-
Wheat (Triticum aestivum L.) is one of the most widely cultivated cereal crops and serves as a cornerstone for global food and nutritional security[1]. As the global population continues to grow, wheat production must increase steadily to meet rising demands. However, achieving this goal is complicated by the challenges posed by climate change, which is driving an increased frequency of extreme weather events such as heat waves, drought, and salinity intrusion. These environmental stresses not only threaten the stability of global wheat production but also limit the potential for achieving yield gains[1]. Therefore, understanding the mechanisms by which wheat adapts to abiotic stress has become an urgent priority in crop biology and breeding.
Crop yield is a complex trait determined by multiple interacting factors, among which spike architecture and development play a central role[2]. The spike, as the direct yield-determining organ, defines the number of spikelets and fertility of florets, ultimately influencing grain number per spike (GNS), and grain size[3]. Recent advances in wheat functional genomics have improved our understanding of spike development and its applications in breeding practices[4]. However, in natural environments, the genetic potential of wheat yield is frequently constrained by abiotic stresses, which exert profound effects on both vegetative and reproductive growth[5−7]. Among these, stress-induced alterations in spike development are of particular concern because they directly compromise reproductive success and harvestable yield.
Abiotic stress can impact spike development in diverse ways. For instance, drought stress during the reproductive phase often reduces spikelet number and grain setting[8], while heat stress disrupts floret fertility, accelerates spike development at the expense of yield components, decreasing the GNS[9]. Salinity stress, on the other hand, affects spike morphology and grain filling by perturbing ion homeostasis and triggering oxidative stress[7]. Similarly, cold stress during sensitive stages of spikelet and floret formation can also result in severe sterility[10]. These stresses act not only as environmental challenges, but also as developmental signals that interfere with the regulatory networks controlling spike architecture. Consequently, understanding how abiotic stresses intersect with developmental pathways is crucial for designing strategies that safeguard yield under changing climatic conditions.
In this review, recent advances in understanding the impacts of abiotic stresses on wheat spike development, with an emphasis on the potential loci/genes, and molecular mechanisms that they are involved in, are summarized. Emerging research directions were also highlighted that may provide new strategies to enhance spike resilience and optimize yield under adverse environments. By bridging developmental biology with stress physiology, such insights are expected to contribute to the development of stress-resilient wheat varieties, ultimately ensuring stable productivity in the face of global climate challenges.
-
Grass crops such as rice, wheat, and maize exhibit a unique evolutionary trajectory in meristem development, which plays a critical role in the coordinated diversification of inflorescence architecture across this large plant family[11−13]. Wheat produces a spike-like inflorescence, and in most cultivated wheat varieties, each rachis node of the spike bears a single sessile spikelet, which is different from that of rice and maize[4,12,13]. The wheat spike development is composed of several programmed phases with distinct meristem morphology[14]. During early spike development, the shoot apical meristem (SAM) is transferred into the inflorescence meristem (IM), shortly after the floral transition from vegetative phase to reproductive phase, after which the spikelet meristems (SM's) are produced along both sides of the rachis at the double-ridge stage. The SM subsequently gives rise to floral meristems (FM's), ultimately forming an unbranched spikelet structure following the SAM-IM-SM-FM transition pathway (Fig. 1). The activity of the IM determines the number of spikelet meristems initiated; after a specific number of SM's are formed, the IM activity ceases and is terminated by a terminal spikelet. Thus, the number of spikelets per spike (SNS) is determined at the early spike development stage. The duration of IM activity is crucial; prolonging it increases the SNS. The IM phase can be further divided into the elongation stage, and the single-ridge, and double-ridge stages, while the SM stage includes the differentiation of glume primordia, and the FM stage corresponds to floret primordia formation[13] (Fig. 1). By the end of these stages, the basic structure of the inflorescence is fully established. At late spike development, when all floret primordia are produced, the floral development and abortion processes are divided into seven stages, including the terminal spikelet stage, white anther stage, green anther stage, yellow anther stage, tipping stage, heading stage, and anthesis, in which floret fertility plays an important role in the determination of final grain number[15]. Extending the duration from green anther stage to anthesis could enhance assimilate accumulation in the spike, and increase the fertility of the floret during the post-anthesis phase[15].
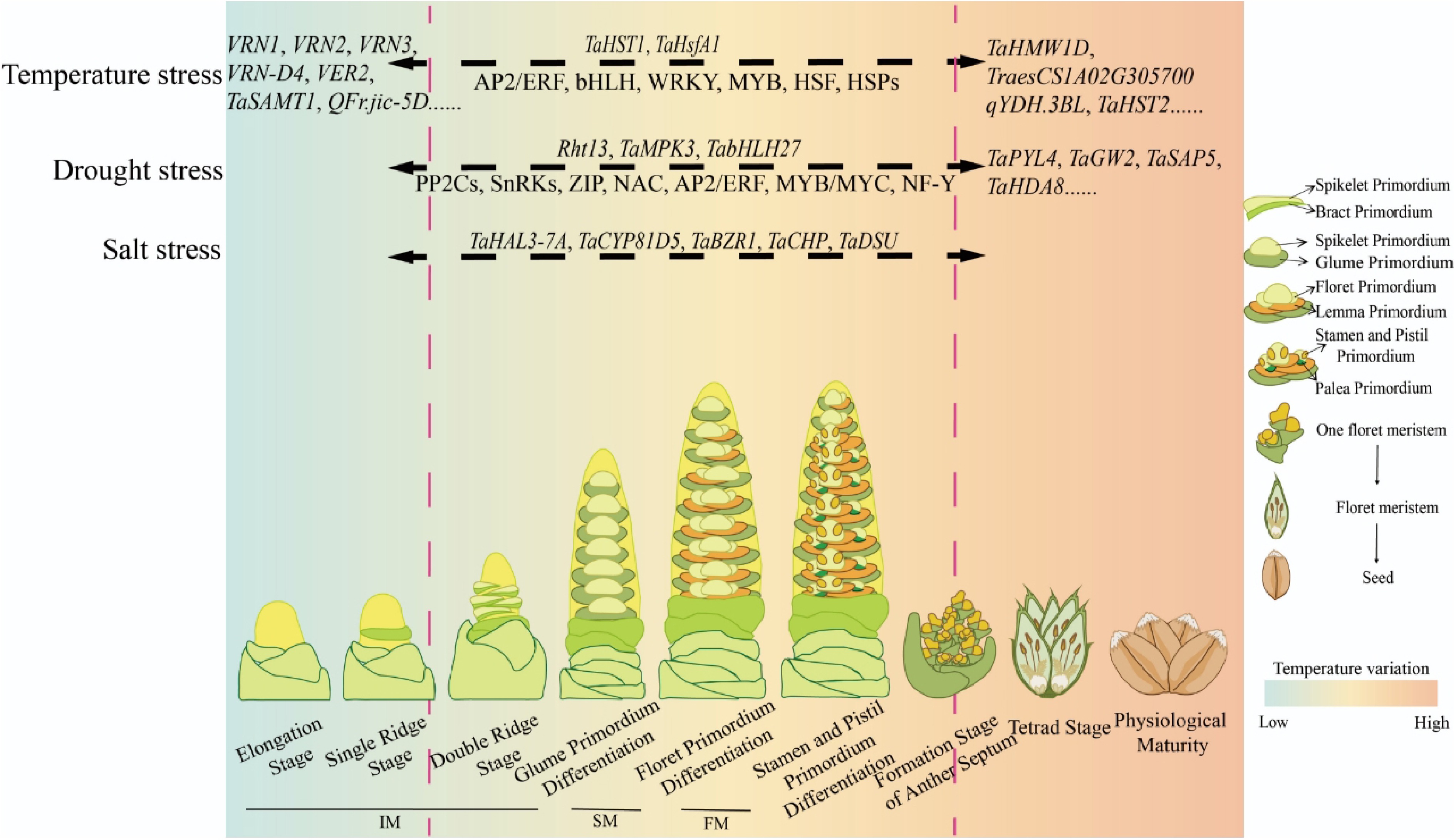
Figure 1.
Spike development and candidate genes involved in the stresses in wheat. Abbreviations: IM, inflorescence meristem; SM, spikelet meristem; FM, floral meristem; Bidirectional dashed arrows indicate functional genes or transcription factor families identified by omics analyses that may influence multiple or all stages of spike development.
The GNS that is determined by SNS and floret fertility, along with the grain weight and the number of spikes per plant, are the three key components determining crop yield. Among these, the GNS is directly influenced by the development of the spike, which is regulated by a complex genetic network interacting with external environmental conditions[4]. Therefore, elucidating the key genes and molecular mechanisms that mediate the interaction between abiotic environmental factors and yield components is essential for advancing crop genetic improvement. Abiotic stress refers to a range of environmental threats that plants encounter, including drought, high/low temperature, salinity, and waterlogging, among others. With ongoing global warming and the increasing frequency of extreme weather events, crop growth and development face severe challenges. In both winter and spring wheat, the transition of the IM's typically occur after spring regrowth, during the reviving stage, which is often vulnerable to late spring cold spells. The spike at the programmed developmental phases demands substantial resources such as water, nutrients, and light, making the yield highly susceptible to climatic conditions. Although the physiological and biochemical impacts of individual stress factors have been increasingly revealed, and their effects on various plant organs, especially roots and leaves, have been systematically summarized[16,17], relatively little is known about how abiotic stress influences wheat spike development and the genetic bases underlying the stress response of wheat spike.
-
Global climate change directly intensifies temperature rise, causing ecosystem fluctuations and imbalances while triggering extreme temperature events—both heatwaves and cold spells. Winter wheat, the world's dominant wheat species, is favored over spring wheat for its higher yield and superior grain quality. However, its extended growing cycle increases its exposure to frequent and severe temperature stress. Consequently, research on temperature stress represents a major focus in wheat breeding. Advances in wheat genome assembly, multi-omics technologies, and the accelerating pace of global climate change are driving rapid progress in this field.
Wheat spike development is a highly temperature-sensitive process that largely determines final grain number and yield. As global climate change intensifies, wheat is increasingly exposed to fluctuating temperatures, including low and freezing temperatures in winter and early spring, heat waves around anthesis, and persistent warm nights during the grain-filling period. Each of these temperature regimes imposes distinct physiological and molecular constraints on spike development, disrupting reproductive organ formation and carbon allocation and corresponding spike development.
-
Global warming, coupled with the breeding emphasis on high-yield and superior grain, has led to the neglect of cold tolerance in winter wheat selection, resulting in cultivars with reduced frost resistance. Extreme low temperatures frequently occur in major wheat-growing regions such as Australia, China, Europe, and the United States. When canopy temperatures fall below 0 °C, late spring frosts severely damage vegetative and reproductive tissues, particularly during the highly temperature-sensitive stages of spike and floret differentiation, ultimately leading to significant yield reduction[10]. At the booting or early floret differentiation stages, low temperature and frost stress highly inhibits spikelet initiation and delays floret maturation. Morphologically, these stresses reduce GNS and thousand-grain weight (TGW). Physiologically, cold exposure alters sucrose metabolism by enhancing sucrose phosphate synthase activity, but suppressing sucrose synthase and invertase, leading to sucrose accumulation in young spikes. Concurrently, low temperatures elevate abscisic acid (ABA) levels while reducing auxin (IAA) and gibberellin (GA) contents, collectively repressing spike elongation[18]. And at the molecular level, transcriptome analyses of young spikes in response to late spring coldness revealed extensive reprogramming of carbohydrate metabolism, hormone signaling, and circadian rhythm pathways. Transcription factors such as AP2/ERF, bHLH, WRKY, MYB, and HSF families were regulated under cold stress, coordinating cold-responsive gene expression[19]. Moreover, temperature-sensitive genic male sterile lines exhibited DNA hypermethylation[20] and microRNA (miR167, tasiRNA-ARF)-mediated regulation of auxin signaling, linking epigenetic control to male sterility under cold stress[21]. In addition, transcriptomic profiling of durum wheat under 5 °C at the reproductive stage identified nearly 900 differentially expressed genes involved in photosynthetic activity, lipid and carbohydrate synthesis, and amino acid and seed protein accumulation, alongside 28 transcription factors and 31 long noncoding RNA's, suggesting that lncRNA-miRNA-TF networks contribute to moderate cold tolerance[22]. These findings collectively suggest that cold stress interferes with carbon partitioning and hormonal homeostasis, leading to delayed or aborted floret development and consequent yield loss.
Current genetic and molecular research on low-temperature response mechanisms in wheat remains relatively limited. Freezing temperatures restrict the geographical distribution of wheat and cause major yield losses. To date, freezing resistance-associated loci in wheat, including Fr1 and Fr2, both containing several tandemly duplicated C-REPEAT BINDING FACTOR (CBF) genes that regulate cold-responsive pathways[23]. Vernalization, required for flowering in winter wheat, involves genes such as VRN1 (VERNALIZATION1), VRN2, VRN3/TaFT1 (FLOWERING LOCUS T1), VRN-D4, and VER2 (VERNALIZATION-RELATED 2)[24], which are critical for the SAM-IM transition to start flowering. Winter wheat exhibits significantly stronger cold hardiness than spring wheat. Vrn-A1 not only determines the spring/winter growth habit but also contributes to freezing tolerance. Notably, distinct haplotypes within the Vrn-A1–Fr-A1 region on chromosome 5AL-where Fr-A1 lies ~2 cM from Vrn-A1-underlie natural variation in freezing tolerance and have been strategically exploited in breeding programs[25]. Freezing tolerance typically declines with the onset of reproductive growth, linking cold response to the developmental phase[26]. Studies using maintained vegetative phase (mvp) diploid wheat mutants, which lack functional VRN-1 revealed that plants without VRN-1 (mvp/mvp) retain high freezing tolerance, whereas those with functional VRN-1 (Mvp/–) flower normally but display reduced freezing tolerance and lower expression of CBF and COR genes. Additional evidence from VRN-1 promoter mutants and expression analyses indicate that VRN-1 initiates a regulatory cascade suppressing cold acclimation under long day conditions, although other photoperiod-regulated genes are also involved. The results demonstrate that allelic variation in VRN-1 alone is sufficient to confer differences in freezing tolerance, suggesting that QTL for freezing tolerance previously mapped to this chromosomal region represents a pleiotropic effect of VRN-1, not the effect of a separate, closely linked Fr-1[26]. Another QTL, QFr.jic-5D, was mapped near Vrn-D1 on chromosome 5D[27]. Breeding efforts have introduced the recessive vrn-D1 allele from the cold-tolerant cultivar Shijiazhuang 8 into Shimai 12, producing the winter cultivar Lunxuan 103, which exhibits enhanced frost tolerance and reduced winterkill compared to spring types. However, how VRNs genes regulate meristems' activity under low-temperature stress at early spike development waits for further investigation.
Wheat possesses 37 CBF genes, five INDUCER of CBF EXPRESSION (ICE) genes, and eleven COLD REGULATED (COR) genes. Phylogenetic analysis has revealed that ICE1 and COR genes exhibit closer evolutionary relationships within monocots compared with CBF genes. Functional studies demonstrate that overexpression of TaCBF14 or TaCBF15 in barley promotes the upregulation of downstream HvCOR14b, thereby increasing barley's cold tolerance[28]. In wheat, both ABA-dependent COR genes and ABA-independent COR genes are significantly upregulated under cold stress[29]. Additionally, plant hormones also regulate cold signaling. SnRK2's are core kinases in ABA signaling, and are induced by low temperatures. For example, PKABA1, encoding an ABA-responsive SnRK2 protein kinase, is induced by drought, low temperature, and osmotic stress, while TaSnRK2.3 and TaSnRK2.4[30] are cold-induced. Epigenetic regulation also contributes to wheat cold response. Salicylic acid (SA), a key defense hormone, can be converted into methyl salicylate (MeSA) through the catalytic activity of SAMT enzymes[31]. MeSA has been shown to enhance cold tolerance in multiple species, including Arabidopsis, tomato, wheat, and several fruit trees[27,32]. In Arabidopsis, this enzyme is S-ADENOSYL-L-METHIONINE-DEPENDENT METHYLTRANSFERASE1 (SAMT1), which uses SA as the substrate and forms MeSA[33]. In wheat, the orthologous gene TaSAMT1 was identified by transcriptome analysis. Overexpression of TaSAMT1 increased MeSA accumulation, reduced SA levels, and markedly improved freezing tolerance, whereas knockout lines were freezing-sensitive. Importantly, yield-related traits such as spike length, spikelet number, and TGW were unaffected under field conditions, suggesting that TaSAMT1 is a promising target for breeding cold-tolerant wheat varieties[34]. However, the direct evidence about the functions and underlying mechanisms of these genes in regulating spike development under low-temperature stress is still missing. Furthermore, cold tolerance in wheat is highly complex. Beyond conserved mechanisms shared with Arabidopsis, whether wheat-specific regulatory pathways exist remains to be elucidated.
-
Global warming threatens wheat production by reducing both yield and grain quality, largely due to heat stress[9]. Heat stress during reproductive stages, particularly during meiosis and pollen development, causes severe fertility loss. Elevated temperature reduces pollen viability, disturbs anther morphology, ultimately decreasing spike fertility and grain weight[35]. At the whole-plant level, heat stress shortens the grain-filling period and promotes premature aging of photosynthetic tissues, thereby limiting carbohydrate supply to developing spikes. Physiologically, this leads to reduced floret fertility and decreased SNS and TGW. Recent physiological and genetic analyses have demonstrated that spike fertility and spike harvest index, two critical determinants of yield formation, are highly sensitive to high-temperature stress. Under heat exposure, disrupted carbon assimilation and restricted assimilate partitioning toward developing spikes weaken sink strength and reproductive success. A genome-wide association study (GWAS) identified 109 marker-trait associations under heat stress, including loci related to spike fertility and spike harvest index on chromosomes 1B, 3B, 4A, 6B, and 7D, with candidate genes encoding heat-responsive proteins such as β-glucanase, α/β-hydrolase, protein kinase, and HSP's, suggesting a coordinated molecular response to maintain carbon flux and spike fertility[36]. GWAS and QTL mapping identified key loci on chromosomes 2A, 4B, and 6B associated with post-anthesis heat tolerance and stay-green traits[37]. Notably, chromosome 5D was shown to confer protection during meiosis, as its absence resulted in enhanced susceptibility to heat-induced sterility[38]. Furthermore, TaHMW1D, a high-molecular-weight glutenin gene, produces multiple transcript variants under heat stress, especially in distal spike grains, suggesting structural plasticity of seed storage proteins as a compensatory mechanism[39]. Collectively, these results indicate that high temperature disrupts both male and female gametophyte development and carbon assimilation, leading to reduced GNS and filling rate.
Unlike daytime heat stress, elevated nighttime temperature (HNT) mainly increases respiration and shortens developmental duration[40,41]. Continuous warm nights during anthesis and grain filling accelerate senescence and increase the number of sterile spikelets, reduce effective tiller number and GNS, with yield reductions of 20%–25% under HNT conditions[40]. Physiologically, increased respiratory carbon loss and reduced assimilate partitioning between source and sink tissues disturb energy balance, leading to poor spike fertility and degraded end-use quality[41]. Gene-based haplotype analysis identified a haplotype Hap1 of TraesCS1A02G305700 exhibited higher biomass and spike number under HNT[42]. Across different thermal stresses, temperature-mediated changes in sucrose metabolism, hormone signaling, transcriptional regulation, and epigenetic modification form an interconnected network governing spike development. QTL's and haplotypes associated with thermal tolerance (e.g., on 2A, 5D, and 6B) provide valuable molecular targets for marker-assisted breeding. Integrating multi-omics approaches, including transcriptomics, metabolomics, and epigenomics will deepen our understanding of spike developmental plasticity under temperature fluctuations. Ultimately, dissecting these regulatory cascades will facilitate the breeding of climate-resilient wheat cultivars capable of maintaining spike fertility and yield stability under temperature stress.
During the grain filling stage, heat stress impairs carbon assimilation and starch biosynthesis, leading to reduced yield and quality. Multiple wheat heat stress tolerance (HST) loci have been identified, including qYDH.3BL and TaHST1[43,44]. The qYDH.3BL QTL is associated with increased yield performance under hot and dry environments. Its delimited genomic region (~ 690 kb) contains 12 putative genes with a sequence variant in the promoter region of the gene Seven in absentia (TaSINA). Near-isogenic lines carrying the positive allele at qYDH.3BL under-expressed TaSINA, and had increased vigor and water use efficiency in development, as well as increased biomass, grain number and grain weight following heat stress[43]. TaHST1 is essential for HST at both the seedling and adult stages, with gene losses in its region, compromising tolerance, and resulting in significantly lower values of GNS, grain weight per spike, TGW, grain length, and grain width under either thermo-stressed or control conditions[44]. In addition, TaHST2 enhances the conversion of soluble sugars into starch during heat-stressed grain filling, thereby enabling faster grain filling and higher yield potential[45].
High temperatures disrupt key developmental and physiological processes, and even modest increases above the optimum can cause significant yield losses. To mitigate these effects, wheat activates various protective mechanisms such as ROS scavenging and the induction of heat shock proteins, and several heat-tolerance genes have been identified, including TaGCN5 and TaOPR3[46]. Heat shock transcription factors (HSF's) are key regulators of heat-induced transcriptional reprogramming. Upon exposure to heat stress, HSF's recognize conserved heat shock elements (HSE's) in promoters of heat-inducible HSP genes. In wheat, multiple HSF genes, including TaHsfA2d, TaHsfA6f, TaHsfC2a, and sHsp26[44,47], function in heat signaling and stress response pathways. Among these, HsfA genes primarily exert a positive regulatory function in abiotic stress response, in which TaABF5b, a critical regulatory factor of the ABA signaling pathway, has been identified as a modulator of the expression of TaHsfA2h. TaHsfA2h interacts with TaHsfC2a, co-regulating key heat-responsive genes such as TaNCED2B, TaPOD4, and TaHSP26, thereby forming a positive regulatory module (ABF5b-HsfA2h/HsfC2a-NCED2b/POD4/HSP26) that enhances wheat heat tolerance. TaHsfA1 is essential for heat tolerance at both the seedling and grain-filling stages. Knockout mutants show severely reduced seedling survival under high temperature, and produce shrunken grains with lower TGW, highlighting its critical regulatory role throughout wheat development under heat stress[47].
During heat acclimation, cereals adjust their metabolism to high temperatures and protect critical cell structures against the effects of high temperatures, and enzymes play a crucial role in this stress defense process. For example, TaSG-D1, a Ser/Thr kinase, negatively regulates brassinosteroid (BR) signaling, and its variant TaSG-D1E286K improves heat tolerance via enhanced phosphorylation of TaPIF4, though at a cost to grain yield[48]. Similarly, the TaSERL2-TaBZR2 module regulates heat tolerance by modulating TaBZR2 stability. Overexpression of TaBZR2 enhances thermotolerance, whereas TaSERL2 promotes degradation of TaBZR2 under stress conditions, demonstrating a negative regulatory mechanism independent of BR signaling[49].
-
Rising global temperatures are anticipated to aggravate crop losses, and agricultural water supplies must be increased by an estimated 17% by 2025 to compensate for global population growth. Drought poses a significant challenge to wheat production globally, leading to substantial yield losses and affecting various agronomic and physiological traits[50]. Plants have strategies to prevent water loss, balancing optimal water allocation to vital organs, maintaining cellular water content, to persevere through periods of drought. Conceptually different from drought tolerance which means the survival of plants under drought stress, water-use efficiency (WUE) refers to the ability of a plant to produce biomass or yield using minimal amounts of water, which measures how efficiently a plant utilizes water to carry out photosynthesis and produce the desired outputs.
Quantitative trait locus (QTL) and GWAS have provided valuable insights into how drought stress reshapes the genetic regulation of wheat spike architecture and reproductive success. In multiple water-deficit field trials, spike-related traits showed high plasticity under drought. A large-scale GWAS across diverse genotypes identified 8 GNS and spikelet fertility loci linked specifically to a drought tolerance coefficient (DTC), which was mapped to chromosomes 1B, 2A, 5B, 6B, and 7D, reflecting how water stress reduces floral fertility gradients from base to tip[51]. Chromosome-specific mapping also revealed novel loci controlling spike fertility and structure. Notably, a 5B:20cM locus (TduRuM_contig25432_1377) showed a stable association with the number of fertile spikes across both favorable and semiarid environments[52]. Likewise, the GA-responsive dwarfing allele Rht13 improved peduncle length, spike length, grain number per spike, and grain weight under drought stress, suggesting that certain semi-dwarf alleles can redistribute assimilates toward the spike rather than merely reducing plant height[53]. In parallel, a high-density linkage map was constructed using genotyping-by-sequencing (GBS) enabled the identification of 85 QTL's across 19 chromosomes for grain yield and spike-related traits under irrigated and water-deficit conditions, explaining 6%–19% of phenotypic variance. Among these, 20 novel loci were located mainly on 1A, 1B, 1D, 2B, 3A, 3B, 6A, 6B, and 7A, and 35 QTL's were functionally linked to drought-responsive genes involved in osmotic regulation and redox balance[54].
Meanwhile, simulating drought stress during the breeding process has been proposed as a useful strategy for screening drought-tolerant lines under otherwise favorable conditions. A GWAS revealed 263 significant marker-trait associations (MTA's), including 246 loci associated with days to anthesis, spike length, SNS, GNS, and TGW[55]. A subsequent multi-environment GWAS further detected 57 stable markers, among which nine were pleiotropic, governing flowering time and spike yield components[56]. These loci provide targets for marker-assisted selection (MAS) to stabilize spike performance under combined heat-drought stress[56]. Collectively, these findings, based on QTL analysis and GWAS, highlight the complex genetic architecture underlying wheat spike development under drought. The integration of chromosome-specific loci (e.g., 5B:20cM, 1B, 2A, 6B, 7D), regulatory alleles such as Rht13, and multi-environment stable markers provides a foundation for dissecting both the structural and physiological bases of spike resilience. By linking spike fertility gradients, assimilate allocation, and flowering-related traits, these studies bridge phenotypic plasticity with genomic control, offering actionable targets for MAS-based breeding of drought-resilient, yield-stable wheat cultivars.
While QTL and GWAS studies have identified key loci controlling spike fertility under drought, multi-omics analyses have now illuminated the underlying molecular and cellular mechanisms by which these loci act to maintain spike development, fertility, and assimilate flow during water deficit. High-resolution RNA-seq analyses under field conditions demonstrate that drought at the early reproductive stages triggers a pronounced reprogramming of gene expression in spike tissues, far more severe than stress at anthesis. Across 115,656 expressed genes, 309 differentially expressed genes (DEG's) were identified, primarily involved in floral organ initiation, photosynthesis, redox regulation, and hormone signaling[57].
Under drought conditions, plants close their stomata, which reduces the intake of CO2, in which ABA mediates stomatal closure and stress-gene upregulation[58]. The ABA signaling pathway has been identified as a central regulator of abiotic stress response in plants, triggering major changes in gene expression and adaptive physiological responses. Currently, extensive research has been conducted on ABA-mediated drought responses in model plants such as Arabidopsis thaliana[59], rice[60], and maize[58,61]. Notably, significant advances have also been made in wheat, especially at the seedling stage[62]. In grass crops, grain number depends on the number of fertile florets at flowering[63,64], and exposure to stress during the young microspore (YM) stage causes a pronounced reduction. At this developmental stage, an adequate assimilate supply to the anthers is critical, with the sub-apical internodes (the two internodes below the spike, TIS), and the flag leaf serving as key organs for assimilate transport[65,66]. Transcriptomic studies have shown that ABA signaling genes, including PP2C's, SnRK's, and ZIP family members are down-regulated in the spike and TIS under water stress at the YM stage. Concurrently, ABA levels increase and correlate positively with cell wall invertase (CWIN), and sucrose:sucrose 1-fructosyl-transferase (1-SST) activities in TIS, suggesting that stress-induced changes in ABA signaling modulate sucrose conversion and allocation, ultimately affecting grain number[8]. PYL proteins, which are abscisic acid (ABA) receptors, play crucial roles in drought stress responses. Overexpression of TaPYL1-1B or TaPYL4 increases wheat ABA sensitivity to improve drought tolerance and increase grain size and weight, tuning water-use efficiency and drought tolerance in wheat[67]. Moreover, TaMPK3 was identified as a candidate interacting protein of TaPYL4. Functional analyses revealed that TaMPK3 overexpression reduces drought tolerance and ABA sensitivity, leading to lower survival rates and decreases in spikelet number, tiller number, panicle length, and grain yield. Conversely, TaMPK3-RNAi lines showed enhanced growth and productivity compared with WT[68]. Meanwhile, several core ABA signaling components such as TaPYL9, TaSnRK2.10[69], and TaABF2[70] in wheat, have been shown to contribute significantly to drought tolerance, while their functions on spike development remains unclear.
The ubiquitin/26S proteasome (UPS) pathway degrades misfolded or unnecessary proteins through ubiquitination and plays a central role in maintaining cellular homeostasis and regulating diverse processes such as hormone signaling, stress response, and plant development[71−73]. Among E3 ligases, RING finger proteins are particularly important. In rice, Grain Width and Weight 2 (GW2), a C5HC2-type E3 ligase regulates grain size by ubiquitinating the cell proliferation regulator WIDE GRAIN 1 (WG1)[74]. Its wheat homolog TaGW2 has been widely studied, with allelic variations linked to drought tolerance. Functional studies reveal that TaGW2 modulates both yield and stress responses. Mechanistically, TaGW2 ubiquitinates and degrades TaARR12, a negative regulator of drought responses, which suppresses bZIP transcription factors. Notably, CRISPR-mediated knockout of TaGW2, combined with TaARR12 knockdown resulted in plants with both improved drought tolerance and higher yields, highlighting the TaGW2-TaARR12 module as a promising breeding target for wheat genetic improvement[75]. In addition to RING-type ligases, stress-associated proteins (SAP's), containing A20/AN1 zinc finger domains, also function as ubiquitin ligases regulating stress adaptation[76]. For instance, TaSAP5 overexpression in wheat increased drought tolerance by maintaining tiller and spike numbers under stress, leading to higher grain weight and grain number without yield penalties under normal conditions, consistent with findings in Arabidopsis and rice[76]. Beyond ubiquitination, other post-translational modifications such as acetylation play equally critical roles in drought stress adaptation[77]. Histone deacetylases (HDAC's), recently termed lysine deacetylases (KDAC's), regulate not only chromatin remodeling but also the stability, activity, and localization of non-histone proteins. In rice and Arabidopsis, HDAC's modulate stress responses and development through selective deacetylation of key regulators. However, their functional roles in wheat remain less understood. Recent work identified TaHDA8 as a crucial regulator of root elongation under drought conditions. Under drought stress, TaHDA8 knockout lines showed higher yield per plant, mainly due to increased GNS. Under stress, reduced TaHDA8 levels restore acetylation-dependent DNA-binding activity of TaAREB3, which subsequently activates TaKOR1 expression to promote root meristem cell proliferation. Moreover, natural variations in the TaKOR1 promoter affecting TaAREB3 binding have been selected in wheat breeding, underscoring the adaptive significance of the TaHDA8-TaAREB3-TaKOR1 module[78]. Nevertheless, the direct functions and mechanisms of these genes or modules in regulation spike development under drought stress are waiting for further investigation.
Different from drought tolerance, WUE of crops prioritizes the balance between maximizing yield and minimizing water consumption[79]. Recently, research on WUE of crops has received increasing attention due to its critical roles in crop production improvement[50]. However, although numbers of QTL's and transcription factors (TF's) in key TF families such as NAC, AP2/ERF, MYB/MYC, bZIP, NF-Y, WRKY, and regulators in various morphological or physiological processes targeting drought adaptation or tolerance have been characterized in wheat[50,80], relatively few key genes were identified to enhance WUE. QTL's for stomatal opening are spotted on chromosome 7A, where TaER2 resides. Further studies revealed that TaER genes can help develop mechanisms to mitigate drought stress in wheat crops[81]. Varieties with enhanced ABA signaling pathways could also improve water-use efficiency. For example, overexpression of TaPYL1-1B genes, an ABA-sensitive gene, consequently increases WUE and photosynthesis. The atypical bHLH TF TabHLH27 directly coordinates both root growth and spike development, thereby affecting WUE and yield-related traits, including spikelet and grain number. The favorable Hap-II allele enhances drought tolerance, increases root system size and improves spikelet number and WUE, and has been positively selected in modern breeding programs[82]. Recently, integrative omics analysis identified 73 QTL's for WUE-traits, in which 85 high-confidence candidate genes, including TaMYB7-A1 were underscored. Overexpression of TaMYB7-A1 enhances photosynthesis, WUE, root development, and grain yield under drought conditions[80].
In summary, these studies revealed that wheat responds to drought stress by coordinating spike development through multilayered regulatory mechanisms, including hormone signaling, ubiquitin/proteasome pathways, metabolic regulation, and transcription factor mediated gene regulatory networks. Understanding and integrating these key molecular modules provides promising strategies for improving drought resilience, while maintaining or enhancing grain yield during breeding[50].
-
Soil salinization affects over 800 million ha worldwide, posing a major threat to crop yield and ecological security. Salt stress induces ionic, osmotic, and oxidative stresses, to which wheat is particularly sensitive, especially during the spring growth period, when surface soil salinity is elevated[83]. High salinity is commonly due to high concentrations of Na+ and Cl- in the soil solution, resulting in hyperosmotic and hyper-ionic conditions, which impede plant absorption of water and nutrients from the soil[84]. Wheat employs multiple mechanisms to cope with the detrimental effects of salinity, in which numbers of key factors were identified in response to salt stress such as TabZIP15, TaASR1-D, TaERF3, TaHAG1, and GCN5[78,85,86]. Na+ exclusion via the key transporter HKT1;5 is central to salt tolerance. Its upstream regulator, TaSPL6-D, represses HKT1;5; natural variation or knockout of TaSPL6-D enhances Na+ exclusion, preserves spike development and improves yield under saline conditions[83]. A group of K+-preferring HKT transporters, TaHKT8, TaHKT9, and TaHKT10 were identified which negatively regulate the wheat shoot K+ accumulation and salt tolerance. A GWAS combined with candidate gene association analysis further revealed that TaHKT9-B substantially underlies the natural variation of wheat shoot K+ accumulation under saline soil conditions. The tae-miR390/TaTAS3/TaARF4 pathway was identified to regulate the salt-induced root development and salt tolerance in wheat, indicative of the trade-off between development and salt tolerance[87].
Crop productivity can be improved by effectively regulating various photosynthesis-related traits, including photosynthetic pigments and photosynthetic efficiency, as well as yield-related traits. Halotolerance 3 (HAL3) is known for its conserved roles in enhancing salt tolerance, cell-cycle control, and cell wall integrity. In wheat, TaHAL3-7A plays a key role in regulating photosynthetic pigments and yield-related traits. It is highly expressed in shoots and grains, and overexpression in wheat and rice increases chlorophyll and carotenoid levels, grain number per spike, and yield per plant. Mechanistically, TaHAL3-7A enhances photosynthesis by upregulating light-harvesting chlorophyll a/b-binding (LHC) genes, and interacts with TaUFD1-3A to coordinate pigment accumulation and grain yield. Combining favorable haplotypes of TaHAL3-7A and TaUFD1-3A further boosts chlorophyll content, TGW, and yield, highlighting its potential as a target for molecular breeding to improve wheat productivity[88]. In addition, TaCYP81D5, a member of the cytochrome P450 monooxygenase (CYP) gene family, enhances wheat salinity tolerance by promoting ROS scavenging. Its expression is induced by salt stress and chromatin modifications, and constitutive overexpression improves both the seedling and reproductive-stage tolerance. Overexpressing lines (TaOE1/TaOE2) produce larger seeds and higher yields under moderate salinity, with Zat12 acting as a crucial component of this ROS-mediated pathway. These findings highlight TaCYP81D5 as a promising target for the development of salt-tolerant, high-yield wheat varieties[89].
BR's are vital plant steroid hormones involved in numerous aspects of plant life, including growth, development, and responses to various stresses. BR signal core transcription factor BRASSINAZOLE-RESISTANT1 (TaBZR1) is significantly upregulated by salt treatment. Overexpression of Tabzr1-1D (a gain-of-function TaBZR1 allele) enhances wheat salt tolerance, accompanied by reduced plant height and spike length, but slightly increased grain length. Mechanistically, TaBZR1 directly activates TaNCED3 to promote ABA biosynthesis and associates with TaGPX2/3 to enhance ROS scavenging. Thus, TaBZR1 confers salt tolerance by coordinately regulating ABA production and ROS detoxification, providing a promising strategy for wheat improvement[90].
Transcriptional regulation is crucial under salt stress, controlling stress-responsive genes to maintain ion balance, antioxidant activity, and growth. For instance, TaCHP, encoding a Cys-, His-, and Pro-rich zinc finger protein, confers enhanced tolerance to saline—alkaline stress in wheat. Overexpression lines displayed stronger germination, photosynthesis, and ROS scavenging, and more importantly, showed increased spike number, grain number per ear, and thousand-grain weight, leading to stable yield gains of ~4% to 14% across multi-year, multi-location field trials without growth penalties under normal conditions. Natural variation analysis further revealed a favorable haplotype (Hap1) associated with stronger stress-induced expression, highlighting TaCHP as a valuable genetic resource for breeding salt-tolerant and high-yielding wheat[91]. While transcription factors play key roles in mediating salt-stress responses, post-translational modifications also provide crucial regulation. Reversible conjugation (sumoylation and desumoylation) of Small Ubiquitin-Like Modifiers (SUMO) is a crucial kind of protein modification. The wheat SUMO protease gene TaDSU confers salt tolerance through desumoylation. TaDSU overexpression enhances salt tolerance in wheat by interacting with the transcription factor MYC2, reducing its sumoylation, and thereby boosting its transcriptional activity, which in turn reinforces TaDSU expression. This regulatory loop leads to lower Na+, higher K+, and improved K+:Na+ balance under salt stress. Beyond stress adaptation, TaDSU also promotes yield-related traits: overexpression lines produce more spikes per plant and larger TGW, resulting in 9.8%–25.0% yield increases in saline soils and 6.8%–9.2% in saline-alkaline fields, without penalties under normal conditions. Moreover, its ectopic expression in Arabidopsis also improves salt tolerance, highlighting its broad role in stress adaptation and agronomic improvement[92]. While more and more key regulators were identified in response to salt stress, and even balance the developmental processes and stress tolerances[83,93], few of them were characterized by showing distinct functions and mechanisms in wheat spike development, which is waiting for further investigation.
-
Recent studies have highlighted the pivotal role of multiple regulations in mediating the crosstalk between stress responses and developmental processes, such as transcriptional control, post-transcriptional modulation, epigenetic regulation, and hormone signaling[6,75,94−96]. Despite the growing body of research, significant knowledge gaps remain. While most studies have focused on stress responses in vegetative tissues at the wheat seedling stage[1,5], research about reproductive development was generally more vulnerable and remain underexplored. Under field conditions, compound heat—drought episodes frequently occur during wheat spike development and early grain filling. Available evidence, largely from field observations and physiological assessments, suggested that such compound events were associated with impaired floral development and fertility, including reduced pollen viability, increased pollen sterility, and decreased stigma receptivity, ultimately shortening effective grain-filling duration and reducing yield[97]. Notably, direct and stage-resolved evidence connecting salt-induced ionic accumulation, or salt signaling perturbations to wheat inflorescence meristem activity, spikelet initiation, or floral meristem determinacy/floral organ patterning, remains scarce (Table 1). Moreover, much of the current understanding derives from model species such as Arabidopsis, rice, soybean, and maize[1,5,6,31], highlighting the requirement for wheat-specific studies that account for its unique polyploid genome and developmental complexity. Addressing these gaps will require integrative approaches that combine genomics, transcriptomics, and functional validation, as well as field-based evaluations under realistic stress conditions.
Although the programmed spike development is critical for GNS, the most important component for the crop yield and QTL related spike morphology traits were mapped[98], limited key genes were identified to regulate spike development[99], particularly under stress conditions. Before heading, the entire wheat spike development is concealed within the leaf sheath, which hinders the real-time observation of spike development and subsequent gene cloning. Previously, several transcriptome profile analyses and multi-omics analysis using meristem tissues uncovered the cascaded gene expression at early spike development, in which genes in different TF families were involved and interact with phytohormones[100]. Subsequently, distinct gene families such as MADS and TCP TFs', as well as particular genes like WFZP, DUO, VRT2, TaSPL17, and TaARF12 were characterized in spike development[101−103]. At the same time, several studies focused on the floral fertility were carried out and QTL's for floral fertility were spotted on different chromosomes[24−26]. Particularly, a number of key genes involved in floral death were identified in cereal crops, including wheat[19]. These genes and loci provide a genetic resource for functional analysis on spike development under stress conditions in future. Recently, new techniques such as Single Cell-RNA Sequencing (scRNA-seq), and Spatial Transcriptomics (ST-seq), combined with other techniques like single-molecule fluorescence in situ hybridization (smFISH), and co-expression network analysis, were utilized to unveil the programmed developmental processes and identify distinct groups of genes in particular tissues in different wheat organs like roots, grains, and spikes[104,105]. Therefore, using these technologies to screen genes that specifically respond to stresses in different cell types will be a promising strategy to discover molecular mechanisms of spike development under different stress conditions, similar to the study in wheat leaf and root[106].
In general, both spatio-temporal expression patterns, and expression levels of genes are determined by the interaction between cis-acting elements and trans-acting factors[107]. The promoters of stress response genes generally contain typical stress response elements. ABA-independent cold- and drought-responsive genes often contain dehydration-responsive elements (DRE) in their promoters, which are recognized and bound by CBF (DREB1), and DREB2 transcription factors, members of the AP2/ERF family, which activate target gene expression[108,109]. Therefore, finding and testing novel stress-inducible promoters for optimization of expression levels of transgenes is one of the critical methodologies to improve plant developmental phenotypes and yields[106]. On the other hand, gene editing using clustered, regularly interspaced, short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9) technique, targeting specific elements in the gene promoter region will precisely identify the function of the gene under stress conditions and will be benefitial to crop breeding[110]. Moreover, taking advantage of CRISPR- Cas9 techniques to generate a promoter random deletion mutation library, followed by spike development defects screening under stress conditions will identify particular genes and cis-elements that respond to stresses and regulate wheat spike development[111,112].
Table 1. Genes associated with abiotic stress and spike development in wheat.
Stress type QTL/gene Function Temperature Low temperature VRN-1 Enabling the SAM-to-IM transition to initiate flowering while reducing freezing tolerance[26]. QFr.jic-5D Enhancing frost tolerance and reducing winterkill compared with spring-type alleles[27]. TaSAMT1 Increasing MeSA accumulation, reducing SA levels, and markedly improving freezing tolerance[34]. High temperature TraesCS1A02G305700 Conferring higher biomass and spike number under high nighttime temperature[42]. qYDH.3BL Increasing biomass, grain number, and grain weight following heat stress[43]. TaHST1 Increasing GNS, grain weight per spike, TGW, grain length and grain width under either thermo-stressed or control conditions[44]. TaHST2 Enhancing the conversion of soluble sugars into starch during heat-stressed grain filling[45]. TaHsfA1 Knockout mutants reducing seedling survival under high temperature and producing shrunken grains with lower TGW[47]. TaSG-D1 Improving heat tolerance though at a cost to grain yield[54]. Drought TduRuM_contig25432_1377 Maintaining stable association with fertile spike number across favorable and semiarid environments[52]. Rht13 Increasing peduncle length, spike length, grain number per spike, and grain weight under drought stress[56]. TaPYL1-1B, TaPYL4 Enhancing ABA sensitivity to improve drought tolerance and increase grain size and weight[67]. TaMPK3 Reducing drought tolerance and ABA sensitivity, thereby lowering survival rates and decreasing in spikelet number, tiller number, panicle length, and grain yield[68]. TaGW2 Improving drought tolerance and higher yields through increasing grain width and weight yields[75]. TaSAP5 Increasing drought tolerance by maintaining tiller and spike numbers under stress, leading to higher grain weight and grain number without yield penalties under normal conditions[76]. TaHDA8 Knockout lines showing higher yield per plant, mainly due to increased GNS under drought stress[78]. TabHLH27 Coordinating root growth and spike development to affect WUE and yield-related traits, including spikelet and grain number[81]. TaMYB7-A1 Enhancing photosynthesis, WUE, root development, and grain yield under drought condition[82]. Salt TaSPL6-D Knockout of TaSPL6-D enhances Na+ exclusion, preserves spike development, and improves yield under saline conditions[83]. TaHAL3-7A Improving salt stress tolerance while further boosts chlorophyll content, TGW, and overall yield[88]. TaCYP81D5 Promoting ROS scavenging to enhance salinity tolerance and producing larger seeds and higher yields under moderate salinity[151]. TaBZR1 Enhancing salt tolerance while reducing plant height and spike length but slightly increasing grain length[89]. TaCHP Enhancing tolerance to saline–alkaline stress strengthening germination, improving photosynthesis, increasing ROS scavenging, and increasing spike number, grain number per spike, and TGW[91]. TaDSU Enhancing salt tolerance and increasing spike number per plant and thousand-grain weight in overexpression lines[92]. -
The authors confirm contributions to the paper as follows: study conception and design: Cui Y, Liu X, and Zhang H; data collection: Ding Y, Li C, Mao R, He M, Zhang H; draft manuscript preparation: Ding Y, Li C, Cui Y, Liu X; language editing: García-Caparros P. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
-
This work was supported by grants from the Natural Science Foundation of China (Grant Nos U24A20391, 32501839, 32300304), the Natural Science Foundation of Hebei Province (Grant Nos C2025205068, C2024205025, C2024205009), and the Seed Science and Technology Innovation Team Project of Shijiazhuang (232490472A).
-
The authors declare that they have no conflict of interest.
-
# Authors contributed equally: Yi Ding, Caihua Li
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of Hainan Yazhou Bay Seed Laboratory. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Ding Y, Li C, Mao R, Garcia-Caparros P, He M, et al. 2026. Advances in the impact of abiotic stress on wheat spike development. Seed Biology 5: e009 doi: 10.48130/seedbio-0026-0005
Advances in the impact of abiotic stress on wheat spike development
- Received: 17 November 2025
- Revised: 05 January 2026
- Accepted: 21 January 2026
- Published online: 30 March 2026
Abstract: Wheat is a staple crop of critical economic and food security importance, with its yield playing a decisive role in ensuring global food supplies. However, the increasing frequency of extreme climatic events poses a substantial challenge to achieving stable and high yields. The wheat spike, as the principal yield-determining organ, has become a central focus of research, and remarkable advances have been achieved in unraveling its developmental regulatory networks with the aid of genomic and biotechnological approaches. Nevertheless, beyond intrinsic genetic potential, environmental stresses remain critical constraints on wheat spike development and subsequent productivity. However, research progress in this field lags far behind that in other areas like root, seedling, and even grain development. Here, current advances on the direct and indirect effects of abiotic stresses such as temperature, drought, and salt on spike development, and QTL's and genes involved in stress responses during spike development, are summarized. Finally, we discussed emerging perspectives that may deepen our understanding of spike–environment interactions, and guide strategies to enhance spike resilience and optimize wheat yield under stress conditions.
-
Key words:
- Wheat spike development /
- Abiotic stresses /
- Drought stress /
- Salt stress /
- Temperature stress













