-
Kratom (Mitragyna spp.) comprises a group of tropical evergreen trees indigenous to South and Southeast Asia, with notable distribution in Thailand, India, China, Myanmar, Malaysia, Laos, Cambodia, Indonesia, the Philippines, and Papua New Guinea[1]. Commonly referred to as kratom, these psychoactive and medicinally significant species include M. speciosa Korth., M. parvifolia (Roxb.) Korth., M. hirsuta Havil., and M. diversifolia (Wall. ex G.Don) Havil. (Plant nomenclature has been verified using World Flora Online and MPNS databases)[2]. The term 'kratom', along with various regional names such as biak-biak, kaim, ketum, kutum, ketumbia, nytum, and the commercial designation maeng da, encompasses both the plant itself and its diverse traditional preparations and extracts (Table 1). Historically, kratom has been utilized by Indigenous populations, particularly in southern Thailand and India, for multiple therapeutic purposes. The leaves have been employed for relief of different forms of pain, including chronic pain, myalgia, and headaches[3]. They have also been used as a stimulant to reduce fatigue and enhance work endurance, especially among manual labourers[4]. Furthermore, kratom has been reported to elevate mood and mitigate symptoms of depression and anxiety[5] and has been used by some individuals as an adjunct in managing opioid withdrawal symptoms[5,6]. Traditional modes of consumption include chewing fresh leaves, preparing decoctions or teas, and ingestion in capsule form[7]. Despite these medicinal applications, kratom is associated with risks of misuse, which may lead to dependence and adverse health outcomes. Recreational consumption, particularly among adolescents and young adults in Southeast Asia, often involves combining kratom with opioids, cough syrups, or energy drinks, thereby increasing the likelihood of serious adverse reactions[8]. Consequently, regulatory approaches differ across countries. Although the United Nations (UN) has not formally scheduled kratom under international drug control conventions, the UN Office on Drugs and Crime (UNODC) advises member states to monitor its use as a psychoactive substance. In India, kratom is not explicitly marketed, and its legal status remains unclear. In contrast, the Thai government has implemented regulatory measures, with ongoing policy discussions aimed at balancing therapeutic potential against risks of misuse[9]. At present, kratom products, including fresh and dried leaves, powders, and extracts, are readily available in Thailand without prescription[8,10]. Scientific investigation into kratom continues to expand, particularly with respect to identifying and characterizing its bioactive constituents and evaluating their pharmacological potential[10−12]. Notably, in 2003, Malaysia classified mitragynine, the principal alkaloid of kratom, under the First and Third Schedules of Psychotropic Substances. Under the Poisons Act 1952, possession or sale of kratom leaves or preparations containing mitragynine may result in penalties of up to RM 10,000 (USD 3,300), imprisonment for up to four years, or both[13].
Table 1. The common names known for Mitragyna spp.
Country Common name Thailand Ithang, Thom, Bai Kratom, Gratom, Kakaum, Katawn, Krathawn, Kratom, Kraton, Katawan, Kratawm, Tawn India Kaim, Guri, Nira Kadamba, Sanna Kadamba, Kayim, Kalam, Vitanah, Kathamamaram, Nichulam Malaysia Biak, Biak-Biak, Ketum, Kutum, Pokok Biak, Pokok Ketum, Sepay Myanmar Beinsa, Bein-Sa-Yawt Lao Nytum Vietnam Giam Indonesia Kadamba, Puri, Keton The Philippines Mambog, Lugub, Polaapupot References: [1,3,14,15]. Taxonomically, Mitragyna is a genus within the family Rubiaceae, the same botanical family as coffee. The genus comprises approximately ten recognized species, including those native to Thailand, such as M. speciosa, M. parvifolia, M. hirsuta, and M. diversifolia, which are collectively known as 'kratom' or 'kratum' [9,16]. Members of the genus are typically small to large trees that may be deciduous, semi-deciduous, or evergreen (Fig. 1). They generally exhibit smooth bark, opposite leaves, and entire stipules that are often keel-shaped. The inflorescences are compound and bear ellipsoidally arranged flower heads ranging in colour from greenish white to yellow. These may appear singly on leafy shoots, arise on sympodially branched axes, or occur in clustered formations resembling umbels. Many shoots also possess small, dormant serial buds beneath the flowering branches[17]. Some species produce sweetly scented flowers that attract pollinators such as bees, butterflies, and beetles. The inflorescence forms a dense, spherical cluster approximately 3–5 cm in diameter and contains both bisexual (perfect) and unisexual (imperfect) flowers. Male (Fig. 2a) and female flowers (Fig. 2b) may occur on the same branch (Fig. 2b-1). The leaves are typically dark green with prominent pinnate venation that may display green, white, or red pigmentation. Kratom varieties are commonly categorized based on mid-vein coloration: green-veined types range from yellowish to deep green, whereas red-veined forms exhibit hues from orange to pinkish red (Fig. 2c, d). Leaf morphology varies and may be lanceolate, ovate, or obovate, with bases described as cuneate, obtuse, or cordate[18]. Variation in vein coloration often occurs within the same tree and may reflect leaf maturity and developmental stage[19]. Mitragyna species primarily reproduce through wind-dispersed seeds, although vegetative propagation via stem or sucker cuttings is also possible in certain species. Their natural habitats include tropical forests, riparian zones, and swampy or marshland environments, particularly within the Malesian floristic region[1,16].

Figure 1.
Tree characteristics of Mitragyna spp. found in southern of Thailand. (a) Close-up view of the leaves showing large, ovate shape and prominent venation. (b) Full grown tree illustrating the overall growth habit.

Figure 2.
Kratom's floral and leaf structures exhibit key taxonomical characteristics. Notably, both (2a) male and (2b) female flowers are present on (2b-1) the same branch, indicating a monoecious reproductive system. The dark green leaves are characterized by prominent pinnate venation, where the veins branch off (2c, 2d) a central midrib. This venation can display a variety of colors, including green, white, or red. A primary classification method for kratom varieties relies on the mid-vein color: 'red vein' varieties display colors from (2c-1) orange, red, to pink, while (2c-2) 'green vein' varieties typically exhibit mid-vein hues ranging from yellow to green.
-
To provide a comprehensive evaluation of Mitragyna species in the context of traditional medicine and NTDs, a systematic literature search was conducted using electronic databases including PubMed, ScienceDirect, Google Scholar, and Scopus. The search strategy utilized Boolean combinations of keywords such as 'Mitragyna', 'Kratom', 'Ethnopharmacology', 'Indole Alkaloids', 'Neglected Tropical Diseases', 'Malaria', and 'Antiparasitic'.
Inclusion criteria encompassed peer-reviewed original research, ethnomedicinal surveys, and toxicological reports published in English. Additionally, traditional pharmacopoeial records from Thai and Indian systems were reviewed to document historical usage. Plant nomenclature for all cited species was verified using the World Flora Online and Medicinal Plant Names Services (MPNS) databases to ensure taxonomic accuracy. The review prioritizes studies that bridge the gap between traditional ethnomedical claims and modern pharmacological validation, with a specific focus on the genus-to-species translational potential for treating infectious diseases.
-
Historically, medicinal plants have been extensively used in the traditional medicine system, and numerous compounds or bioactives have been isolated from these sources, which have contributed to the development of modern medicines. Kratom occupies a distinctive and multifaceted position within Southeast Asian societies, where it intersects traditional medicine, social practices, and emerging recreational patterns. In contrast, within the Indian context, kratom has historically been regarded primarily for its medicinal value in local folklore, with no documented evidence of recreational use[20]. Its longstanding historical relevance, broad spectrum of applications, and evolving sociocultural role highlight the importance of continued scientific investigation to better define its therapeutic potential and associated risks. The traditional use of kratom in South and Southeast Asia extends back at least 150 years and reflects a complex pharmacological profile underlying its diverse effects. Fresh leaves have traditionally been chewed to enhance stamina and endurance during physically demanding labour, whereas decoctions or teas have been consumed for their analgesic and calming properties[9,16]. Kratom has also been incorporated into household remedies for conditions such as fever, diarrhoea, diabetes, and various pain disorders[9,21,22]. Beyond its therapeutic roles, kratom holds notable social significance. It is frequently consumed as a communal beverage, particularly among men, and has served as an alternative to alcohol in Muslim communities[23−25]. In recent decades, patterns of use have shifted. Kratom has gained prominence as a recreational substance, especially among youth in Thailand, where it is sometimes ingested in concentrated mixtures known as '4 × 100'[26]. Although less common, dried leaves may also be smoked for relaxation. In certain settings, kratom has been utilized as an accessible and affordable means to alleviate symptoms of opioid withdrawal[27]. The plant's versatility is reflected in the use of multiple parts, including leaves, bark, and roots, for medicinal preparations[16]. While its use among men is socially accepted in many Southeast Asian communities, recreational consumption by women is often culturally discouraged[1,21].
Kratom also has a well-documented history within traditional Thai medicine. Pharmacopoeial records dating back to the seventeenth century describe its application in treating abdominal pain, severe diarrhoea, and symptoms associated with opium withdrawal[23]. The Department of Thai Traditional and Alternative Medicine has catalogued 18 formulations from the Royal Pharmacopeia that incorporate kratom as a principal component[28]. Representative formulations are presented in Table 2. In these preparations, kratom leaves are typically combined with other medicinal herbs to address cramps, dysentery, and severe diarrhoea[29]. In southern Thailand, kratom has been integrated into broader herbal formulations extending beyond gastrointestinal indications. It has been traditionally employed to treat 'ไข้มหาสันนิบาต' (K̄hị̂ mh̄ā s̄ạnnibāt), a febrile condition historically associated with retained menstrual products in women in their third and fourth decades of life and characterized by neuropsychiatric symptoms such as delirium, agitation, and memory disturbances. Additionally, kratom has been used in managing anaemia, haematological irregularities, and menstrual disorders. Its reported antiparasitic activity has also supported its inclusion in deworming remedies[30]. Beyond its therapeutic applications, kratom carries cultural and symbolic importance in Thailand. It has traditionally been offered to guests as a gesture of hospitality and has featured in ancestral rites and religious ceremonies[15]. In southern Thailand and peninsular Malaysia, kratom consumption has long been embedded within daily life, reflecting deep-rooted cultural acceptance[31]. Sociological research in southern Thailand has suggested that kratom use—often in combination with other substances—may be associated with comparatively lower social stigma than other illicit drugs[32]. Preferences within these regions frequently favour red-veined varieties, valued for their pronounced bitterness and reportedly prolonged effects[33].
Table 2. Traditional Thai medicinal recipes using kratom.
Thai medicinal formula Method of preparation Treatments ยาประกระท่อม (Yā pra krath̀xm) Kratom leaves, combined with other herbs, are the primary ingredient in a decoction prepared with spirits. Reduce pain and stomach cramps Opium withdrawal Combine cannabis with other herbs and boil. Consume the resulting liquid as a tea. ยาหนุมานจองถนนปิดมหาสมุทร (Yā h̄numān cxng t̄hnn pid mh̄ās̄muthr) Dried leaves, ground and combined with other herbs, may be dissolved in drinking water. Relieve severe diarrhea Cramp treatments A decoction prepared with spirit and administered concurrently with opium or boil with opium and other ingredients and patchouli. Reduce menstrual and pregnancy cramps ยาประสะกาฬแดง (Yā pras̄a kāḷ dæng) Mix with spices and ground. Relieve severe diarrhea ยาเหลืองกระท่อม (Yā h̄elụ̄xng krath̀xm) Grind the leaves and other tropical herbs with lime juice and vinegar. Reduce cramps and measles In India, the fresh leaf sap of M. parvifolia has been traditionally employed by the Chenchu, Yerukula, Yanadi, and Sugalis tribes across Andhra Pradesh, Telangana, Karnataka, and Tamil Nadu for the management of jaundice. In addition, communities in Tumakuru and adjacent regions of Karnataka have utilized the leaves and bark as analgesics and in the treatment of myalgia and bilious disorders[34]. A decoction prepared from the bark of M. parvifolia has been reported as an antipyretic remedy and for alleviating rheumatic pain among the Sonaghati tribes of Uttar Pradesh and the Valaiyan tribe of Tamil Nadu, respectively. More broadly, the bark and roots of M. parvifolia have been used to address diverse conditions, including inflammatory disorders, toxic exposures, gynecological ailments, and edema. Ethnobotanical records further indicate its traditional application as a lactagogue and aphrodisiac[35]. Globally, regulatory approaches to kratom vary significantly. While Thailand moves toward a regulated medicinal market and Malaysia enforces strict criminalization under the Poisons Act, the United Nations maintains a 'monitoring' status without international scheduling. In India, the plant occupies a legal grey area with unclear status, whereas in Western settings (e.g., USA), regulatory attention focuses on public health surveillance via the US Food and Drug Administration's Adverse Event Reporting System (FAERS), driven by the prevalence of potent concentrated extracts rather than traditional leaf consumption. Over the past decade, FAERS documented 497 serious cases, including 356 fatalities. Detailed evaluation of these reports indicates that kratom was seldom the sole agent implicated; most cases involved concomitant use of other prescription medications[36]. This finding is significant considering evidence that two major kratom alkaloids, mitragynine and corynantheidine, act as moderate inhibitors of cytochrome P450 2D6 (CYP2D6). Co-administration of kratom products may therefore result in competitive inhibition of CYP2D6-mediated drug metabolism, potentially elevating plasma concentrations of co-administered drugs and increasing the risk of adverse outcomes[37]. The likelihood of such interactions may be greater in Western settings, where concentrated kratom extracts, often containing higher mitragynine levels than traditional preparations, are widely available. Nonetheless, acute toxicity studies in vivo using standardized methanolic extracts of M. speciosa Korth. administered to four-week-old Sprague–Dawley rats (100, 500, and 1,000 mg/kg) demonstrated no mortality over a 14-d observation period, underscoring the importance of formulation type and co-exposure context in safety assessments[38]. Since CYP2D6 metabolizes many common medications, co-administration with kratom causes a competitive inhibition. This blocks the body's ability to break down other drugs, potentially leading to toxic accumulation of those co-administered substances in the plasma. The danger is greatest in polypharmacy settings. This explains why, despite Kratom's traditional safety record, modern usage serves as a catalyst for adverse events when combined with prescription medicines.
-
The isolation and structural characterization of mitragynine, a principal indole alkaloid of M. speciosa, has stimulated substantial scientific interest in elucidating its pharmacological mechanisms. Early investigations, including the first isolation of mitragynine in 1921 at the University of Edinburgh, primarily examined its stimulant properties. Clinical observations from that period noted that kratom users exhibited minimal respiratory depression and lacked the characteristic symptoms of opium intoxication; instead, individuals were described as appearing mentally and physically 'quite normal'[39]. Phytochemical analyses indicate that immature leaves of M. speciosa contain mitragynine as the predominant alkaloid (66.2%), accompanied by lower concentrations of speciogynine (6.6%), speciociliatine (0.8%), paynantheine (8.6%), and the structurally related 7α-hydroxy-7H-mitragynine (7-HMG) (2.0%) (Fig. 3). The structure of 7-HMG was elucidated through spectroscopic techniques and confirmed via chemical transformation from mitragynine[40]. Receptor-binding studies demonstrate that mitragynine exhibits significant affinity for
$\mu $ $\mu $ 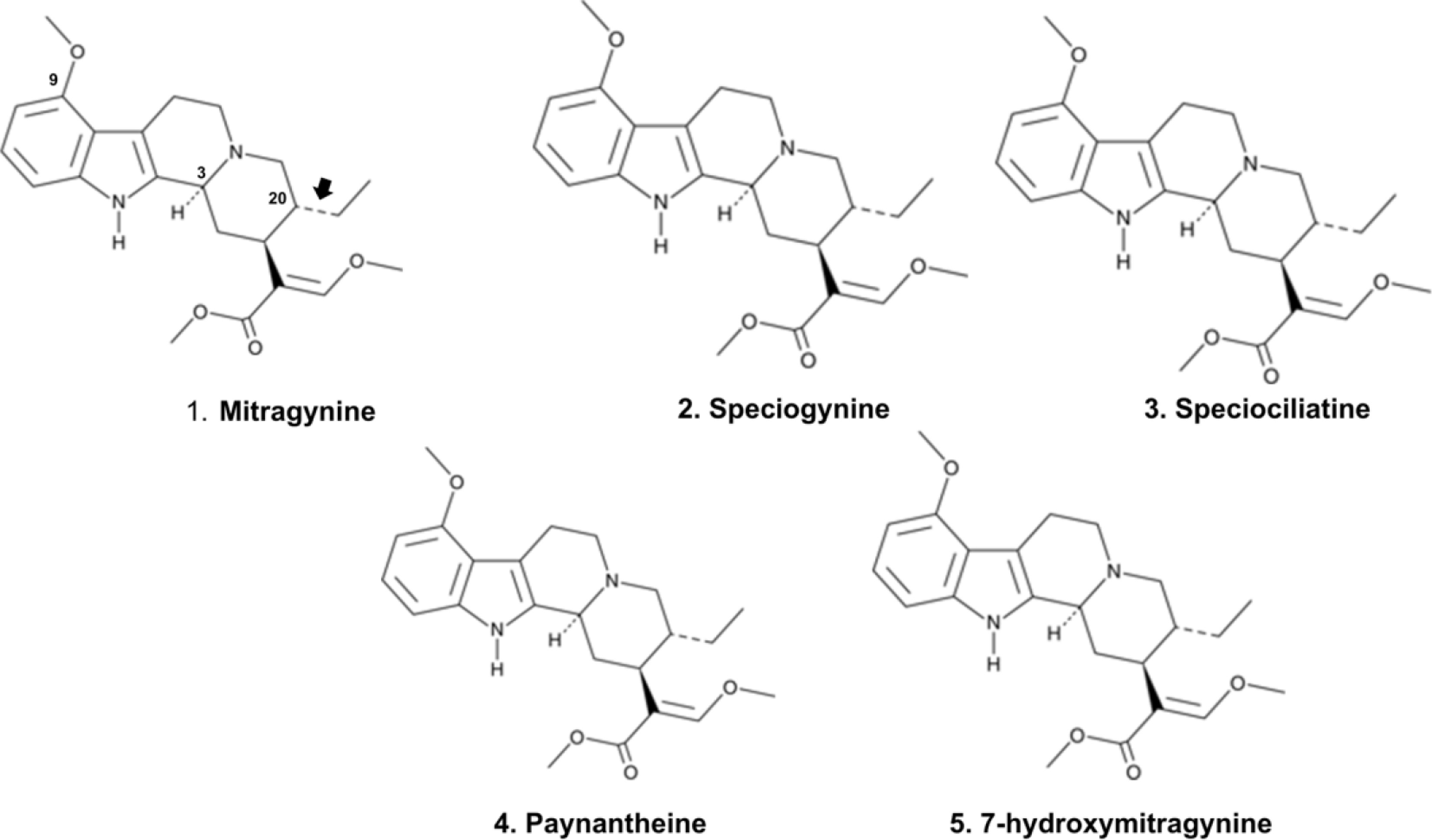
Figure 3.
Chemical structures of the major indole alkaloids isolated from Mitragyna spp., including Mitragynine, Speciogynine, Speciociliatine, Paynantheine, and 7-Hydroxymitragynine.
Comparative studies utilizing tail-flick assays in mice have demonstrated that 7-HMG exhibits potent antinociceptive activity. It shows approximately 13-fold higher potency than morphine following subcutaneous administration, as evidenced by dose-response curves where 7-hydroxymitragynine (2.5–10 mg/kg) outperformed morphine in both tail-flick and hot-plate tests. In contrast, mitragynine displays a lower potency profile, comparable to codeine, though specific efficacy varies significantly depending on the route of administration (oral vs intraperitoneal) and the opioid receptor subtype engaged[22,43]. In silico study reported that certain oxindole alkaloids such as Mitrafoline, Isospecionoxeine, Corynoxine B, Mitraphylline, from M. speciosa exhibit strong predicted interactions with CYP3A4, along with a moderate to high likelihood of CYP2D6, and CYP2C19 inhibition, suggesting that their higher lipophilicity and favorable oral absorption may contribute to increased systemic exposure and a potential risk of drug–drug interactions[44].
In addition to
$\mu $ Beyond opioid receptor modulation, mitragynine has been reported to suppress locomotor activity through effects on presynaptic dopaminergic pathways, potentially explaining its traditional use in alleviating muscle cramps and chronic pain. Moreover, experimental studies indicate that mitragynine reduces circulating corticosterone levels, suggesting modulation of the hypothalamic–pituitary–adrenal (HPA) axis. Because HPA axis dysregulation is associated with depressive states, this neuroendocrine influence may partially account for the antidepressant-like effects observed in preclinical models[46].
The anti-inflammatory activity of kratom leaves appears to be mediated through multiple complementary mechanisms, including suppression of pro-inflammatory mediator release and attenuation of vascular permeability. In addition, evidence suggests modulation of immune responses and facilitation of tissue repair processes[47]. Central to these effects are the cyclooxygenase (COX) and lipoxygenase (LOX) pathways, particularly the inducible COX-2 isoform[48]. COX-2 catalyses the synthesis of prostaglandin E2 (PGE2), a key mediator of inflammation. Experimental studies indicate that mitragynine downregulates COX-2 mRNA and protein expression, resulting in reduced PGE2 production. At lower concentrations, this inhibition appears relatively selective for COX-2, with minimal impact on COX-1, an enzyme essential for physiological homeostasis. However, at higher doses, reduced selectivity may occur, potentially affecting COX-1 activity and warranting cautious interpretation[49]. This dual mechanism suggests Kratom could serve as a bridge therapy for pain management that addresses both nociception (opioid pathway) and inflammation (COX-2 pathway) without the full respiratory depression profile associated with classical opioids like morphine. Beyond mitragynine and structurally related alkaloids, additional bioactive constituents of M. speciosa may contribute to the mitigation of oxidative stress. Elevated intracellular reactive oxygen species (ROS), including hydrogen peroxide (H2O2), can trigger neuronal injury and apoptosis. Activation of antioxidant defense genes such as heme oxygenase-1 (HO-1) and NAD(P)H-quinone oxidoreductase-1 (NQO1) is primarily regulated through nuclear translocation of the transcription factor Nrf2[50]. Kratom extracts have demonstrated antioxidant capacity across multiple in vitro platforms, including DPPH radical scavenging assays, reducing power assessments, oxygen radical absorbance capacity (ORAC), ferric reducing antioxidant power (FRAP), and cupric reducing antioxidant capacity (CUPRAC) assays[50−52]. Collectively, these findings support a meaningful antioxidant component to kratom's pharmacological profile. Mitragynine and 7-HMG also exert measurable effects on gastrointestinal physiology. In anesthetized rat models, intracerebroventricular administration of mitragynine into the fourth ventricle produced dose-dependent suppression of gastric acid secretion induced by 2-deoxy-D-glucose. Similarly, subcutaneous administration of 7-HMG in mice significantly reduced gastrointestinal transit, indicating modulatory effects on gut motility[53,54].
-
Neglected tropical diseases (NTDs), including malaria, tuberculosis, and Chagas disease, impose a disproportionate burden on economically disadvantaged populations, hindering socioeconomic development and contributing to substantial morbidity and mortality. Despite their global impact, these conditions remain chronically underfunded and insufficiently researched, leading to limited therapeutic options and the increasing prevalence of drug-resistant strains. Plant-derived natural products, particularly those obtained from underexplored or underutilized species, represent a promising strategy for addressing these gaps. Their structural diversity, relative affordability, and potential to provide novel pharmacophores capable of overcoming resistance mechanisms position them as valuable candidates for new drug discovery. Nevertheless, translating such natural compounds into clinically viable interventions requires a comprehensive approach, encompassing rigorous pharmacological validation, standardized phytochemical characterization, sustainable sourcing practices, and collaborative engagement with affected communities to promote equitable access.
Kratom, traditionally recognized for its biphasic stimulant and sedative effects, has more recently attracted scientific interest for its broader pharmacological properties. Emerging investigations have begun to assess its therapeutic potential beyond conventional indications, including exploratory studies evaluating its relevance in the context of NTD management.
To critically evaluate the therapeutic potential of Mitragyna species, it is essential to distinguish between three distinct modes of action identified in the literature:
(1) Direct anti-parasitic activity: Interventions where bioactives directly inhibit or kill the pathogen within the host (e.g., macrofilaricidal effects on adult worms or antiplasmodial activity).
(2) Vector control (Indirect action): Strategies targeting the disease vectors, such as larvicidal or repellent effects against Aedes and Culex mosquitoes, to interrupt transmission.
(3) Symptomatic management: The use of kratom's analgesic and anti-inflammatory properties to manage clinical manifestations (e.g., fever in Dengue) without necessarily clearing the causative agent.
Malaria
-
In traditional Malaysian and Indonesian medical practices, M. speciosa has been employed for the treatment of ailments such as malaria, cough, hypertension, and pain[55,56]. Other species within the genus, including M. ciliata, have similarly been used for comparable therapeutic indications[57−60]. The growing prevalence of drug-resistant Plasmodium strains has intensified the challenge of effective malaria management. Malaria, caused by protozoan parasites of the genus Plasmodium and transmitted by female Anopheles mosquitoes, remains a major public health concern, particularly in endemic regions such as southern Thailand and India, where kratom is naturally distributed.
Although kratom has been proposed to possess antimalarial properties, the available scientific evidence remains limited and inconclusive. In one experimental study using P. yoelii-infected mice, kratom extract did not demonstrate direct parasitic clearance but was associated with a significant extension of survival time, suggesting possible host-modulatory effects that merit further investigation[61]. A separate study explored an alternative antimalarial strategy, demonstrating that kratom constituents, including mitragynine and 7-hydroxymitragynine, can function as reducing and stabilizing agents in the biosynthesis of silver nanoparticles (AgNPs)[62]. These kratom-mediated AgNPs exhibited potent in vitro antiplasmodial activity, with IC50 values below 5 µg/mL, indicating substantial parasite growth inhibition. This moves Kratom research beyond simple herbal extracts into nanomedicine[63]. It implies that Kratom's value might not just be as a direct drug, but as a bio-functional scaffold for stabilizing novel drug delivery systems against resistant malaria strains.
Further investigation is required to clarify the mechanistic basis of these effects and to evaluate the translational potential of kratom-derived compounds and nanostructures as antimalarial candidates. Additional studies on related species provide contextual support: triterpenes have shown activity against both chloroquine-sensitive and chloroquine-resistant P. falciparum strains, and extracts from leaves, roots, and stem bark of M. inermis have demonstrated antiplasmodial activity with IC50 values ranging from 15 to 18 µg/mL. In vitro assays also revealed reductions in parasitemia in infected erythrocytes treated with M. inermis extracts compared with untreated controls[64].
Trypanosomiasis
-
Trypanosomiasis comprises a spectrum of parasitic diseases caused by pathogenic species of the genus Trypanosoma, affecting both humans and livestock[65]. As a neglected tropical disease, it exerts profound health and economic consequences in endemic regions. Human African Trypanosomiasis (HAT), commonly referred to as sleeping sickness, predominantly affects populations in sub-Saharan Africa, whereas Animal African Trypanosomiasis (AAT), also known as Nagana, impacts livestock and agricultural productivity[66]. Transmission occurs primarily through bites of tsetse flies (Glossina spp.), which act as vectors for multiple Trypanosoma species[67]. In contrast to American trypanosomiasis (Chagas disease), caused by T. cruzi and transmitted by reduviid insects, African trypanosomiasis continues to pose a substantial public health challenge across sub-Saharan Africa.
Members of the genus Mitragyna have demonstrated antiprotozoal properties[68]. M. ciliata, traditionally employed by healers in southeastern Nigeria for the treatment of trypanosomiasis, has shown notable experimental activity[69]. Investigations reveal that root extracts of M. ciliata exhibit significant trypanocidal effects; administration at 50 mg/kg resulted in approximately 76% suppressive activity and 54.24% therapeutic efficacy against the parasite in vivo[70]. Complementary biochemical analyses further support the safety and potential efficacy of these extracts, including evaluations of hepatic enzyme profiles, modulation of oxidative stress parameters associated with parasite clearance, and effects on calcium homeostasis in infected animal models. Collectively, these findings indicate that M. ciliata may represent a promising source of bioactive compounds for anti-trypanosomal drug development. The antiparasitic efficacy demonstrated by related Mitragyna species, including M. ciliata against trypanosomiasis and M. parvifolia against lymphatic filariasis, provides a strong foundation for investigating M. speciosa's pharmacological potential. These species share similar indole alkaloid profiles, suggesting that M. speciosa may possess analogous but unexplored antiparasitic properties. This phylogenetic relationship supports adopting a systematic screening approach wherein bioactive compounds identified in one Mitragyna species guide targeted investigation of M. speciosa, potentially accelerating the discovery of novel therapeutic agents.
Leishmaniasis
-
Leishmaniasis represents a complex group of neglected tropical diseases caused by protozoan parasites of the genus Leishmania and remains a major global health concern. The disease presents in three principal clinical forms: cutaneous, mucocutaneous, and visceral leishmaniasis. Cutaneous leishmaniasis generally manifests as localized skin ulcers that may resolve spontaneously, whereas mucocutaneous disease can result in destructive lesions affecting the nasal and oral mucosa. Visceral leishmaniasis, the most severe manifestation, is associated with systemic infection and, if untreated, may result in mortality rates as high as 95% in certain settings[71].
Current therapeutic regimens for leishmaniasis are constrained by substantial limitations, including toxicity, high treatment costs, prolonged administration schedules, and the growing incidence of drug resistance. The absence of broadly effective vaccines or definitive curative therapies further highlights the urgent need for alternative pharmacological strategies. Indole alkaloids and triterpenoids, classes of compounds commonly found in Mitragyna spp. and other members of the Rubiaceae family, have demonstrated antiparasitic and antitrypanosomal activities in experimental models[68,72]. Mitragyna inermis, a species native to Africa, has attracted particular interest for its potential antileishmanial activity. Extracts derived from its stem bark and leaves have shown inhibitory effects against Leishmania species, including L. donovani[73]. Moreover, synthetic derivatives of mitragynine, especially those incorporating the indolo[2,3-a]quinolizidine scaffold, are considered promising leads in drug discovery. These derivatives exhibit a range of biological activities, including anti-allergic, antibacterial, antiviral, and potentially antileishmanial effects[74,75].
Dengue fever
-
Dengue fever (DF) is one of the most prevalent mosquito-borne viral illnesses in tropical regions and is caused by the dengue virus (DENV). The disease is transmitted primarily by Aedes aegypti and Ae. albopictus mosquitoes and represent a major public health concern in endemic areas. Clinical manifestations may include gastrointestinal symptoms such as abdominal pain, nausea, vomiting, jaundice, hepatomegaly, and, in severe cases, hepatic dysfunction or failure.
While M. speciosa is traditionally utilized to manage the severe muscle pain associated with dengue via its opioid and COX-2 inhibiting pathways, modern scientific evaluation has focused primarily on vector control. Investigations into the larvicidal potential of kratom leaf extracts have yielded limited efficacy. One study reported that only a relatively high concentration (20 mg/mL) induced significant larval mortality, whereas lower concentrations (10.5 and 2.5 mg/mL) did not produce statistically meaningful effects[76]. Additionally, evaluation of the repellent activity of M. speciosa extract against Ae. albopictus indicated a transient deterrent effect during the initial three minutes of exposure; however, this protection diminished rapidly and was not sustained beyond five minutes, suggesting limited practical utility as a mosquito repellent[77].
Schistosomiasis
-
Urinary, or urogenital, schistosomiasis is a parasitic infection caused by blood flukes belonging to the genus Schistosoma. These digenetic trematodes require freshwater snails of the genus Bulinus as intermediate hosts for completion of their life cycle. Species of Bulinus exhibit relatively limited morphological diversity. Human transmission occurs through contact with freshwater contaminated by free-swimming larval forms (cercariae), which penetrate intact skin. Schistosomiasis remains a major public health issue, particularly in rural populations living near slow-moving or stagnant water bodies in tropical and subtropical regions[78].
Clinical manifestations vary depending on the infecting Schistosoma species and the organs involved, and may include diarrhoea, inflammatory responses, and haemorrhagic complications. In severe or untreated cases, the disease can progress to life-threatening outcomes. Within traditional medicine, M. stipulosa has been employed as a remedy for bilharziasis, a term historically used to describe schistosomal infections caused by parasitic flatworms[79]. M. inermis is also utilized in certain regions, although its application depends on local ethnomedical practices and availability. The bark of these species is traditionally prescribed for a range of conditions, including pregnancy- and childbirth-related indications, abdominal pain, dysentery, and schistosomiasis[59].
Lymphatic filariasis
-
Lymphatic filariasis (LF) is a neglected tropical disease affecting more than 882 million people across 44 countries and is primarily transmitted through mosquito vectors. Although both malaria and LF are mosquito-borne diseases, they differ in their principal vectors: malaria is predominantly transmitted by Anopheles mosquitoes, whereas LF is spread by several genera, including Culex, Aedes, and Mansonia[80]. Among these, Culex quinquefasciatus is considered a major vector due to its broad geographic distribution and significant public health impact. Investigations into the potential role of M. stipulosa and M. parvifolia leaves in LF management have reported encouraging preliminary findings. Phytochemical analysis identified key bioactive constituents within the extracts, which demonstrated notable biological activities[81]. The extracts exhibited antibacterial effects against pathogens such as Staphylococcus epidermidis, Bacillus cereus, and Salmonella typhi. In vitro assays further revealed significant macrofilaricidal activity, characterized by inhibition of worm motility and suppression of metabolic function, suggesting potential efficacy against adult filarial worms. Larvicidal evaluations showed measurable activity against C. quinquefasciatus larvae, with higher concentrations producing greater mortality. This dual activity suggests M. parvifolia could theoretically contribute to both transmission control and host treatment, although data for M. speciosa remain absent. Additionally, the extracts displayed concentration-dependent antioxidant properties, which may confer ancillary health benefits. However, to date, there is no direct experimental evidence specifically evaluating kratom for therapeutic application in lymphatic filariasis.
Table 3 provides a comprehensive pharmacological overview mapping specific Mitragyna species, including M. speciosa, M. ciliata, and M. parvifolia, to their respective bioactive constituents, molecular targets, and therapeutic outcomes for NTDs.
Table 3. Pharmacological mapping of Mitragyna species against neglected tropical diseases (NTDs).
NTD/condition Targeted Mitragyna species Bioactive constituent/preparation Molecular target or mechanism Therapeutic significance and outcome Malaria M. speciosa (kratom); M. inermis Silver nanoparticles (AgNPs); Crude extracts Nanoparticle stability: AgNPs inhibit parasite growth. Host Modulation: Mechanism for survival extension remains unknown (no clearance). High potential (nano): AgNPs showed potent activity (IC50 < 5 μg/mL). Low potential (extract): Failed to clear parasites. Trypanosomiasis (sleeping sickness) M. ciliata Root extracts (indole alkaloids) Enzymatic/Ca2+ modulation: Affects hepatic enzymes, oxidative stress, and calcium homeostasis in hosts. High efficacy (in vivo): Demonstrated ~76% suppressive activity and 54.24% therapeutic efficacy in models. Leishmaniasis M. inermis; M. speciosa (derivatives) Stem bark and leaf extracts; Synthetic indolo[2,3-a]quinolizidine Antiparasitic action: Direct inhibition of Leishmania donovani. Structural Scaffolds: Synthetic derivatives target parasite viability. Promising lead: Significant inhibitory effects; synthetic scaffolds identified for drug discovery. Dengue fever (vector control) M. speciosa (kratom) Leaf extracts Larvicidal: Toxicity to Aedes aegypti larvae. Repellent: Sensory deterrence against Aedes albopictus. Low viability: Larvicidal activity required impractical concentrations (20 mg/mL); repellent effect lasted < 5 min. Lymphatic filariasis M. parvifolia; M. stipulosa Leaf extracts Macrofilaricidal: Inhibits worm motility and suppresses metabolic function of adult worms. High potential (in vitro): Significant activity against adult worms; no current data for M. speciosa. Schistosomiasis M. stipulosa; M. inermis Bark extracts Ethnomedicinal: Traditional remedy for 'bilharziasis' (parasitic flatworms). Ethnobotanical: Historically used; specific molecular targets require further validation. NTD symptom management (fever/pain) M. speciosa (kratom) Mitragynine; 7-Hydroxymitragynine COX-2 pathway: Downregulates COX-2 mRNA; inhibits PGE2. Opioid receptors: Agonist activity at $\mu $-opioid receptors. Supportive therapy: Validated mechanism for managing pain/inflammation in infections (e.g., dengue). -
Despite compelling ethnopharmacological evidence supporting Mitragyna species for NTD treatment, substantial gaps exist between current data and the clinical potential of kratom. First, a critical phylogenetic and geographic disconnect undermines direct efficacy translation. Most antiparasitic evidence originates from African species (M. ciliata for trypanosomiasis; M. inermis for leishmaniasis) rather than Southeast Asian M. speciosa. Without comprehensive chemotaxonomic comparison, assuming shared antiparasitic alkaloid profiles across geographically distinct species is premature. Second, available M. speciosa data suggest symptomatic relief rather than parasitocidal activity. In P. yoelii malaria models, crude extracts extended survival without eliminating parasitemia, indicating anti-inflammatory or analgesic mechanisms rather than direct antiparasitic effects. Notably, antiplasmodial efficacy appears dependent on technological enhancement, specifically, silver nanoparticle (AgNP) synthesis—rather than inherent phytochemical potency. Third, vector control applications show limited practicality. Larvicidal activity against Ae. aegypti required impractically high concentrations (20 mg/mL), while repellency lasted under five minutes, far below the performance of established botanical insecticides. Moreover, despite M. parvifolia's macrofilaricidal activity, no published studies have evaluated M. speciosa against lymphatic filariasis, representing a significant knowledge gap. These limitations underscore the need to move beyond phylogenetic inference toward direct pharmacological evaluation of M. speciosa compounds. Future research must establish whether this species offers disease-modifying potential or functions primarily as adjunctive symptomatic therapy.
-
Kratom holds a distinctive ethnobotanical role across South and Southeast Asia, particularly in southern Thailand and parts of India, where it has long been incorporated into traditional healthcare practices. Historically valued for its biphasic stimulant and sedative effects, kratom has been used to manage a variety of conditions, including pain, fever, diarrhea, and metabolic disorders such as diabetes. Its enduring presence in traditional medicine reflects the complex integration of therapeutic use, social practice, and cultural meaning associated with the plant. Despite its potential medicinal benefits, concerns have emerged regarding misuse, dependence, and public health implications. In Thailand, regulatory measures have been introduced, with continuing policy discussions focused on developing a balanced framework that reconciles therapeutic potential with risks of abuse. In contrast, reports of kratom misuse in India remain limited. Contemporary scientific investigations are increasingly exploring kratom's pharmacological properties and its possible applications, including exploratory research into its relevance for NTDs. Additionally, developing NTD treatments from locally available flora like Mitragyna spp. aligns with sustainable healthcare models. It offers a potentially cost-effective alternative to expensive synthetic drugs, reducing dependency on imported pharmaceuticals for diseases like Dengue and Leishmaniasis. This review aims to encourage further rigorous research and to support the rational development of therapeutic strategies derived from kratom's bioactive constituents. A thorough understanding of its pharmacodynamics, toxicological profile, and clinical applicability, particularly in the NTD context, is essential to optimize therapeutic potential while ensuring safety and responsible utilization.
-
The authors confirm their contributions to the paper as follows: study conception and design: Sommano SR; literature review: Ruksiriwanich W; writing: Sommano SR, Ruksiriwanich W, Jantrawut P, Devegowda D, Jahagirdar S, Bhat SS, Prasad SK; editing: Sommano SR, Jantrawut P, Devegowda D, Prasad SK; formatting: Jahagirdar S, Bhat SS; manuscript review and proofreading: Devegowda D, Prasad SK. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
-
Sommano SR, Ruksiriwanich W, Jantrawut P thank Chiang Mai University, Thailand, for the support towards the conduct and publication of this work. Prasad SK, Jahagirdar S, Bhat SS, Devegowda D acknowledge the infrastructure and support provided by JSS Academy of Higher Education and Research, Mysuru. This work was partially funded by Chiang Mai University.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Sommano SR, Ruksiriwanich W, Jantrawut P, Devegowda D, Jahagirdar S, et al. 2026. Kratom (Mitragyna spp.) in Thai and Indian traditional medicine: ethnopharmacology, phytochemistry, and therapeutic potential for neglected tropical diseases. Medicinal Plant Biology 5: e010 doi: 10.48130/mpb-0026-0006
Kratom (Mitragyna spp.) in Thai and Indian traditional medicine: ethnopharmacology, phytochemistry, and therapeutic potential for neglected tropical diseases
- Received: 24 November 2025
- Revised: 11 February 2026
- Accepted: 18 March 2026
- Published online: 23 April 2026
Abstract: Kratom, a collective designation for species within the genus Mitragyna, comprises tropical evergreen trees indigenous to South and Southeast Asia. The plant holds considerable ethnobotanical relevance, particularly in the southern regions of Thailand and India, where it has been traditionally employed for its biphasic stimulant and sedative effects and in the management of conditions such as pain, fever, and diarrhoea. This review presents a comprehensive evaluation of kratom within the framework of Thai and Indian traditional medicine, with emphasis on its ethnopharmacological applications and its proposed relevance to neglected tropical diseases (NTDs). It examines the historical and cultural dimensions of kratom use across South and Southeast Asia, underscoring its integration into local medicinal systems and sociocultural practices. The phytochemical profile of kratom is analysed, focusing on its principal indole alkaloids, including mitragynine, speciogynine, speciociliatine, and 7-hydroxy-7H-mitragynine. In addition, the review outlines the prevailing regulatory landscape in both countries, highlighting ongoing policy discussions in Thailand aimed at balancing therapeutic potential against risks of misuse. Finally, it underscores the necessity for rigorous scientific investigation to clarify pharmacological mechanisms, establish safety parameters, and support the development of evidence-based therapeutic strategies derived from kratom's bioactive constituents, particularly in relation to NTDs. This review innovatively repositions M. speciosa beyond its conventional role in pain and addiction management by applying a 'genus-to-species' translational framework to predict its efficacy against NTDs, specifically highlighting novel applications in the green synthesis of antimalarial silver nanoparticles.
-
Key words:
- Ethnopharmacology /
- Indole Alkaloids /
- Psychoactives /
- Pain relieving /
- Tropical medicinal plants /
- NTDs












