (Spain) equipped with an R4 spindle at a speed of 100 rpm at 23 ± 1 °C.
Texture analysis
-
The texture study was performed using an EZ-test-SX universal testing machine (Shimadzu Corporation, Suzhou Instruments Manufacture, Suzhou, Jiangsu, China). Samples with a volume of 120 mL, pre-tempered at 23 ± 1 °C, were subjected to compression under load with a plate (diameter 35 mm and height 0.8 cm). The plate immersion rate before sample detection was 5 cm/min (pretest), after detection, 1 mm/sec (test), and the sample penetration depth was 20 mm. In texture analysis, the following were determined: 'Hardness' (N) – this is the maximum test force loaded on food products using a plunger, 'Young's modulus' (N/mm2) – a parameter related to the elasticity of the sample calculated from the stress (force)-strain curves in compression, and 'Energy' (kJ) – load areas. The data were processed using TrapeziumX software (Shimadzu, Japan).
Study of phase transitions
-
Phase transitions were studied using a differential scanning calorimeter DSC 3 (Mettler-Toledo, Switzerland). Before the study, the rate of heat flow was calibrated using indium. Samples weighing 8−10 mg were placed in 40 µL aluminum crucibles. The study cycle consisted of the following stages: stabilization, cooling from 25 to –20 °C at a rate of 5 °C/min, stabilization, and heating from –20 to 90 °C at a rate of 7 °C/min. The data obtained were analyzed using STARe software (Mettler-Toledo).
Total polyphenol content determination
-
The content of total polyphenolic compounds in samples was determined by the Folin–Chocalteu spectrophotometric method using a Spectro Quest 2800 spectrophotometer (UNICO, USA). For the study, the sample (100 μL) was diluted with water (900 μL) and analyzed according to the method described in the study by Frolova et al.[23].
Organoleptic evaluation
-
The sensory assessors were 11 people with at least five years of experience − three men and eight women (aged 28 to 72 years) − selected according to their perception threshold for basic taste, smell, and color according to the ISO 8586:2023 (Sensory analysis - Selection and training of sensory assessors) standard. All participants signed a volunteer consent to participate in the degustation. Following the ISO 13299:2016 (Sensory analysis—Methodology—General guidance for establishing a sensory profile), a standard sensory profile of samples was established. Sensory evaluation of the studied thickened samples was carried out according to the criteria 'Appearance', 'Color', 'Smell', and 'Flavor'. The description of the criteria is shown in Table 1. The sensory assessors had to evaluate the compliance of the investigated samples with the given criteria on a five-point scale (5 – very good, 4 – good, 3 – average, 2 – bad, 1 – very bad).
Table 1. Criteria for organoleptic evaluation.
Parameters Description Appearance Transparent or slightly opalescent liquid. May be cloudy when hydrocolloids are added. Should not contain lumps. Color From golden amber to dark brown, depending on the variety of tea and the degree of fermentation. Flavor Refreshing, with light acidity, slightly tart. May have fruity or floral notes. Sometimes slightly carbonated. Smell Sour, with fruity or vinegary tones (depending on maturity). There may be a flavor of tea. Each participant was given a spout pouch containing 50 mL of the sample. The assessment was carried out in individual specialized booths designed in accordance with the recommendations of ISO 8589:2014 (Sensory Analysis. Design of Test Rooms). Participants were given drinking water (22 ± 1 °C) to purify the taste buds to reduce the influence of the precedence effect.
Additionally, according to Spectrum Descriptive Analysis, 'Acid Intensity' and 'Smell Intensity' scores were evaluated on a scale from 0 to 15[29]. Here, zero denotes an absence of smell, and 15 is the maximum expression. This enables a comparative analysis of different products. In this case, the original kombucha and the thickened samples. Citric acid prepared in different concentrations was used as a reference sample for 'Acid Intensity'. To establish a range of smell intensity values, a series of reference substances was selected, including sunflower oil, orange juice, and chewing gum with cinnamon flavoring.
At the second stage of the research, an organoleptic evaluation of the studied samples of thickened kombucha with added flavoring was carried out. Three different water-soluble flavorings were used: raspberry, apple, and blackcurrant. The flavorings were added directly to the kombucha solution in the amount recommended by the manufacturer, then thickened using hydrocolloids, mixed thoroughly on a magnetic stirrer, and then packaged in spout pouches with a lid of 50 mL each. The samples were then incubated for 24 h at 4 °C.
Vitamin stability study
-
The used vitamin premix 45 mg (the component composition of the premix is presented in paragraph 2.1) and 112.5 mg ascorbic acid were added to 250 mL of water-diluted kombucha (1:1), mixed thoroughly, and incubated in an ES-20/60 shaker incubator (Biosan, Latvia) at 65 °C. Sampling was performed after 0, 16, 24 and 48 h.
Hydrocolloid-structured samples were prepared according to the technique described in 2.4, but a kombucha solution with vitamins was used instead of a kombucha solution. The formed samples were placed in a shaker-incubator ES-20/60 (Biosan, Latvia) at 65 °C. Sampling was carried out after 0, 16, 24, and 48 h.
Changes in vitamin content were monitored according to Bendryshev et al.[30] using an Agilent Technologies 1100 high-performance liquid chromatograph with a diode array detector (Agilent Technologies, Santa Clara, CA, USA).
Statistical analysis
-
One-way analysis of variance (ANOVA) was used to assess the variation in the groups, followed by Tukey’s post hoc test. The significance level was p < 0.05 with a 95% confidence level. Statistical analysis of the data was performed using SPSS Statistics 20 and OriginPro 2018 software package.
-
Kombucha was obtained by fermentation of sweetened black tea solution for 10−14 d. Characterization of the obtained kombucha is presented in Table 2.
Table 2. Characteristics of initial kombucha.
Parameter Value L* 22.4 ± 0.1 a* 4.9 ± 0.1 b* 5.7 ± 0.2 WI 77.9 ± 0.1 YI 36.5 ± 1.0 Cab 37.8 ± 1.8 pH 3.25 ± 0.05 Acidity, mL (NaOH)/100mL 2.29 ± 0.02 Dry matter content, °Brix 5.0 ± 0.1 The color of the obtained kombucha was characterized by a light brown shade. It is known that the color of the final kombucha depends greatly on the type and concentration of tea used to produce it[31]. After stopping the fermentation process, the pH value of the initial kombucha was 3.35 ± 0.05, and the acidity was 2.29 ± 0.02 mL (NaOH)/100mL. This ratio resulted in a mildly acidic flavor system. Similar pH values for fermentation for 10−14 d or more were obtained by other researchers[32]. The advantage of kombucha utilization includes not only its diverse chemical composition but also its low pH values, which help monitor the development of pathogens[1].
During the fermentation process, kombucha acquires the buffer capacity of the solution[3], which allows dilution of the original kombucha while maintaining low pH values to ensure the microbiological stability of the final product. For further studies, a 50% kombucha solution (base) with a pH of 3.57 ± 0.05 was used.
Characteristics of kombucha structured with hydrocolloids
-
In this study, kombucha solution-based samples structured with XG and CAR were obtained. The concentrations of the added hydrocolloids were 0.3%, 0.6%, and 0.9%. Experimental samples based on kombucha solution, structured XG and CAR were denoted with index "k" – XGk and CARk, respectively, and water-based samples had index "w" – XGw and CARw accordingly.
Color
-
Kombucha has specific color characteristics (Table 2), which can be directly reflected in the color characteristics of the final product (Fig. 1).

Figure 1.
(a) Appearance of samples (0.9% hydrocolloids). (b) Diagram of color characteristics (L*, a*, b*, whiteness index [WI], yellowness index [YI] and chroma [Cab]) of samples. (WI' – whiteness index values are reduced by a factor of ten in the diagram). XGk and CARk – samples based on 50% kombucha solution structured with xanthan gum and carrageenan. XGw and CARw – water-based samples structured with xanthan gum and carrageenan.
It was observed that upon visual evaluation (Fig. 1a), kombucha-based samples acquire a yellowish tint characteristic of kombucha itself, which was confirmed by color determination (according to Table 2, yellowness index [YI] 36.5 ± 1.0). Independent of the hydrocolloid concentration, the greatest color change was characteristic of the samples CARk (Fig. 1b). Thus, the CARk sample was characterized by the highest redness (a*) and the XGw and CARw samples by the lowest. The CARk sample had the highest yellowness (b*), which is confirmed by the highest YI value, among all the samples studied. At the same time, it is worth noting that no significant differences were found for the WI index. In terms of lightness index (L*), samples XGw and CARw had higher values than those of CARk and XGk, indicating a decrease in L* when structuring the kombucha solution with these hydrocolloids. In color saturation (Cab), the CARk sample exceeded all samples except the kombucha base solution. It is worth noting that no significant differences were observed between the XGk and CARw samples in terms of color characteristics. This may be because the XGk sample has some turbidity. According to the study by Gordillo et al.[33], turbidity in the samples under study causes changes in the qualitative and quantitative components of color (L*, Cab). The authors state that the increase in scattering power in the mixture due to the increase in turbidity should be the reason for the observed decrease in L* values, since as the amount of reflected light increases, less light passes through the sample. As turbidity increased, using the oil in the mixture as an example, it was shown that the color of the samples became more achromatic (a* and b* tended to zero), darker (L* decreased), and with a lower color intensity. Additionally, based on the obtained values (L*, a*, b*), a total color difference index (ΔE*) was calculated, thanks to which it is possible to predict whether the consumer will notice the color change. According to Quintanilla et al.[34], the color difference perception ΔE* varies depending on the observed color and the sensitivity of the human eye: ΔE* < 1 – no color differences could be detected by the human eye, 1 < ΔE* < 3 – minor color differences could be detected by the human eye, and ΔE* > 3 – color differences could be detected by the human eye. When calculating ΔE*, the values of the samples ranged from 6.0 to 7.7 for the samples with CAR and XG, respectively. It indicates that the expected color change of CARk and XGk samples is due to the use of kombucha as a base. It is important to understand that the color of finished products plays a major role in consumer evaluation and can influence behavior in choosing a particular product[35].
Rheological properties
-
In this study, a comparative analysis was carried out on changes in the rheological properties of the samples depending on the composition of the aqueous phase and the concentration of hydrocolloids (Fig. 2). When developing structured products, it is necessary to take into account the possible influence of pH on the rheological characteristics of the final product. The pH value of the studied samples based on the kombucha solution was 3.72 ± 0.05 for XG-based samples and 3.87 ± 0.03 for CAR-based samples.
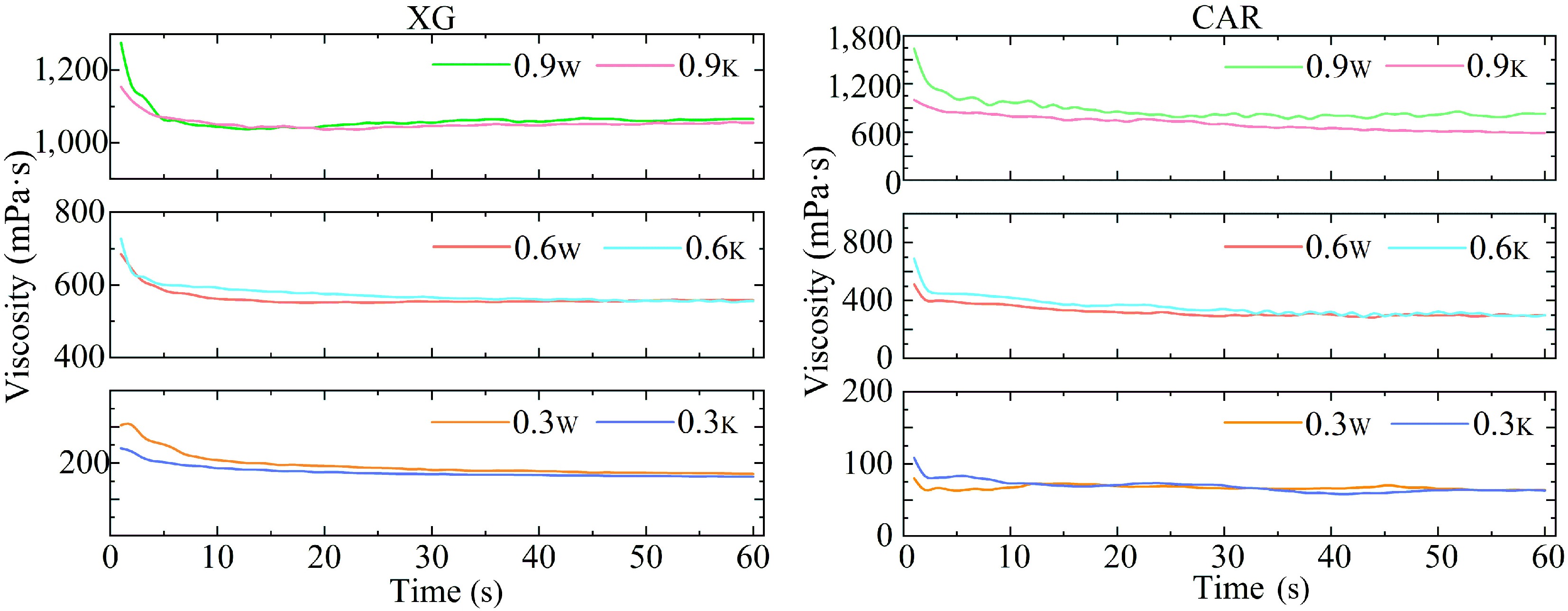
Figure 2.
Variation of viscosity of the investigated samples. XG and CAR – samples structured with xanthan gum and carrageenan respectively. Index 'w' denotes samples based on water; index 'k' denotes samples based on kombucha.
In the study, there was a decrease in viscosity during the first 10 s, after which all samples reached a plateau, which can be explained by the influence of dynamic forces occurring during shear. According to Wang et al.[36], this behavior was often observed with various gel structures and may be useful in the application of such structured systems in the food industry. For example, when they are used in the final finished product packaged in a tube or a doy-pack packaging. When passing through the narrow neck, the product, under the pressure exerted, will be easily squeezed out.
The use of XG and CAR allows the production of kombucha-based samples with a viscosity identical to that of water-based samples (Fig. 2). For XG-based samples, this is due to the ability of gum solutions to maintain a viscosity value over a wide range of pH values from 2 to 12[37]. For CAR-based samples, viscosity reduction at pH below 4.3 is possible due to the auto-hydrolysis process[37]. However, the autohydrolysis process can be prevented by using CAR-based systems below the gelation point. With this approach, it retains potassium ions bound by sulfate groups, if any, which in turn prevents the autohydrolysis process[37]. In the systems studied, a decrease in viscosity (Fig. 2) and strength (Fig. 3) was only observed at a CAR concentration of 0.9%; no significant difference (p ˃ 0.05) in viscosity was found in the other samples studied.
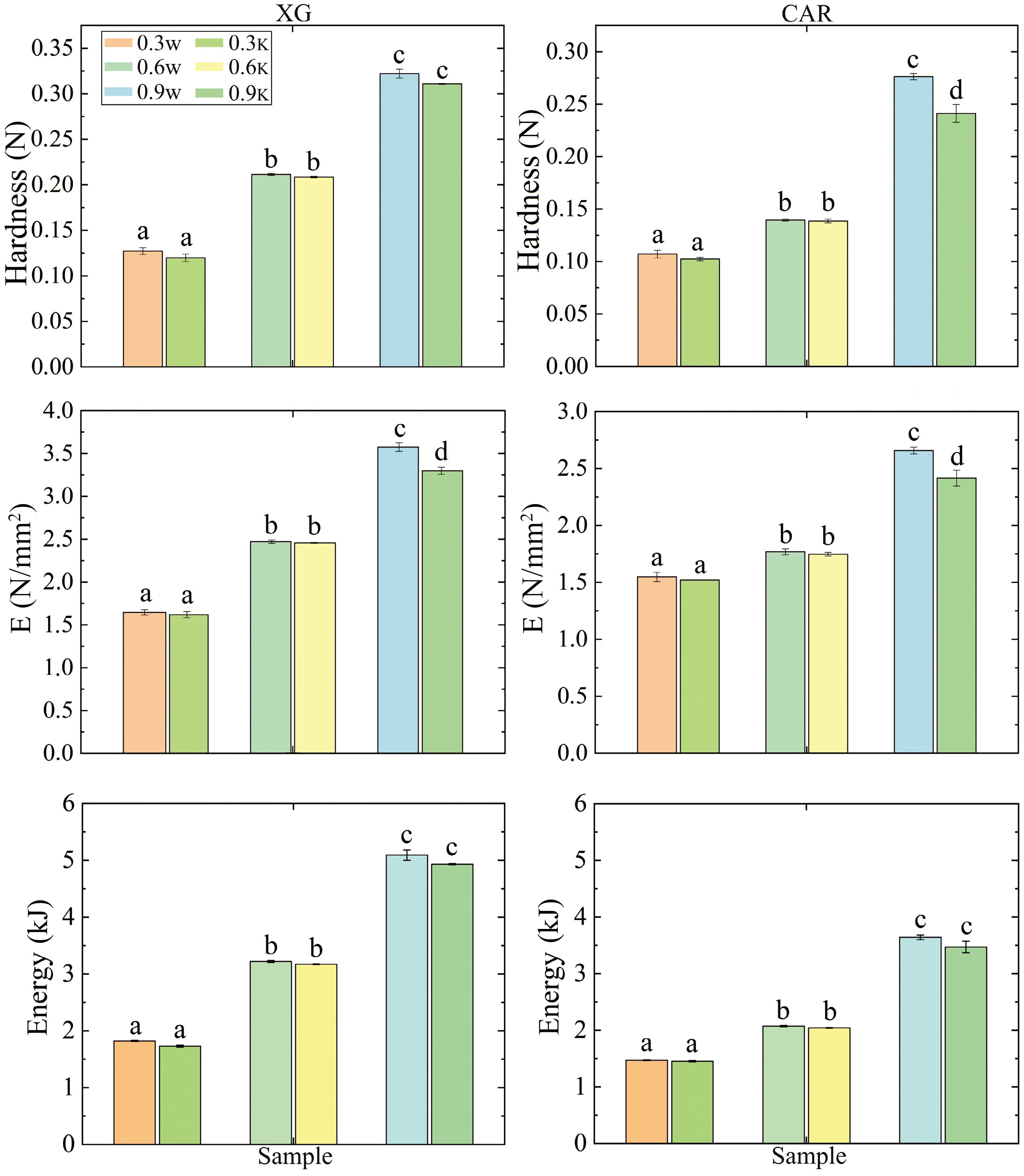
Figure 3.
Determination of the strength of the tested samples. XG and CAR – samples structured with xanthan gum and carrageenan respectively. Index 'w' denotes samples based on water; index 'k' denotes samples based on kombucha. Different letters denote samples that differ from each other (p < 0.05).
Textural properties
-
Texture is important both in terms of food flavor and food safety. It represents the integration of mechanical and thermal properties perceived in both oral and pharyngeal phases during food ingestion[38].
Comparative characterization of textural properties of the studied samples is presented by the parameters 'Hardness', 'Young's modulus' (E), and 'Energy' (Fig. 3). For all samples based on XG, there was a linear growth of parameters as a function of polysaccharide concentration R2 = 1, for samples based on CAR − R2 = 0.8875. When using XG and CAR at concentrations of 0.3% and 0.6%, the differences among textural characteristics were not significant (p > 0.05) in both water and kombucha-based samples. All samples structured with XG exhibited higher values than samples structured with CAR at each hydrocolloid concentration. It is noteworthy that a significant (p < 0.05) decrease in values was observed when determining the hardness and Young's modulus of the CAR-structured (0.9%) kombucha-based sample, relative to the water-based sample. A comparable trend was identified during the assessment of Young's modulus of samples structured with XG at a concentration of 0.9%. This can be explained by the fact that CAR and XG are anionic polysaccharides. XG is known to exhibit certain sensitivity to pH changes since its charge density is changed, and therefore, the molecular associations between the single molecules are influenced[39]. In the case of CAR, this appears to be due to its autohydrolysis, whereby CAR in the acid form cleaves at the 3,6-anhydrogalactose linkage in the molecule[37].
Thermal analysis
-
The features of phase transitions should be taken into account in the development of food products containing hydrocolloids. These features will allow not only regulating the rheological properties of food products but also choosing the optimal temperature conditions for the introduction of thermally unstable essential nutrients. As a result of these studies, it was found that the samples (at a concentration of 0.9% of the gelator) have a weakly pronounced broad peak from ~28 to 60 °C, at which a phase transition occurs, characteristic of both the XG sample[40] and CAR sample[41]. Notably, no differences were found between water-based and kombucha solution-based samples in this range. Food product development should also take into account the intended storage and marketing conditions of the final product. For example, when investigating phase transitions in frozen foods, recrystallization of ice may occur, resulting in changes in the quality of the final product[42]. An endothermic peak on heating was observed in all the samples studied in the temperature range of 2.15 to 3.84 °C (Table 3).
Table 3. Thermal characteristics of the studied samples (the initial temperature (Tonset), peak temperature (Tpeak), final temperature (Tend), enthalpy change (∆Hm)).
Sample Tonset (°C) Tpeak (°C) Tend (°C) ∆Hm (J/g) Water 1.14 ± 0.04 3.84 ± 0.25 8.05 ± 0.45 230.55 ± 31.19 Base −0.41 ± 0.18 3.10 ± 0.45 6.57 ± 0.87 220.31 ± 44.52 XGw 0.3 0.86 ± 0.02* 3.26 ± 0.13* 7.53 ± 0.93 291.61 ± 9.37 XGw 0.6 0.84 ± 0.02* 3.03 ± 0.16* 8.02 ± 0.59 272.21 ± 36.64 XGw 0.9 0.81 ± 0.06* 3.09 ± 0.13* 8.24 ± 0.48 284.84 ± 13.16 CARw 0.3 0.61 ± 0.01* 3.15 ± 0.03* 7.82 ± 0.76 301.67 ± 0.55* CARw 0.6 0.60 ± 0.04* 3.23 ± 0.25* 6.95 ± 0.11* 293.31 ± 17.79 CARw 0.9 0.56 ± 0.01* 2.95 ± 0.01* 7.56 ± 0.35 298.17 ± 1.36* XGk 0.3 −0.46 ± 0.04 2.49 ± 0.04** 6.91 ± 1.43 255.29 ± 23.76 XGk 0.6 −0.45 ± 0.01 2.35 ± 0.01** 6.24 ± 0.04 266.40 ± 2.83 XGk 0.9 −0.45 ± 0.02 2.32 ± 0.10** 6.31 ± 0.11 269.24 ± 5.75 CARk 0.3 −0.64 ± 0.11** 2.30 ± 0.01** 6.30 ± 1.15 265.43 ± 13.46 CARk 0.6 −0.61 ± 0.06** 2.42 ± 0.13** 6.58 ± 0.81 274.41 ± 15.20 CARk 0.9 −0.64 ± 0.01** 2.15 ± 0.03** 5.78 ± 0.68 247.21 ± 21.11 XGw and CARw – water-based samples structured with xanthan gum and carrageenan. XGk and CARk – kombucha-based samples structured with xanthan gum and carrageenan. Numerical indices show the concentration of structural agent in the sample, e.g., XGw 0.3 – indicated that the sample contained 0.3% xanthan gum. * Samples having significant differences from Water (p < 0.05). ** Samples with significant differences from Base (p < 0.05). Notably, no significant displacement of the peak value (Tpeak) of the phase transition temperature, depending on the hydrocolloid concentration in the range from 0.3% to 0.9% was found, which is in agreement with the data[43]. However, according to a study by Ahad et al.[44], when a sample with xanthan gum at a higher concentration (10%) was studied, there was a shift of the peak to higher temperatures. When comparing structured samples on water (XGw and CARw) with samples on kombucha (XGk and CARk), it was found that for all samples, there was a significant (p < 0.05) shift of Tpeak to a lower side, apparently due to the chemical composition of kombucha, specifically the presence of various acids in its composition. It is worth noting that this tendency was observed for all structured samples, with no significant differences depending on the hydrocolloid concentration. In terms of Tonset, all samples except XGk (0.3%–0.9%) differed from water and base solution. The final temperature (Tend) and enthalpy of melting of gels (∆Hm) in most cases had no significant differences (p > 0.05) compared to water and base solution.
Organoleptic evaluation
-
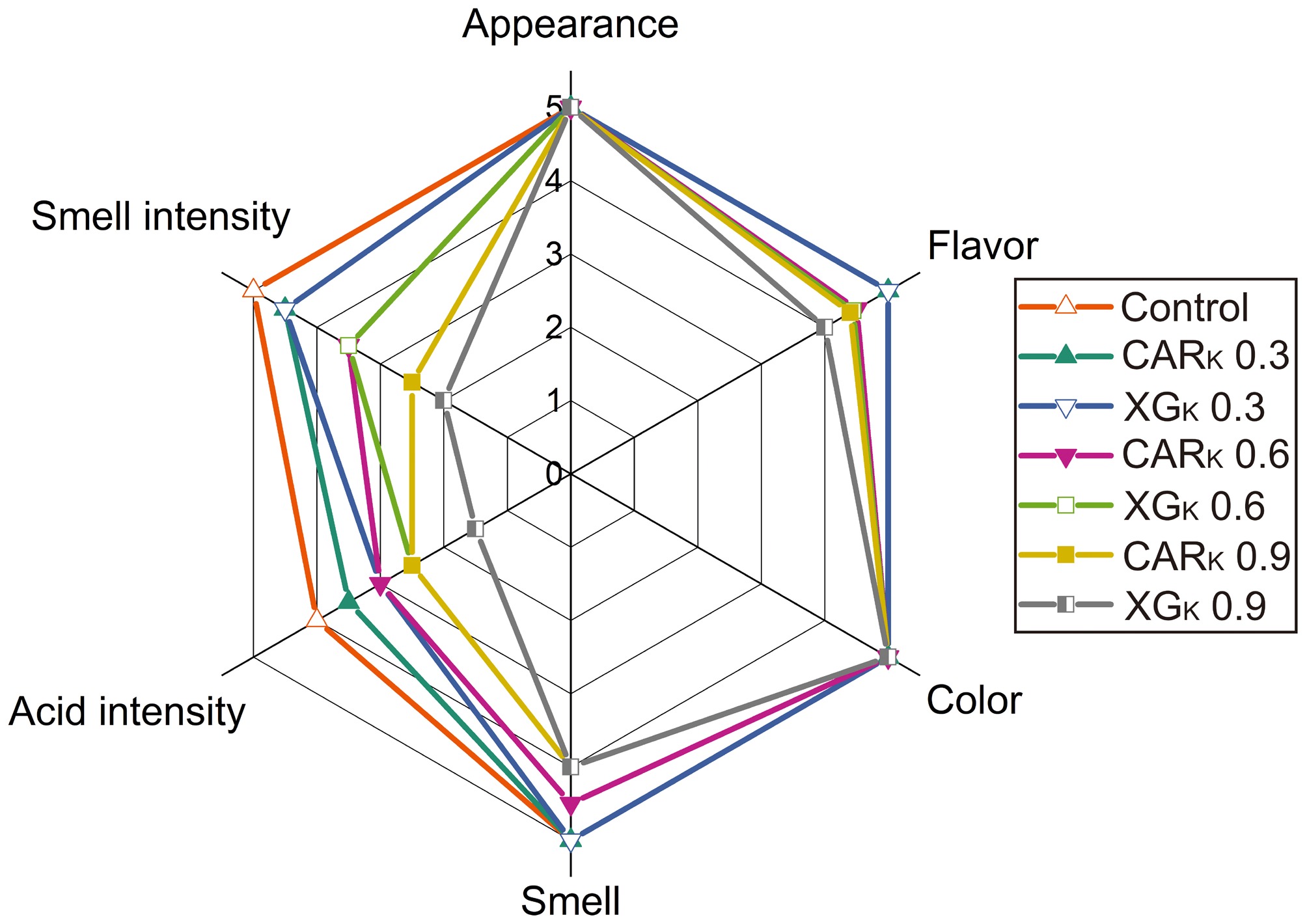
In addition to rheological properties, during product development, it is essential to pay attention to its organoleptic properties, especially when working with thickened liquids, because changes in rheology and texture directly affect the perception of the product under study[45]. It is known that the incorporation of diverse thickeners (e.g., pectin, xanthan gum) can modify not only the texture but also the flavor profile of the product. It is hypothesized that more viscous solutions may retard the diffusion of flavor compounds, which has the potential to modify the flavor profile. For instance, this could result in the masking of the natural sourness and fruity notes of kombucha, thereby rendering its flavor 'flat'. The organoleptic evaluation process facilitates the selection of a structuring agent that optimally balances texture and flavor intensity. Organoleptic analysis was carried out on kombucha-based samples structured with XG and CAR. Since kombucha itself has a specific taste, smell, and color, this step aimed to investigate the effect of the hydrocolloid used on the organoleptic profile. The results of the organoleptic evaluation of the samples that were tested are presented in Fig. 4.
The analysis revealed that the 'Appearance' and 'Color' of all the samples under study were evaluated with the maximum score (5.0), although samples structured with XG were characterized by low turbidity (Fig. 1). As the concentration of structuring agents increased from 0.6%, a decline in the 'Flavor' and 'Smell' scores was evident. The control sample (50% kombucha solution) had the highest scores for these parameters. Furthermore, an evaluation of all samples was conducted for the parameters of 'Acid intensity' and 'Smell intensity'. The 'Acid Intensity' in the control sample was four points. It was found that the use of XG and CAR in combination with kombucha resulted in a decrease in 'Acid Intensity', with the value of this index exhibiting a decline as the hydrocolloid concentration increased. It is noteworthy that a more pronounced decrease in 'Acid Intensity' values was found for the structured XG samples. A decline in 'Smell intensity' was observed in both XG and CAR structured samples, particularly at concentrations of 0.6%, which likely explains the reduced 'Flavor' and 'Smell' scores. In a subsequent note, sensory assessors noted that adding hydrocolloids to the kombucha solution resulted in a milder flavor profile and the appearance of a 'body'. A similar effect is observed when thickening drinks with hydrocolloids, as reported in the study by Pegg et al.[46].
At the second stage of the research, an organoleptic evaluation of the studied samples of thickened kombucha with added flavoring was carried out. This evaluation aimed to ascertain the possibility of additional flavoring. To achieve this objective, three pre-selected flavorings – raspberry, apple, and blackcurrant – were introduced during the development of thickened kombucha samples. The results obtained from the survey revealed a clear preference for the 'raspberry' flavoring, with a proportion of over 60% of respondents opting for this option. In comparison, the 'apple' flavoring was selected by more than 25% of participants, while the "blackcurrant" option was chosen by less than 10%. In addition, the sensory assessors were tasked with determining which of the samples exhibited the most vivid 'Flavor' and 'Smell'. It was found that the sample structured with CAR had a more pronounced flavor and smell.
Stability study
-
It has been established that insufficient intake of vitamins is a risk factor for a multitude of nutrition-related diseases[47]. In this regard, it is recommended that vitamins be incorporated into food products, particularly beverages. This recommendation is consistent with public health initiatives that are aimed at reducing the burden of diet-related diseases and promoting healthy lifestyles[48]. Given that the stability of vitamins is affected by light, temperature, pH, and the presence of oxidizing and reducing agents, stability studies were conducted on the stability of added vitamins in kombucha (50% kombucha solution) and in thickened samples (results are presented for a sample thickened with 0.9% XG). To study the stability of thermolabile components in the base and the hydrocolloid-thickened sample, an experiment was conducted in which the influencing factors were pH (3.57 ± 0.05) and temperature exposure (65 °C for 48 h). It is known that an increase in temperature leads to an increase in the rate of chemical and biochemical processes. Thus, with increasing temperature, there is a faster destruction of vitamins, in particular vitamin C[49,50]. In this regard, the study of the stability of ascorbic acid, one of the most heat-sensitive micronutrients, has received special attention. B vitamins (B1, B2, B6), folic acid, nicotinamide, and ascorbic acid were quantified using HPLC-DAD analysis. The results of the study are shown in Table 4.
Table 4. Stability of added vitamins in the base and developed sample structured with xanthan (0.9%).
Vitamins Concentration (mg/100 g) 0 h 16 h 24 h 48 h Base XGk Base XGk Base XGk Base XGk Vitamin B1 0.340 ± 0.014a 0.315 ± 0.035a 0.335 ± 0.007a 0.380 ± 0.014a 0.335 ± 0.007a 0.325 ± 0.007a 0.295 ± 0.007a 0.305 ± 0.007a Vitamin B2 0.255 ± 0.007a 0.265 ± 0.007a 0.250 ± 0.000a 0.275 ± 0.021а 0.280 ± 0.028a 0.245 ± 0.007а 0.230 ± 0.000a 0.250 ± 0.000а Vitamin B6 0.300 ± 0.028a 0.335 ± 0.021a 0.340 ± 0.014a 0.335 ± 0.021a 0.360 ± 0.028a 0.355 ± 0.035a 0.312 ± 0.014a 0.325 ± 0.007a Folic acid 0.040 ± 0.005a 0.039 ± 0.005a 0.040 ± 0.006a 0.035 ± 0.001a 0.040 ± 0.009a 0.034 ± 0.002a 0.039 ± 0.004a 0.033 ± 0.004a Nicotinamide 2.445 ± 0.176a 2.900 ± 0.099a 2.345 ± 0.134a 2.515 ± 0.177a 2.660 ± 0.127a 2.710 ± 0.061a 2.520 ± 0.057a 2.700 ± 0.113a Ascorbic acid 38.20 ± 0.43a 33.70 ± 0.42a 24.25 ± 0.78b 23.75 ± 0.07b 20.55 ± 0.19c 16.65 ± 0.21c 11.40 ± 0.57d 5.01 ± 4.101d Similar letters indicate no significant difference (p > 0.05) within-sample between the control points. As a result of this study, it was found that under conditions of acidic environment and elevated temperature for 48 h, all used vitamins (B1, B2, B6, folic acid, nicotinamide) were stable, except ascorbic acid. This tendency was maintained in both the base (degradation of ascorbic acid more than 70%) and the sample structured with 0.9% xanthan gum (degradation of ascorbic acid more than 80%). It is known that the stability of ascorbic acid in a food product depends greatly on various factors, including its components[51]. Kombucha is a complex system with a low pH value and contains various substances: polyphenolic compounds, fructose, glucose, sucrose, etc.[23]. Under the conditions of this experiment (storage at 65 °C), all the above factors could both increase and decrease the stability of ascorbic acid. The introduction of polysaccharides leads to accelerated degradation of this vitamin at high temperatures, but the exact mechanism of action is still unknown. Further studies are needed to determine the specific cause of ascorbic acid degradation in the presence of xanthan gum.
The main disadvantage of water-soluble vitamins is their high sensitivity to oxidation, resulting in discoloration and undesirable flavor[52]. However, as previously shown, they were fully preserved under the conditions analyzed and should not lead to negative changes in organoleptic properties. The conducted experiment allowed us to establish that kombucha and hydrocolloid-structured kombucha can be fortified with vitamins (B1, B2, B6, folic acid, and nicotinamide). The revealed stability of the analyzed vitamins at a temperature of 65 °C allows, if necessary, to carry out prolonged pasteurization of the studied samples to ensure microbiological safety with the preservation of the added vitamins.
In aqueous tea solutions, some polyphenolic compounds are susceptible to degradation by temperature and pH. For example, Li et al.[53] showed that at 24 °C for 3 h, the amount of catechins and theaflavins did not change, but at 70 °C for 3 h, the amount of catechins decreased by 25% and theaflavins by 56%.
In this work, preliminary data were obtained on the changes in the content of polyphenolic compounds in kombucha and thickened samples conditioned at 65 °C for 9 h. The total polyphenolic content in the initial samples was: in a 50% kombucha solution, 19.72 ± 0.28 mg/L, in structured XG and CAR samples, 12.14 ± 0.36 mg/L and 14.58 ± 0.22 mg/L, respectively. As a result of the studies, it was found that the number of polyphenolic compounds in kombucha for 9 h of thermostatic decreased by 20%, in 50% kombucha solution – by 24%, while in samples based on XG and CAR, no significant change (p > 0.05) in the total amount of polyphenolic compounds was detected. These results are most likely related to the ability of polysaccharides to bind to flavonoids to form so-called carbohydrate-polyphenol complexes. The interaction of polyphenols with carbohydrates depends on the molecular weight of the polyphenols, the hydrophilicity of the polyphenols, and the structure of the carbohydrate[54]. The work[55] states that xanthan can form a secondary structure in solution, forming hydrophobic pockets, due to which the encapsulation and binding of polyphenols occurs.
-
A comparative analysis of samples based on a 50% kombucha solution, structured by xanthan gum and carrageenan, in comparison with samples based on water at different concentrations of hydrocolloids, has been carried out. It was found that the rheological and textural characteristics of the studied samples change naturally depending on the concentration of hydrocolloids. It is shown that due to the low pH of the kombucha solution, the viscosity of the samples based on it decreases at a hydrocolloid concentration equal to 0.9%. It was found that for all the studied samples, there is a reliable shift of Tpeak to lower values. It is shown that in the studied samples under the influence of temperature (65 °C) for 9 h, there is no degradation of polyphenolic compounds, while in the kombucha itself, without added hydrocolloids, the decrease in the total amount of polyphenolic compounds is an average of 20%. It was obtained that the content of added vitamins (B1, B2, B6, folic acid, nicotinamide) does not change in the solution of kombucha and thickened samples in an acidic environment for 48 h at 65 °C, except for ascorbic acid (a three times decrease). The obtained results showed the possibility of structuring fermented kombucha beverages using hydrocolloids (xanthan gum and carrageenan).
Prospects for the use of the findings
-
The structured samples of kombucha obtained as a result of the study can be used both as a final product and as a basic food matrix for the development of food products with a given composition and properties.
For example, the findings could be used in the development of products for people with dysphagia. It is known that the thickening of liquids provides safety for swallowing and prevents aspiration[56]. According to the American Dietetic Association's classification of the national diet in dysphagia, four types of liquid consistency are distinguished: Thin liquid − 1−50 cP, syrup/nectar-like liquid − 51−350 cP, honey/cream-like liquid − 351−1,750 cP, and spoon-thick liquid − > 1,750 cP (1 cP = 1 mPa·s). Based on this classification, the samples developed in this study can be characterized as nectar-like/syrup-like at 0.3% thickener and honey-like/pudding-like at 0.6% and 0.9%. Nectar/syrup-like and honey/pudding-like liquids are typically described as non-sippable through a straw. As demonstrated in studies[56,57], thickened liquids are easier to control because their higher viscosity slows bolus movement both in the oral cavity and pharynx. Additionally, thickened liquids promote prolonged pharyngeal contractions and prolonged opening of the upper esophageal sphincter.
When using the developed samples as a basis for functional and specialized products, the influence of other components on stability and properties should be taken into account. For example, the salt content can significantly change the properties of gels based on xanthan gum, and gels based on carrageenan become more elastic in the presence of calcium ions. To investigate such interactions and prospects for use, it is necessary to conduct additional research on the stability and interaction of components, including under the influence of various factors.
The research was supported by a grant from the Russian Science Foundation (Grant No. 19-76-30014-π). We sincerely thank Prof. Kochetkova AA (Head of the Laboratory, Academician of the Russian Academy of Sciences, Dr. Sci. Tech.) for her expert advice and leadership throughout this study. We are also grateful to Bogachuk MN (Cand. Sci. Pharm., Laboratory of Food Chemistry) for performing the vitamin content analysis.
-
The authors confirm their contributions to the paper as follows: study conception and design: Frolova Y, Sobolev R; data collection: Sobolev R; analysis and interpretation of results: Frolova Y; draft manuscript preparation: Frolova Y, Sobolev R. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author (Frolova Y) on reasonable request.
-
The authors declare that they have no conflict of interest.
- Supplementary Fig. S1 Technological scheme of thickening of a kombucha drink.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Frolova Y, Sobolev R. 2026. Study of the properties of kombucha structured with xanthan gum and carrageenan. Beverage Plant Research 6: e005 doi: 10.48130/bpr-0025-0034
Study of the properties of kombucha structured with xanthan gum and carrageenan
- Received: 28 May 2025
- Revised: 31 July 2025
- Accepted: 13 September 2025
- Published online: 10 February 2026
Abstract: Kombucha-type fermented drinks have health benefits due to their diverse chemical composition, which is why they have gained popularity. Concurrently, active research is being conducted to develop food products containing kombucha. In this study, a method for producing hydrocolloid-structured kombucha (using xanthan gum or carrageenan) was developed, and its properties were investigated. The initial characteristics of kombucha used as a base were determined, including its color, pH, acidity, and dry matter content. The stability of various vitamins at 65 °C in 50% kombucha solution and structured kombucha was analyzed. The absence of degradation of all vitamins studied (B1, B2, B6, folic acid, and nicotinamide) was found, except for ascorbic acid. Variation of color characteristics depending on the hydrocolloid used was shown. The viscosity and strength of the samples increased consistently depending on the hydrocolloid concentration. A decrease in the peak phase transition temperature was observed in kombucha-based samples compared to aqueous samples. It is shown that the addition of hydrocolloids makes the flavor less intense compared to the initial kombucha. The study revealed that, in the presence of temperature, there was no degradation of polyphenolic compounds in samples containing hydrocolloids. However, an average reduction of 20% was observed in the kombucha solution. The present study provides information on a new application of a vitamin-fortified fermented kombucha beverage combined with hydrocolloids.
-
Key words:
- Kombucha /
- Hydrocolloids /
- Xanthan gum /
- Carrageenan /
- Vitamins /
- Polyphenolic compounds /
- Texture /
- Stability