-
Nitrogen is the fundamental element for all living organisms, acting as a primary limiting nutrient for plant productivity. Nitrogen biogeochemical cycling represents a cornerstone ecological process that supports terrestrial ecosystem structure and function. Nitrogen cycling encompasses a suite of interconnected processes, including biological fixation, mineralization, nitrification, assimilation, retention, and gaseous emission or leaching, which collectively regulate N availability in terrestrial ecosystems. Since the 1860s, however, human activities have drastically altered terrestrial N cycling patterns and processes by increasing biological N fixation, fossil fuel combustion, and converting inert N2 into reactive N (Nr) via industrial fertilizer synthesis, especially over the last half century[1]. These interventions have led to increased Nr emissions and subsequent deposition of both oxidized (NOy) and reduced N (NHx) at the global scale, threatening ecosystem structure and function through acidification and eutrophication[2−4]. Nowadays, human interference with the N cycle has exceeded the safe operating space for humanity[5], and N deposition has been recognized as one of the major drivers of global environmental change.
Forests are the main component of terrestrial ecosystems and play a vital role in harboring biodiversity, sequestering atmospheric CO2, mitigating global change, and supporting ecological functions and services. Over the past two decades, the understanding of N biogeochemical cycling in forest ecosystems has undergone a significant shift, driven by advances in molecular biology, isotopic tracing techniques, and modeling approaches. These developments have revealed spatial patterns in N deposition rates at the global scale, as well as uncovered previously unrecognized N cycling pathways and microbial contributions to N cycling processes[3,6,7]. Under elevated deposition, forests across varied climatic zones and land-use histories exhibit divergent shifts in N status and stocks, giving rise to emerging spatial and temporal patterns[2,8]. These progresses have enhanced the understanding of global N deposition patterns and the mechanisms in regulating N availability, turnover, and loss in forest ecosystems. Although several comprehensive reviews have addressed soil N cycling processes in forest ecosystems, they have mainly focused on specific forest types and discrete components of the soil N cycle, such as nitrification, mineralization, and denitrification[9,10].
Until now, relatively little attention has been paid to integrating the complete N biogeochemical cycle across diverse forest ecosystems, particularly in tropical and temperate forests, which exhibit distinct climatic conditions, vegetation structures, and soil properties. Furthermore, interactions among plants, soils, and microbial communities, such as root exudates and mycorrhizal associations, are frequently studied in isolation rather than as integral components of a coupled plant–soil–microbe system that regulates N transformation rates. Moreover, key N cycling mechanisms, such as biological N fixation, microbial-mediated transformations, plant-soil interactions, and N loss pathways, are being reconfigured in complex and often nonlinear ways[6−9]. Therefore, a synthesized, mechanistic understanding of these processes is urgently needed to forecast ecosystem responses across large spatial scales under ongoing atmospheric N enrichment.
This review presents recent developments in forest N biogeochemical cycling within the context of global N deposition. It addresses spatiotemporal patterns of N deposition, deposition dynamics, forest N status, N-cycling processes, underlying mechanisms, and relevant theoretical advances. By clarifying the interconnections among these processes, the review aims to deepen understanding of the implications of N deposition for forest health, biodiversity, and ecosystem services, especially their role as carbon sinks. The review also identifies critical knowledge gaps and prioritizes future research directions to address the challenges of anthropogenic N deposition, thereby supporting the sustainable management of global forest resources in the Anthropocene.
-
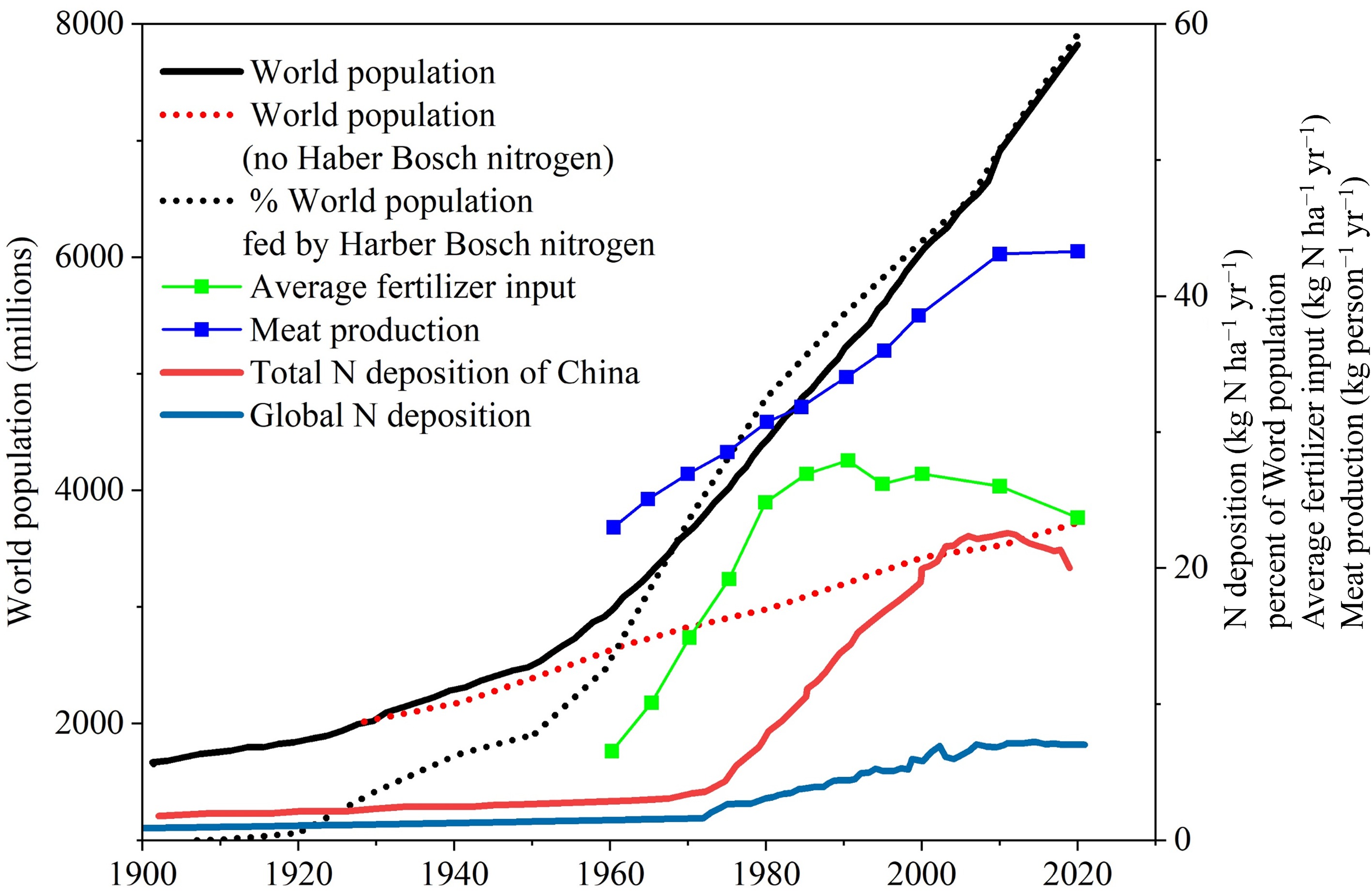
Two anthropogenic activities have greatly increased reactive nitrogen (Nr) availability: food and energy production, which have grown with population[1] (Fig. 1). At the beginning of the 21st century, food and energy production increased the anthropogenic Nr creation rate by over a factor of ten compared to the late 19th century. Global N deposition was estimated at only 31.6 Tg N yr−1 in 1860 but rose to 103 Tg N yr−1 by the early 1990s. Between 1990 and 2010, deposition rates remained relatively stable, ranging from 93.0–96.1 Tg N yr−1[1,11]. During this period, North America, Europe, and Asia were identified as hotspots of N deposition[3,12]. Following severe atmospheric N pollution and high deposition loads, deposition rates in the United States and Europe began to stabilize or decline from the late 1980s to early 1990s, mainly due to the implementation of stricter air pollution control legislation[13,14]. A global synthesis of more than 52,000 site-years (1977–2021) revealed that land N deposition reached approximately 92.7 Tg N in 2020, peaking around 2015[14].
With shifts in socioeconomic development patterns, a recent assessment estimates the global average terrestrial N deposition at ~7.0 kg N ha−1 yr−1 in 2020 (Fig. 1). The hotspots have shifted from developed regions to developing regions, particularly in South Asia, Southeast Asia, and South America at low to mid-latitudes[1,14]. This spatial redistribution reflects differing emission trends: the increase in N deposition across developing regions is primarily driven by rising NH4+, while the decline in Europe and North America stems primarily from reduced NO3− concentrations. Guo et al. systematically evaluated the effects of reactive N control policies and projected that, by 2050, global anthropogenic emissions of NH3 and NOx could be reduced by 40% and 52%, respectively, relative to the 2015 levels[15]. In fact, N deposition has declined since the mid-1990s in the United States and Europe, and more recently in China[16−19]. However, many countries (such as China, India, and the United States) still have a long way to go to address NH3 pollution, as they lack policies to reduce N overuse. In fact, current N deposition remains very high, especially in developing countries, and accumulated N deposition will continue to increase in the future.
China represents the core of this southward shift of deposition hotspots. The global fertilizer production and Haber–Bosch-fixed N rose exponentially after the 1950s (Fig. 1). Accordingly, total N deposition in China deviated markedly from the global background after 1980, closely tracking the national curves of livestock numbers and urea consumption, forming a pronounced high-deposition center. Using nationwide datasets on bulk N deposition across China between 1980 and 2010, Liu et al. found that annual bulk N deposition averaged 13.2 and 21.1 kg N ha−1 in the 1980s and 2000s, respectively, indicating an increase of approximately 60%[20]. The maximum was in south-central China, where N deposition reached 65 kg N ha−1 yr−1, ranking the country among the world's three highest deposition regions[14,21]. There are three significant transitions in N deposition in China: (1) the total N deposition began to stabilize in 2001–2005 mostly due to a decline in wet NH4+ deposition; (2) approximately equal wet and dry N deposition occurred in 2011–2015; and (3) the contribution of reduced N components in the deposition decreased due to increasing NO3− deposition[22]. In fact, systematic efforts to control atmospheric reactive N emissions were initiated during the 2011–2015 period[23].
Spatial patterns of N deposition are shaped by emission sources and atmospheric processes. While NOᵧ deposition has declined in many industrialized regions due to emission controls, NHx from agriculture often persists, leading to continued exceedances of ecosystem critical loads in sensitive areas. These exceedances drive biodiversity loss, with a global synthesis confirming that N deposition significantly reduces plant species richness across habitats[24]. Table 1 synthesizes N deposition rates and critical load exceedance across continents. Some studies projected that N deposition will continue to decline under current policies, yet reducing NHx emissions will depend on targeted agricultural measures[14,21,23]. Without such interventions, critical load exceedances are expected to persist, highlighting that future trends in N deposition hinge on the effective mitigation of ammonia.
Table 1. Selected continental-scale nitrogen deposition fluxes and exceedance of critical loads
Region Estimated total deposition (kg N ha−1 yr−1) Main forms (NHx/NOγ) Critical load exceedance (% of area) Ref. Europe ~10–20 (temperate) ~60% NH4+/40% NO3− ~40% of the forest area [134] North America ~5–15 (variable) Increasing NHx fraction ~25% of the forest area [135] East and South Asia ~15–40 (hotspots > 30) NHx dominated in some sites ~60%–70% in hotspot zones [14] Sub-Saharan Africa ~2–8 (less measured) NHx emerging Unknown/limited data [14] Global average ~8 NHx:NOy = 1.5:1 Limited data [14] Data compiled from recent monitoring and modelling syntheses. -
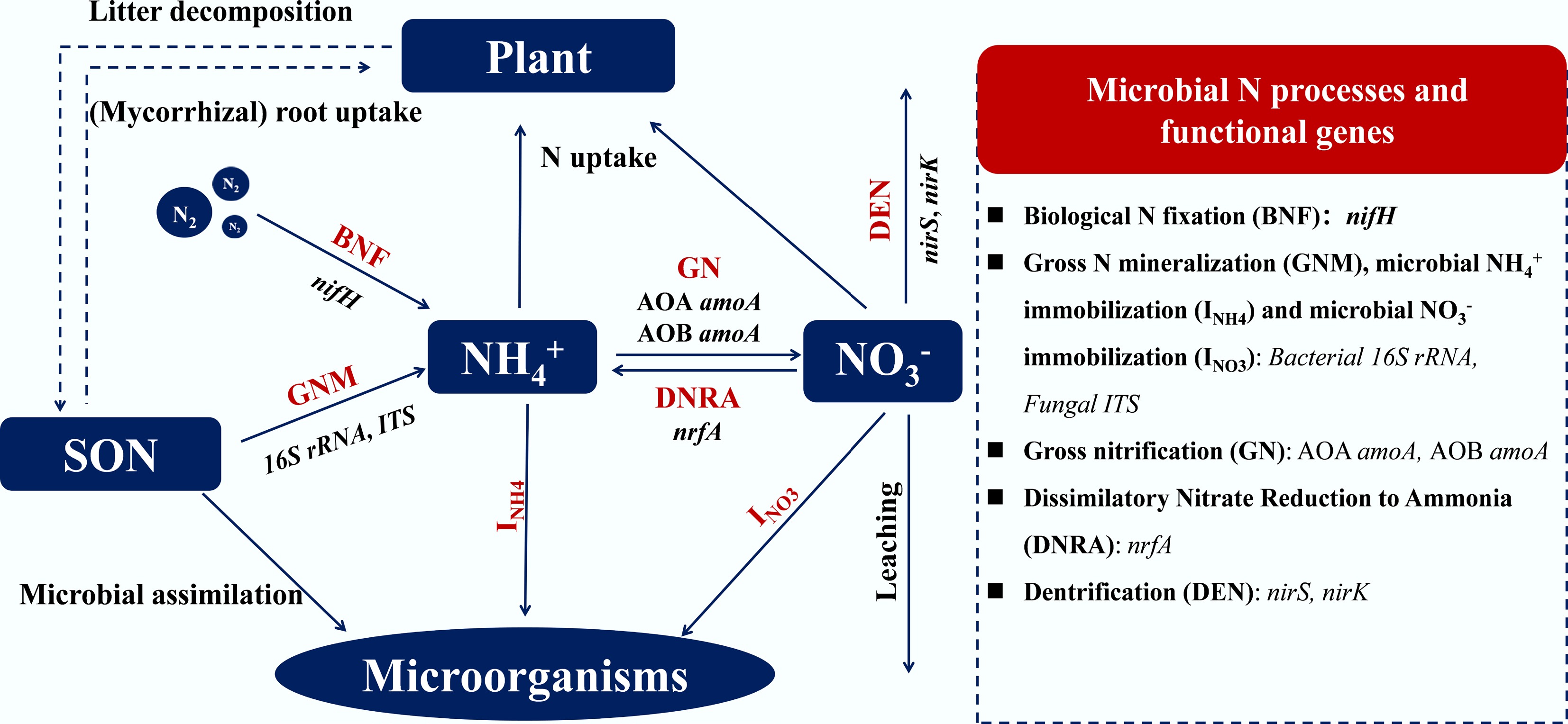
Terrestrial N cycling represents a complex network of biogeochemical processes that mediate the transformation, partitioning, and flux of N between atmospheric, terrestrial, and aquatic reservoirs, ultimately regulating ecosystem productivity, greenhouse gas balance, and environmental quality. The terrestrial N cycle comprises biological N2 fixation (BNF), gross N mineralization (GNM), gross nitrification (GN), denitrification (DEN), microbial NH4+ and NO3− immobilization (INH4 + INO3), dissimilatory nitrate reduction to ammonium (DNRA) and anaerobic ammonium oxidation (Anammox) (which occurs in select waterlogged soils and sedimentary environments), plant uptake and loss pathways via leaching (e.g., NO3− and DON) and gaseous emissions (e.g., NO, N2O and N2; Fig. 2). These processes are tightly coupled to carbon (C), phosphorus (P), and water cycles and are sensitive to climate change, substrate supply, and biotic interactions[25,26]. To contextualize the complexity of these interconnected processes, Table 2 summarizes the major pathways of the terrestrial N cycle, their key controlling factors (e.g., redox conditions, temperature, and soil C/N ratio), and representative indicators used in field monitoring (e.g., enzyme activities and gene abundances) and modeling (e.g., flux estimates and pool sizes).
Table 2. Major nitrogen cycling processes in terrestrial ecosystems, their controlling factors, and representative indicators
Process Description/transformation Key controlling factors Representative indicators/measurements Biological N2
fixation (BNF)Conversion of atmospheric N2 → NH3 by diazotrophs or symbiotic bacteria Soil P and Mo availability, temperature, moisture, and vegetation type Acetylene-reduction, 15N2 tracer,
δ15N natural abundanceMineralization (ammonification) Organic N → NH4+ C : N ratio, microbial biomass, temperature NH4+ flux, gross mineralization
(15N pool dilution)Nitrification NH4+ → NO2− → NO3− (by AOB/AOA) O2 availability, pH, temperature NO3− production, nitrifier gene abundance (amoA) Denitrification NO3− → NO → N2O → N2 under anoxia Soil moisture, C availability, redox potential N2O/N2 flux ratios (15N gas tracing) DNRA/Anammox NO3− → NH4+ (retention)/NH4+ + NO2− → N2 (loss) Organic C : NO3− ratio, anaerobiosis Isotope pairing, 15N mass balance Plant uptake and resorption Root/fungal uptake of NH4+, NO3−, or DON; N resorption from senescent leaves Root traits, mycorrhizae, soil N form availability Leaf/root N content, resorption efficiency Leaching and gaseous losses Export of NO3−/DON; emission of NO, N2O, N2 Rainfall, texture, drainage, and land use Stream NO3−, N2O flux, NO emission -
Elevated N deposition significantly alters the forms and stocks of soil N in forest ecosystems (Table 3). While atmospheric N deposition enriches the soil N pool, excessive inputs can disrupt C-N coupling, stimulating microbial activity and reducing soil organic carbon (SOC) stability[27−29]. Elevated N deposition generally increases soil NH4+ and NO3− concentrations, thereby enhancing N cycling[8,30]. However, prolonged N inputs can shift ecosystem N status from N limitation (defined as N availability fails to meet plant and microbial demand) to N saturation (defined as N supply exceeds the ecosystem's capacity to retain or utilize it), leading to nitrate leaching and nutrient imbalances[31−33]. In N-limited forests, ecosystem N stocks are relatively low, and N is tightly cycled and retained within the system to support plant and microbial growth, leading to ecosystem N accumulation with succession. In N-saturated ecosystems, however, most of the deposited N is lost through leaching and denitrification, without stimulation of CO2 sequestration[34]. Forest age and type can further mediate these responses.
Table 3. Effects of elevated nitrogen deposition on ecosystem nitrogen forms in forests
N deposition/experiment N form studied Findings from studies Future implications Ref. Atmospheric N deposition NH4+, NO3− Elevated N deposition increased both NH4+ and NO3− concentrations in forest soils in the Netherlands forest Prolonged N loading may lead to N saturation and enhanced nitrate leaching [30,136] Atmospheric N deposition (linked to urban/industrial regions) NH4+, NO3− Increased soil available nitrogen (mainly NH4+ and NO3−), especially in areas with higher population density and industrialization Continued emissions may accelerate soil N enrichment and N loss in urban ecosystems [8,30,31,38] Atmospheric N deposition NH4+, NO3− Chronic high N inputs lead to N saturation and elevated nitrate leaching Long-term high N deposition disrupts nutrient cycling and soil carbon stability [32] Atmospheric N deposition TN, NH4+, NO3− Enrichment of total and available inorganic N, accelerating nitrogen cycling and altering soil chemistry even at low deposition rates Alters ecosystem structure and functioning, e.g., species composition and productivity [33] Long-term N deposition TN, NH4+, NO3− Significantly higher total and available inorganic N in monsoon evergreen broadleaved forest than in
younger successional forestsIndicates older forests accumulate more inorganic N, reflecting stage-dependent N retention [137] Atmospheric N deposition NH4+, NO3− Enhanced inorganic N quantity and mobility through stimulated mineralization and transformation; older stands showed higher inorganic N fluxes Ecosystems are still efficient in N retention, but may reach N saturation with continued inputs [138] Atmospheric N deposition in Central African tropical forests DON, NH4+, NO3− Lowland forests lost N mainly as DON, others as inorganic N; tight N cycling but growing input-output imbalance Potential for future N saturation or shifts in dominant N forms [2,35] Meta-analysis of N deposition studies NH4+, NO3− Enhanced mineralization and nitrification increased inorganic N availability and possible N losses Broad-scale acceleration of N cycling and potential for increased N leaching [9] Long-term ammonium nitrate deposition NH4+, NO3− Despite long-term deposition, NH4+ and NO3− remained stable, indicating strong N limitation and ecosystem resilience Boreal forests remain resistant to N saturation, unlike temperate/tropical systems [36] In tropical/subtropical and temperate forests, soil N forms experience remarkable change from NH4+ to NO3− dominance, and increased N efflux[35], while the ratio of NH4+ to NO3− remains stable under long-term N deposition in boreal forests[36]. Regarding soil N stocks, existing results are inconsistent. Some studies reported no significant increase in soil N stocks due to N deposition[37] or noted that deposited N was largely assimilated by vegetation[38]. Nitrogen amendment experiments showed that, while total soil N may increase initially, the accumulation rate declines over prolonged periods and at higher input levels[39]. Moreover,15N-tracing experiments and a global meta-analysis confirm that forest soils act as a significant sink for atmospheric N deposition[34,40,41]. Lu et al.[42] found that soil N sequestration can keep pace with soil C (e.g., N-induced soil C accumulation rate at ~9 kg C/kg N), and developed a soil C sequestration hypothesis, suggesting N deposition can accelerate soil C sequestration in forest ecosystems at the global scale, regardless of ecosystem N status and climate zones.
-
Biological N fixation (BNF) is the largest natural source of new N input that supports terrestrial productivity. The BNF in natural biomes is 65–88 Tg N y−1[43,44]. Terrestrial biological N fixation shows a discrete latitudinal gradient, with tropical ecosystems demonstrating high rates of fixation and higher abundance of N-fixing species due to warm temperatures, heavy rainfall, and greater diversity of N-fixing species, while boreal and temperate ecosystems experience lower rates and lower abundance[45,46]. The rate of BNF is controlled by environmental factors (e.g., light and temperature), soil nutrients (e.g., P, iron, molybdenum, and vanadium), and N-fixers (e.g., the composition and abundance of rhizobial community, and nitrogenase activity)[47] (Table 4). The impact of N deposition on BNF in forests is complex and regulated by multiple factors. The conventional understanding is that N addition suppresses BNF because N fixation is energy-intensive, and elevated soil N availability may reduce the activity of N-fixing microorganisms. A meta-analysis showed that N addition reduced BNF by 31%[48]. However, the suppressive effect of N deposition is not universal and can be modulated by factors such as SOC content. Studies have shown that the adverse impact of N addition on BNF diminishes with increasing SOC, which can supply energy for N fixation and thereby alleviate energy limitation[49]. Furthermore, the response of BNF to N addition is influenced by both the forest succession stage and location. For instance, while N addition significantly suppresses BNF in disturbed and urban forests, it has no significant effect in suburban/rural and rehabilitative forests. These differential responses can be well explained by variations in the sensitivity of N-fixing microbial communities across forest types[50].
Table 4. Limiting factors of biological nitrogen fixation
Controlling factor Mechanism of action Main affected systems Adaptive responses Iron deficiency Limited synthesis of nitrogenase cofactors Marine systems, calcareous soils Increased secretion of iron chelators Phosphorus deficiency Inadequate ATP supply, constrained energy metabolism Tropical soils, lateritic soils Enhanced phosphatase activity, increased
phosphorus uptakeMolybdenum limitation Impaired function of iron-molybdenum cofactors Acidic soils, marine environments Alternative pathways (e.g., using iron-vanadium
nitrogenase)Oxygen sensitivity Nitrogenase deactivation Aerobic environments Formation of heterocysts, increased respiration,
spatial-temporal separationCarbon limitation Insufficient energy supply Barren soils, deep-sea environments Symbiosis with photosynthetic organisms, utilization of organic carbon Soil N transformations
Patterns and key regulatory factors of soil N cycling
-
Soil gross N cycling represents a fundamental nutrient process in forest ecosystems and collectively governs productivity patterns, soil C sequestration, and nutrient retention capacity. Due to pronounced differences in climate conditions, vegetation types, and soil properties across global climatic zones, both the rates and patterns of soil N cycling exhibit significant spatial heterogeneity among forest types. Understanding these variations is critical for predicting how forest ecosystem structure and function respond and adapt to global change. Current research on gross N transformation rates reveals that tropical forests exhibit the highest rates of GNM, INH4, GN, INO3, and DNRA, followed by subtropical forests, while temperate forests generally demonstrate the lowest rates (Fig. 3). In contrast, subtropical forests show the highest levels of soil ammonium and nitrate content, as well as the highest denitrification rates. Microbial N immobilization (INH4 + INO3) in subtropical forests is comparable to that in temperate forests. The ratios of soil NO3− to NH4+ (NO3−/NH4+) and that of GN to INH4 (GN/INH4) are indicators to estimate the potential risk of soil N loss[51−53]. Thus, the highest NO3−/NH4+ and GN/INH4 ratios in the subtropical forests indicate an N-leaky ecosystem. Elevated N inputs generally lead to a leaky N cycle in tropical forests, considering the increase in GN/INH4, NO3−/NH4+, and nitrous oxide emissions and the decrease in INO3 and DNRA[54].
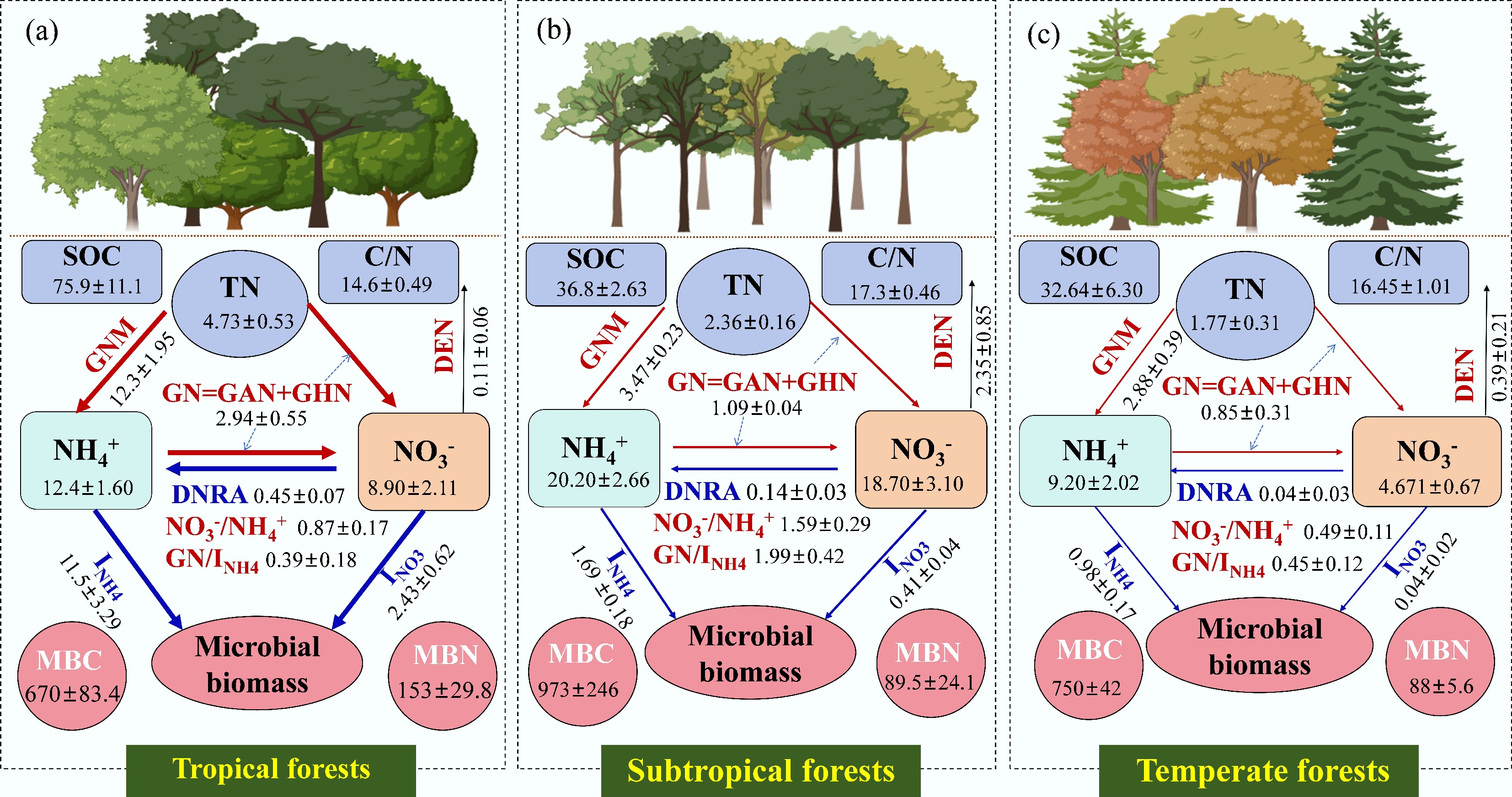
Figure 3.
Global patterns of SOC, TN, C/N ratio, available N, microbial biomass, and N transformation rates in (a) tropical, (b) subtropical, and (c) temperate forest soils. The red and blue line represents the processes of soil N production and retention, respectively. The thickness of line and arrows refers to the degree of each N transformation rate. GNM, gross N mineralization; GN, gross nitrification; GHN, gross heterotrophic nitrification; GAN, gross autotrophic nitrification; INH4, microbial NH4+ immobilization; INO3, microbial NO3− immobilization; DEN, denitrification; MBC, microbial biomass carbon; MBN, microbial biomass nitrogen. SOC, soil organic carbon; NH4+, extractable ammonium; NO3−, extractable nitrate. SOC: g kg−1, TN: g kg−1, NH4+: mg kg−1, NO3−: mg kg−1, MBC and MBN: mg kg−1; GNM, INH4, GN (GAN+GHN), INO3, DNRA and DEN: mg kg−1day−1. Data are cited from [54,126−133].
The spatial patterns of soil N cycling across forest biomes arise from distinct environmental and biological factors. Climate factors, particularly temperature and precipitation, directly regulate microbial and enzyme activities, serving as the 'master switch' for N transformation processes. In tropical forests, consistently warm and humid conditions sustain a rapid and continuous N cycle[54]. These soils, often acidic and rich in kaolinite and iron-aluminum oxides, strongly adsorb ammonium, thereby reducing nitrogen loss[55]. Under low pH (usually pH: 4.0−5.5), nitrification is primarily driven by ammonia-oxidizing archaea, albeit at relatively lower rates[56]. High rates of microbial N immobilization, alongside significant DNRA rates in anaerobic microsites, further promote efficient nitrogen retention[57,58]. In contrast, subtropical forests experience distinct seasonal shifts in dry/wet and cool/warm conditions, leading to a pulsed N cycle. Mineralization and nitrification intensify during wet/warm seasons, and NO3− is prone to leaching during rainy periods, while these processes are suppressed during dry/cold seasons[54,59]. Subtropical soils are generally less acidic (e.g., pH: 5.0−6.5), allowing both ammonia-oxidizing bacteria and archaea to contribute to nitrification, often resulting in a higher nitrification potential compared to tropical forests[56,60]. Thus, while tropical and subtropical forests are governed by climate, soil physicochemical properties, and microbial communities, the specific mechanisms differ markedly, shaping their distinct N cycling dynamics. In temperate forests, however, low temperatures and higher soil C/N ratios are the primary constraints, leading to lower N transformation rates[61,62]. Beyond these type-specific factors, tree species, soil properties (e.g., SOC, total N, and texture), microbial community traits (e.g., functional gene abundance such as amoA and nirK/S), and mycorrhizal types (e.g., ectomycorrhizal vs. arbuscular mycorrhizal) are further shaping soil N cycling[63−65]. Hence, it is necessary to integrate these biotic and abiotic factors to build more universal predictive models of soil N cycling.
Soil N production and retention
-
Studies from different periods have consistently showed that N addition promotes both GNM and GN rates in forest soils, although the increase magnitude is modulated by N dosage, climate zone, and soil depth[66−69] (Fig. 4). This stimulatory effect primarily arises because external N inputs directly enhance soil available N pool, thereby supplying more abundant substrates for microbe-mediated N transformation processes. In N-limited forest ecosystems, exogenous N can stimulate soil microbial activity and accelerate the decomposition and transformation of organic N. Moreover, N deposition has been shown to suppress microbial N immobilization, particularly in N-rich tropical and subtropical forests[54]. Meta-analyses and reviews on DNRA and denitrification in global forest soils under N deposition remain scarce. The limited number of case studies available suggests that these processes either decrease or show non-significant responses to N addition[70,71]. Furthermore, Elrys et al. indicated that N deposition may impair soil N retention capacity in tropical forests compared to subtropical systems, as reflected by increased ratios of NO3−/NH4+ and GN/INH4, along with reduced DNRA rates. These shifts could elevate the risk of N loss from tropical forest soils[69].
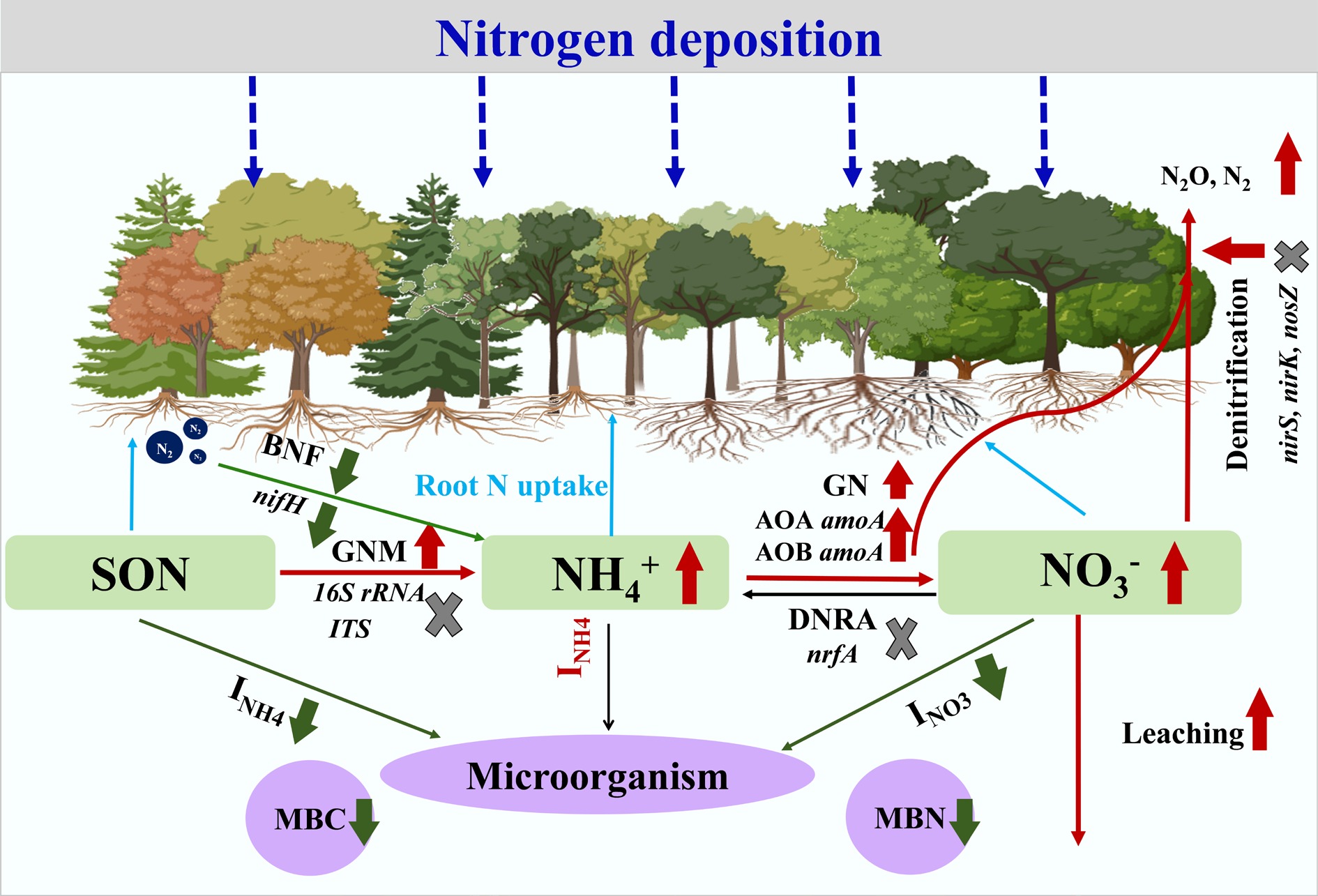
Figure 4.
The general pattern on the responses of soil microbes-driven gross N transformation to elevated N deposition in forest ecosystems. Red arrows indicate that N deposition exerts stimulatory effects on N pool size, N transformation processes, and associated fluxes, whereas gray arrows represent inhibitory effects of N deposition on these aspects. 'x' symbols denote uncertain effects under N addition.
Soil N losses
-
Soil N2O emissions and NO3− leaching are key indicators reflecting the N saturation status and loss risk in forest ecosystems. N2O is primarily produced by microbial nitrification and denitrification processes[72]. Most studies have shown that N deposition significantly stimulates N2O emissions from forest soils. For instance, long-term N addition in temperate and tropical forests has been found to increase N2O emissions by 113% to 250%[73]. Global meta-analyses further confirm that N addition enhances forest N2O emissions by 216% to 591%[74,75], contributing directly to 9.0% of global forest N2O emissions[76]. However, some tropical montane forests show no significant response to high N inputs[77], a variability that can be explained by the ecosystem N saturation hypothesis[2]. This theory also predicts an increase in N leaching with elevated N deposition[78]. Multiple long-term experiments have observed significant increases in NO3− and total dissolved N leaching under N addition[79,80] (Fig. 4), although the extent of increase is influenced by forest type, N application rate, and duration[81].
Underlying mechanisms of how N deposition affects soil N transformation
-
Nitrogen deposition directly or indirectly affects soil N transformation rates by altering soil physical and chemical properties (e.g., increasing SOC, NH4+, and NO3− content, and reducing soil pH), thereby changing the community composition, function, and activity of microorganisms involved in soil N transformation.
(1) N effect on microbial biomass and community composition. The effects of N deposition on soil microbes vary spatiotemporally, depending on duration and ecosystem type[54,82,83]. In N-limited ecosystems, short-term N input elevates soil available N, directly stimulating microbial growth and increasing microbial biomass C and N[84]. Microbes act as a critical N sink, assimilating available N into their biomass. This process can reduce substrates used for nitrification and denitrification, potentially lowering short-term N loss. However, long-term N deposition can reduce the biomass and activity of bacteria and fungi through abiotic and biotic pathways such as soil acidification and exacerbated microbial C limitation, thereby suppressing the mineralization of soil organic matter (SOM)[85,86]. Furthermore, N deposition can reshape microbial community structure and diversity, directly interfering with soil N cycling. It can increase the relative abundance of copiotrophic taxa such as Bacteroidetes, Gemmatimonadetes, Actinomycetes, and Firmicutes, which preferentially utilize labile organic C and have higher requirements for inorganic N. Simultaneously, elevated N input can reduce the relative abundance of oligotrophic taxa, including Verrucomicrobia, Acidobacteria, and Basidiomycetes among fungi, which primarily acquire nutrients by mineralizing complex SOM. This shift can decrease activities of various extracellular enzymes involved in SOM decomposition, thereby affecting the release and transformation of soil N[86−88]. In addition, long-term input simplifies community structure and increases the gram-positive-to-gram-negative ratio, indicating a strategic shift from a 'rapid investment–return' to an 'energy-saving tolerance' mode[27,89].
(2) N effect on the activity of enzymes involved in soil N transformation. Studies demonstrate that elevated N deposition directly constrains the rate of organic N mineralization by strongly suppressing microbe-derived N-releasing enzymes, such as glycine aminopeptidase, while exerting minimal effect on nonspecific proteases. This selective inhibition effectively slows the rate-limiting step in the depolymerization of organic N[67]. The resulting pronounced increase in C-acquiring enzyme activity, coupled with only marginal changes in N-acquiring enzymes, significantly raises the enzymatic C/N ratio[90,91]. These results suggest that microbial communities can adapt to excess N by shifting their enzymatic investment patterns, triggering a fundamental change in their resource-allocation strategy from mining N toward securing C and acquiring P[89]. Consequently, the soil N cycle is progressively steered toward a new dynamic paradigm characterized by accelerated nitrification rates and enhanced microbial N immobilization.
(3) N effect on N cycling-associated microbial functional genes. An increasing number of studies focus on key functional genes for N fixation (nifH), nitrification (amoA), and denitrification (nirS, nirK, nosZ), as their abundance often serves as a proxy for potential N transformation rates. Their responses to N addition are highly variable and rate-dependent, shaped by N application rates, forest types, and soil properties[92,93]. This variability leads to divergent effects on N transformation rates across biomes. For instance, in tropical/subtropical forests, N addition often decreases archaeal amoA while increasing nirK abundance[92,94]. Conversely, in temperate/boreal forests, it can enhance bacterial amoA but reduce denitrifier genes[71,94,95]. A recent meta-analysis showed that N addition increased the abundance of archaeal and bacterial amoA genes but suppressed the abundance of the N-fixing gene nifH[94]. In contrast, responses of denitrification genes (nirS, nirK, nosZ) are generally less consistent and often non-significant[93]. This response pattern highlights a fundamental shift in the soil N cycle under enrichment: deceleration in N fixation, pronounced acceleration in nitrification, and variable impact on denitrification, which can steer the soil N cycle toward greater inorganic N turnover and loss potential[94,96] (Fig. 4).
(4) N effect on rhizosphere processes. Root exudates, which include sugars, amino acids, organic acids, and phenolic acids, represent a crucial plant adaptation mechanism and a significant carbon flux (5%–21% of net fixed C) into soils[97]. They critically regulate soil N transformations, including mineralization, nitrification, immobilization, denitrification, and DNRA[98,99]. First, root exudates can stimulate N mineralization by supplying labile C, which enhances microbial and enzyme activity[100,101]. Second, root exudates suppress nitrification by releasing biological nitrification inhibitors (BNIs), which immobilize NH4+ and facilitate plant N absorption due to the lower pH in the rhizosphere[102]. Third, root exudates can enhance microbial N immobilization due to their low C/N ratio and regulate denitrification by providing energy and redox conditions[103]. Fourth, root exudates favor DNRA over denitrification under C-rich, anaerobic conditions[99]. In sum, N inputs can alter mycorrhizal types and the quantity and quality of root C input, modifying exudate composition and C/N stoichiometry, and further altering microbial nutrient limitations, gene expression, and enzyme activities[104,105]. These changes are expected to alter rhizosphere N cycling. However, the mechanisms by which N deposition influences specific rhizosphere N-cycle processes remain poorly understood. Comprehensive, long-term studies are required to clarify the mechanisms underlying N deposition's effects on rhizosphere N losses.
-
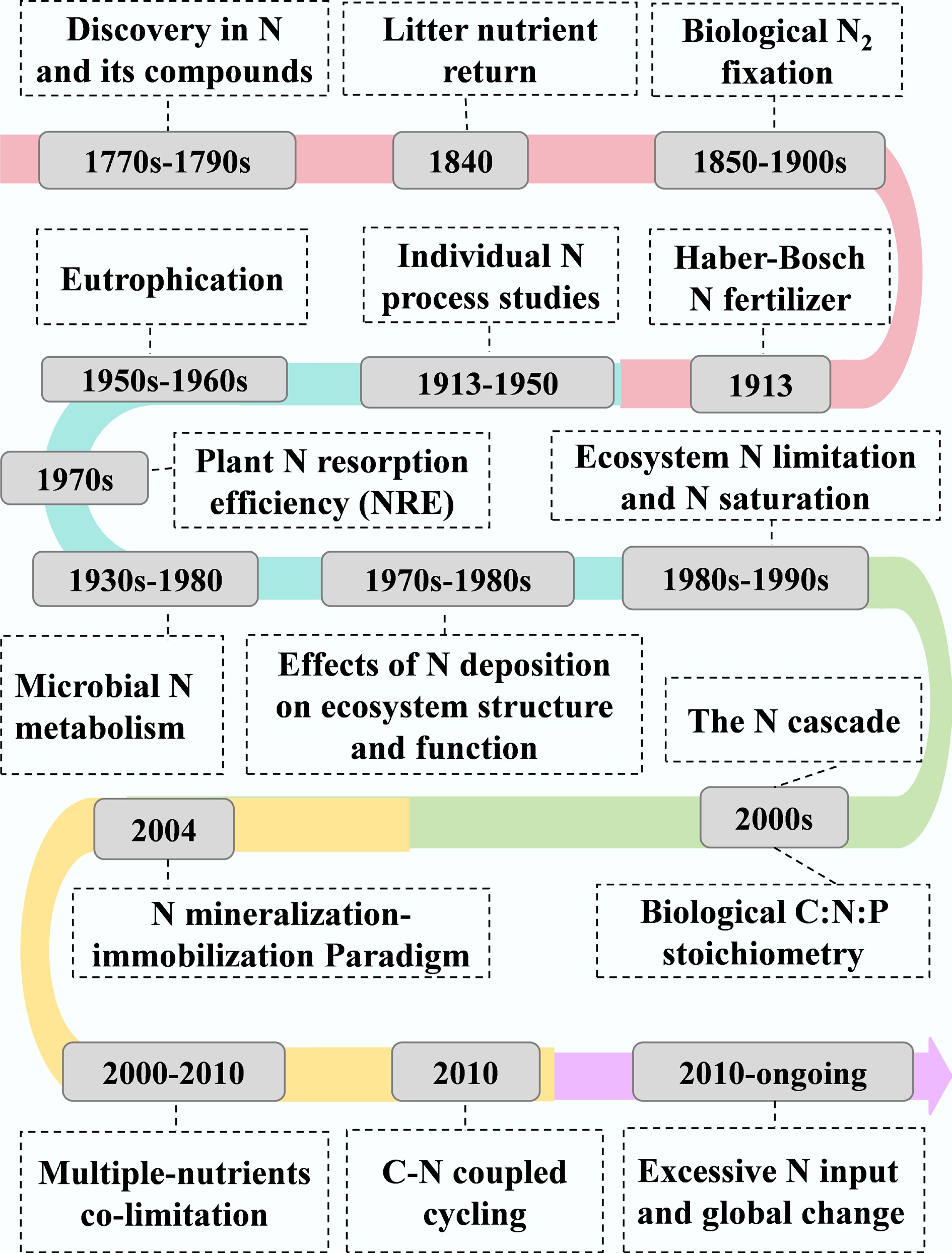
Nitrogen was formally named the 7th element of the periodic table by Jean Antoine Claude Chaptal (1,756–1,832) in 1790. Over the past two centuries, N biogeochemical cycling has evolved from the exploration of phenomena and processes to the in-depth elucidation of mechanisms and multi-element coupling in the context of global change. A critical thread in this ecological evolution is the growing recognition of anthropogenic disruption on the ecosystem N cycle, particularly through atmospheric deposition of reactive N and its cascading consequences for forest ecosystems. This historical perspective can be categorized into three distinct stages (Fig. 5).
Stage (I): foundational discoveries (late 18th–late 19th century)
-
This era established the fundamental components of the N cycle and the perceived balance of the natural N cycle. Key milestones included the identification of crucial N compounds (e.g., NO, NH3, and HNO3) by chemists such as Priestley since 1772, and the important role of litter N returning in forest nutrient retention[106]. The confirmation of biological N fixation highlighted the primary natural pathway for N entry into ecosystems[107]. Collectively, this stage describes a largely closed and self-regulating N cycle, providing an essential baseline against which the magnitude and consequences of future atmospheric N deposition can be quantified and contextualized.
Stage (II): ecosystem processes and mechanisms, and the emergence of the N deposition problem (early 20th–late 20th century)
-
The mid-20th century marked a shift toward mechanistic and ecosystem-scale research on N cycling. Key microbial processes such as mineralization, nitrification, immobilization, DNRA, and denitrification were intensively studied[108,109]. Since the 1980s, atmospheric N deposition has emerged as a central ecological issue, with pioneering studies documenting its cascade impacts on forests and freshwater systems[110−112]. This period gave rise to foundational concepts such as ecosystem N saturation[2], which described sequential forest decline under chronic N loading. The concept of N resorption efficiency was developed to assess alterations in plant nutrient use strategies[113,114]. This stage culminated in the 'Nitrogen Cascade' framework[110], which illustrates how increasing reactive N can sequentially exacerbate watershed eutrophication, soil acidification, climate change, and biodiversity loss. This stage redefined N from a historically limiting nutrient to a pervasive environmental pollutant, with atmospheric deposition emerging as a primary dispersal vector for its widespread distribution.
Stage (III): integration with global change and sustainable management (21st century and ongoing)
-
During this stage, biological stoichiometry coupled the N cycle with C and P, and the theory of multi-nutrient co-limitation replaced the simplistic view of single-nutrient limitation, emphasizing that N deposition effects can be mediated by P[115,116]. C-N coupled cycling has received widespread attention for predicting ecosystem C sequestration and its feedback to climate change under elevated N deposition[40,42]. Current research also links excessive N inputs to global challenges, including climate feedbacks and biodiversity loss, and frames N management as critical for achieving Sustainable Development Goals[24,117]. In China, studies on the ecological effects of N deposition began in 2002 at the Dinghushan Nature Reserve[4,34,42,72]. Since then, many N addition experiments have been established across different ecosystem types, including forests, grasslands, shrublands, and croplands.
-
This review examines the historical development of research on forest N biogeochemical cycling, a field that has been fundamentally transformed by global-scale atmospheric N deposition. Over the past two decades, advances in key methodologies and conceptual frameworks have substantially enhanced scientific understanding.
First, molecular biological tools such as metagenomics, metatranscriptomics, and proteomics offer unprecedented opportunities to describe microbial regulatory networks of functional N cycling genes. Coupling these approaches with stable isotope labeling (e.g., 15N2, 15NH4+, and 15NO3−) allows precise tracking of N transformation pathways and associated microbial metabolic fluxes. For example, DNA-stable isotope probing (DNA-SIP) directly links metabolic function to microbial identity, clarifying niche differentiation within N-cycling networks[118−120]. Second, the view of C-N-P stoichiometric coupling redefines the framework for N-induced ecological effects, shifting from nitrogen's isolated role to a balanced multi-element synergistic regulation. For example, N deposition modulates the nonlinear relationships between microbial C use efficiency (CUE) and N use efficiency (NUE) by altering microbial stoichiometric imbalances. High N inputs can exacerbate C–P imbalances in the organic layer and reduce NUE, whereas P supplementation can restore microbial functional potential by mitigating stoichiometric constraints[121,122]. Third, biotic interactions and interface processes in regulating N cycling have attracted increasing attention, specifically highlighting the ecological contributions of the rhizosphere and mycorrhizal networks[100,123].
However, current understanding remains limited in terms of underlying mechanisms and predictive certainty. Moving forward, interdisciplinary integration is essential to overcome existing research bottlenecks, with emphasis on the following key directions.
(1) Integrating novel technologies and methods: Deep integration of multi-omics with high-resolution in situ techniques like stable isotope tracing (NanoSIMS) is needed. This will quantitatively link the activity of key functional genes (e.g., nifH, amoA, and nosZ) to actual microbial metabolic fluxes, mapping regulatory networks in soil N transformation.
(2) Quantifying C-N-P-H2O coupling mechanisms: The coupling mechanisms among key elements, such as C, N, and P, and water, are complex and fundamental to understanding the functioning and stability of forest ecosystems. Water acts as a carrier for nutrient transport, linking belowground with aboveground, and individual transpiration with ecosystem hydrology. The coupling of C, N, and P use efficiencies across forest types, especially across different mycorrhizal associations, should be systematically quantified to identify critical thresholds where microbial community structure may drive these changes under chronic N inputs.
(3) Elucidating plant-microbe-soil interactions: Exudates not only provide energy for microorganisms but also stimulate the rhizosphere priming effect, significantly accelerating soil organic matter mineralization and shaping soil N cycling. It remains unclear, however, how enhanced or reduced root exudation due to environmental change (e.g., elevated N deposition) affects soil N biogeochemical cycles, such as BNF, nitrification, and denitrification, and determines the trade-off between mycorrhizal C sequestration and nutrient transport. This requires deciphering chemical and molecular signaling in plant-microbe-soil interactions.
(4) Assessing interactive effects among global change factors: Interactions among atmospheric N deposition, climate warming, and altered precipitation can modulate—or even override—the ecological impacts of individual drivers. Future research priorities should include establishing long-term, multi-factor experimental platforms in emerging low-latitude N deposition hotspots, considering these regions are also biodiversity hotspots and are subject to frequent extreme climate events (e.g., warming and heatwaves, changes in precipitation patterns, and typhoons). It is urgent to address the synergistic effects of these stressors on ecosystem N cycling and overall ecosystem stability.
(5) Evaluating ecosystem responses to the declining rates of N deposition: Most existing studies have focused on the effects of elevated N deposition on forest ecosystems. As N deposition rates decline, however, there is growing scientific concern about whether and how forest ecosystems can recover following reductions in N input. Future experimental and modeling approaches are required to predict the biogeochemical and ecological responses of forest ecosystems to the decline in N deposition, both individually and in combination with other global change factors.
-
The authors confirm contributions to the paper as follows: Xiankai Lu: study conception and design, writing, revision and funding acquisition; Xiaomin Zhu: data collection and analysis, draft manuscript preparation and revision; Weibin Chen: data collection and analysis, draft manuscript preparation and revision; Yushu Gao: draft manuscript preparation; Abdoul Kader Mounkaila Hamani: draft manuscript preparation; Lasisi Hammed Tobiloba: draft manuscript preparation; Nazar Abbas: draft manuscript preparation; Junbing Bai: draft manuscript preparation; Zhihao Qiu: draft manuscript preparation; Jun Pan: draft manuscript preparation; Nimra Maqsood: draft manuscript preparation; Zebing Xiao: draft manuscript preparation; Yingxue Xuan: draft manuscript preparation; Jiangming Mo: draft manuscript preparation and suggestions. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
-
This work was supported by Guangdong S&T Program (Grant No. 2022B1111230001), National Natural Science Foundation of China (Grant Nos 32371735, 32271687), Guangdong Basic and Applied Basic Research Foundation (Grant No. 2024A1515011088), and Guangzhou Science and Technology Plan Project (Grant No. 2024A04J4592).
-
The authors declare that they have no conflict of interest.
-
Anthropogenic N deposition has substantially altered terrestrial N cycling at global scale, and human interference with the N cycle has exceeded the safe operating space for humanity.
The hotspots of N deposition have shifted from developed regions to developing ones.
Despite a global decline in atmospheric N deposition, levels remain high in developing countries, where accumulated N deposition is projected to further increase in the future.
Forest soils act as a significant sink for atmospheric N deposition, which can accelerate soil carbon sequestration, regardless of ecosystem N status and climate zones.
Nitrogen biogeochemical cycling has evolved from the exploration of phenomena and processes to the in-depth elucidation of mechanisms and multi-element coupling in the context of global change.
-
# Authors contributed equally: Xiankai Lu, Xiaomin Zhu, Weibin Chen
Full list of author information is available at the end of the article. - Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Lu X, Zhu X, Chen W, Gao Y, Hamani AKM, et al. 2026. Nitrogen biogeochemical cycling in forest ecosystems with the globalization of nitrogen deposition. Nitrogen Cycling 2: e015 doi: 10.48130/nc-0026-0002
Nitrogen biogeochemical cycling in forest ecosystems with the globalization of nitrogen deposition
- Received: 05 November 2025
- Revised: 12 December 2025
- Accepted: 12 January 2026
- Published online: 28 February 2026
Abstract: Nitrogen (N) is an essential nutrient for living organisms and a key limiting factor in ecosystem productivity, influencing both ecosystem structure and function. Over the past fifty years, increased anthropogenic N deposition has substantially altered terrestrial N cycling, resulting in cascading effects such as soil acidification, nutrient imbalances, changes in carbon sequestration, and impacts on biodiversity within forest ecosystems. While previous reviews have examined specific aspects of N deposition, few have systematically integrated multi-scale interactions between global N deposition patterns and the entire N biogeochemical cycle in forests. This review provides a comprehensive overview of research progress on forest N biogeochemical cycling in the context of globalized N deposition. Key topics include N deposition dynamics, changes in forest N status and stocks, and critical N-cycling processes, including biological N fixation, mineralization, nitrification, assimilation, and loss pathways. The review also traces the historical development of N cycling research, identifying three distinct stages since the 1860s. In addition, it examines underlying mechanisms, including microbial transformations and plant–soil interactions, and highlights theoretical advances in forest N cycling. By integrating patterns, processes, and mechanisms, this review enhances understanding of ecosystem responses to anthropogenic N enrichment and identifies critical research directions to address these challenges under global change.