-
A 75-year-old man with a known diagnosis of hepatocellular carcinoma (HCC) was admitted after pulmonary space-occupying lesions were identified one week following transarterial chemoembolization (TACE). Physical examination on admission revealed no significant abnormalities. The patient was conscious and in fair general condition. Vital signs were stable, and there was no clinical evidence of jaundice, superficial lymphadenopathy, or peripheral edema. Abdominal examination was unremarkable, with no palpable hepatosplenomegaly. Neurological examination showed no focal deficits.
Contrast-enhanced magnetic resonance imaging (MRI) of the liver demonstrated residual enhancing nodules in segments VII and VIII, consistent with residual tumor activity following TACE. Contrast-enhanced computed tomography (CT) of the chest revealed a nodule in the left lower lobe accompanied by widespread lymphadenopathy. Brain MRI identified multiple enhancing nodules distributed throughout the brain, suggestive of metastatic disease. Whole-body positron emission tomography–computed tomography (PET-CT) demonstrated intense hypermetabolic activity in the hepatic lesions, left lung nodule, lymph nodes, bones, and brain, confirming multifocal metastatic dissemination. Representative imaging findings are shown in Fig. 1. Whole-body PET-CT findings demonstrating multifocal metastatic disease are shown in Fig. 2.
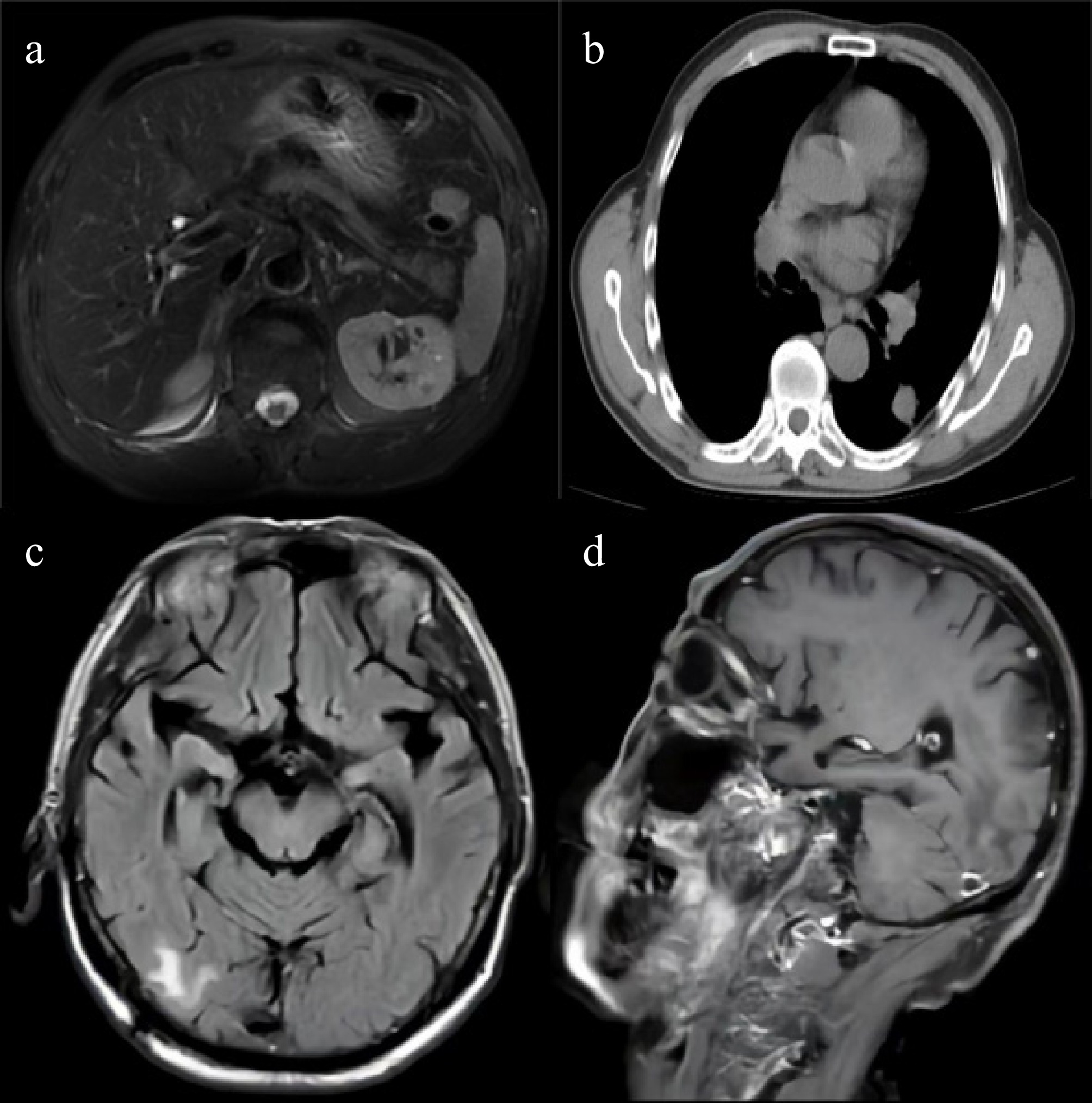
Figure 1.
Cross-sectional contrast-enhanced imaging of the patient. (a) Contrast-enhanced MR of the liver: images show hypervascular nodules near the inferior vena cava in segment VIII and in segment VII, primarily considered early hepatocellular carcinoma (eHCC). (b) Contrast-enhanced CT of the chest: images reveal a nodule in the left lower lobe, multiple lymph nodes in the left neck, supraclavicular area, mediastinum, and left pulmonary hilum, and osteolytic destruction of the left scapula, primarily suggesting pulmonary malignancy with metastases. (c) Contrast-enhanced MR of the brain: Images demonstrate multiple enhancing nodules in the brain, with metastasis as the primary consideration. (d) Contrast-enhanced MR of the brain: images show multiple enhancing nodules in another region of the brain.
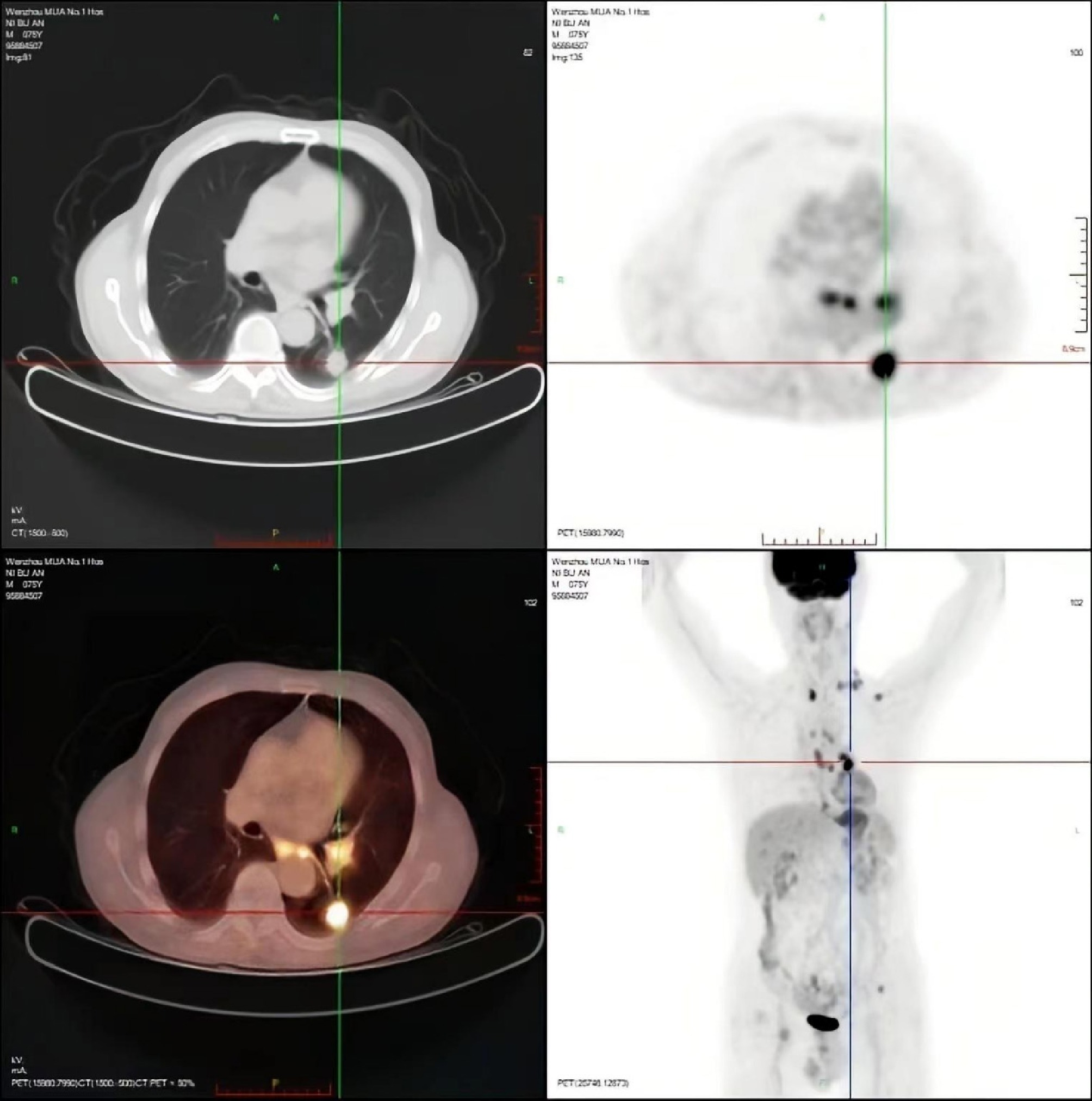
Figure 2.
PET-CT evaluation. (a) PET maximum intensity projection (MIP) whole-body image: shows multiple hypermetabolic foci in the left lower lung, multiple lymph node stations, brain (left frontal and right temporal lobes), left scapula, and left ilium, suggesting malignant metastases. (b) Fused PET/CT image of the chest: corresponds to Fig. 1b, confirming hypermetabolism in the left lower lung nodule and mediastinal lymph nodes. (c) Fused PET/CT image of the abdomen: corresponds to Fig. 1a, shows residual increased metabolism in the hepatic nodules (segments VII and VIII), suggesting residual tumor activity post-treatment. (d) Fused PET/CT image of the brain: corresponds to Fig. 1c, d, reveals hypermetabolic small nodules in the brain (left frontal and right temporal lobes).
A percutaneous biopsy of the left lung lesion was performed. Histopathological examination revealed a poorly differentiated malignant tumor. Tumor cells were arranged in sheets and irregular nests, exhibiting marked nuclear atypia, hyperchromatic nuclei, a high nuclear-to-cytoplasmic ratio, and focally eosinophilic cytoplasm. Areas of tumor necrosis were present. Given the undifferentiated morphology and the absence of features typical of well-differentiated HCC, further immunohistochemical evaluation was required to clarify the tumor origin. Representative hematoxylin and eosin–stained sections are shown in Fig. 3.
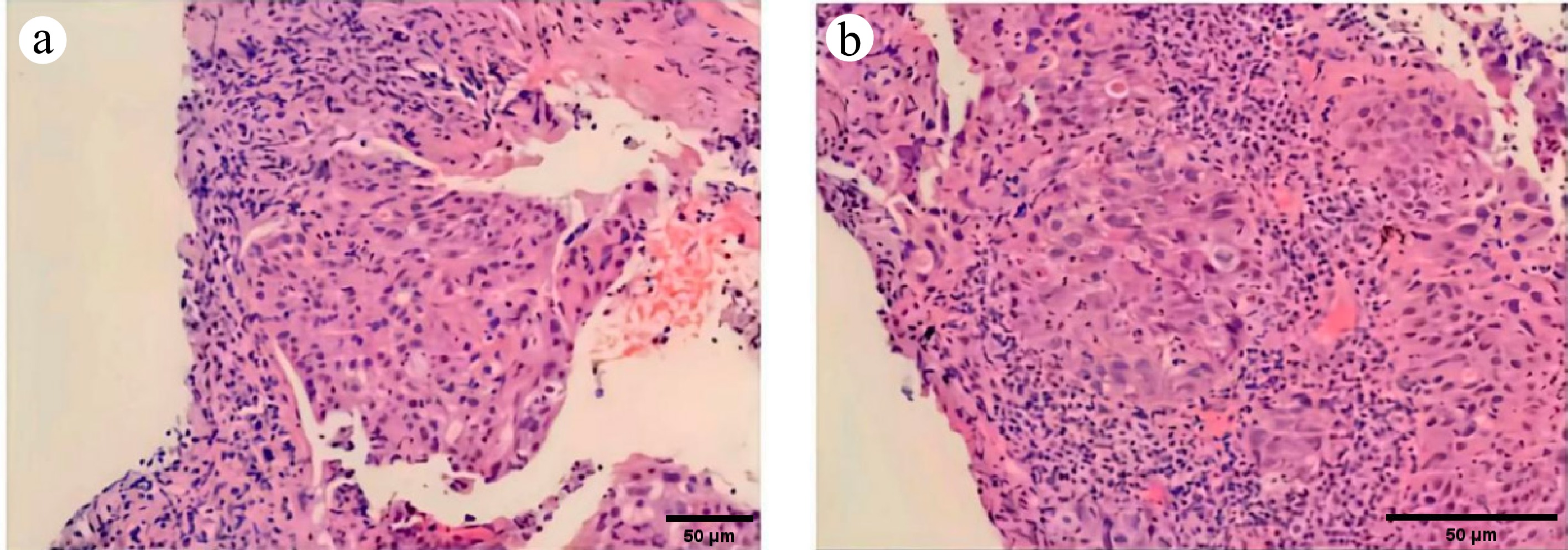
Figure 3.
Histopathological images of the lung metastasis biopsy: (a) 100×, (b) 200×. H & E-stained micrograph: shows the tumor tissue and the morphology of tumor cells.
Histopathological examination revealed a poorly differentiated malignant tumor with highly atypical morphology. Given the poorly differentiated features and aggressive clinical behavior, the initial routine immunohistochemical panel was insufficient to reach a definitive diagnosis. Considering the possibility of a SMARCA4-deficient tumor, we performed additional immunohistochemical staining for SMARCA4/BRG1. Immunohistochemical analysis demonstrated positivity for hepatocytic markers, including Hepatocyte and focal glypican-3 (GPC-3), confirming hepatic origin. The tumor cells also expressed biliary markers, with positivity for cytokeratin 19 (CK19) and partial positivity for cytokeratin 7 (CK7). Markers associated with primary lung malignancies, including thyroid transcription factor-1 (TTF-1), Napsin A, P40, and cytokeratin 5/6 (CK5/6), were negative. The Ki-67 proliferation index was approximately 60%, indicating high proliferative activity. Notably, immunohistochemistry demonstrated complete loss of nuclear SMARCA4/BRG1 expression in tumor cells, with retained expression in non-neoplastic cells serving as internal controls. Programmed death-ligand 1 (PD-L1) expression was low, with a tumor proportion score (TPS) of 10%. Immunohistochemical findings, including complete loss of SMARCA4/BRG1 expression, are illustrated in Fig. 4. A summary of the immunohistochemical profile is provided in Table 1.
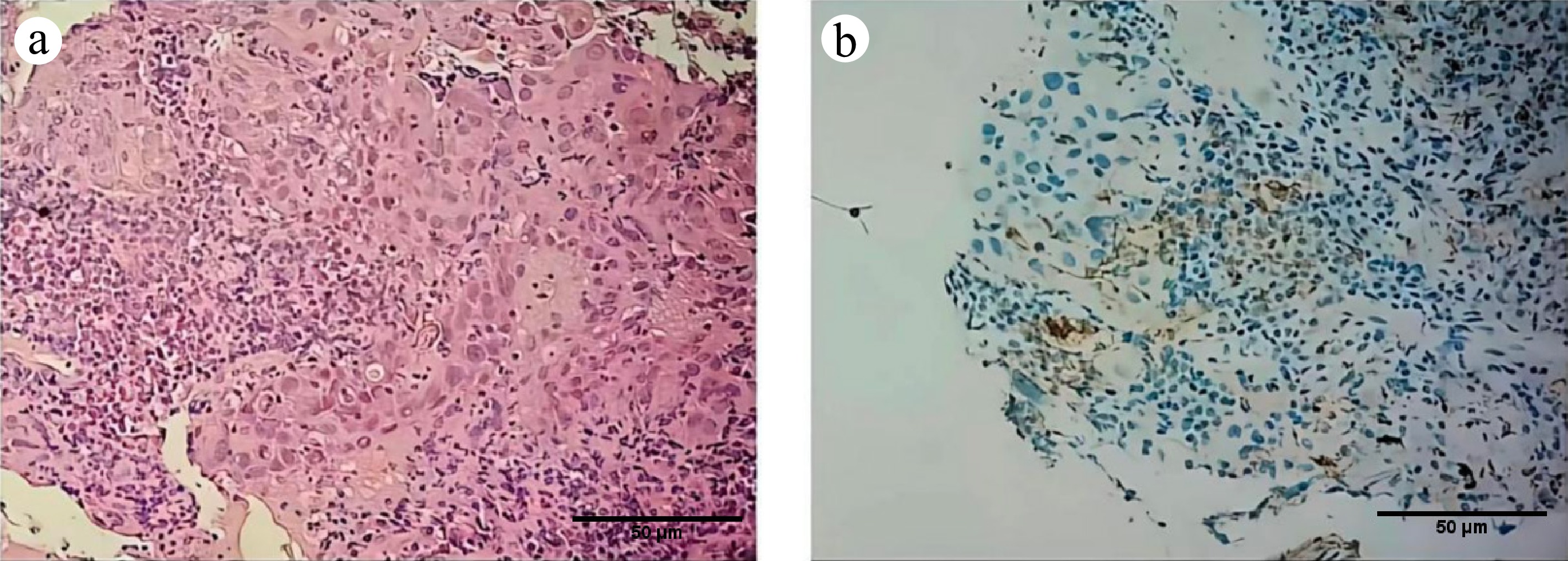
Figure 4.
Supplementary pathological examination of the lung metastasis: (a) 200×, (b) 200×. H & E-stained micrograph: shows the morphology of the tumor area.
Table 1. Immunohistochemical profile of the lung metastasis biopsy.
Antibody Result Staining intensity/extent Notes/significance Hepatocytic markers HepPar-1 Positive Diffuse strong positivity (> 80%) Confirms hepatocellular origin GPC-3 Focally positive ~30% of cells, moderate intensity Supports HCC diagnosis SMARCA4/BRG1 Negative Complete loss of nuclear staining in tumor cells Key diagnostic finding Proliferation index Ki-67 High ~60% nuclear positivity in tumor cells Indicates high proliferative activity Immune checkpoint PD-L1 (Clone E1L3N) Low expression TPS = 10% Epithelial and differential diagnosis markers CK7 Partially positive ~40% of cells positive CK19 Positive Diffuse positivity TTF-1 Negative 0% Rules out lung adenocarcinoma Napsin A Negative 0% Rules out lung adenocarcinoma P40 Negative 0% Rules out squamous cell carcinoma CK5/6 Negative 0% Rules out squamous cell carcinoma Based on these findings, a diagnosis of metastatic SMARCA4-deficient hepatocellular carcinoma was established. Following multidisciplinary discussion, the patient was treated with lenvatinib in combination with atezolizumab. Despite systemic therapy, the disease progressed rapidly, and the patient survived approximately one year following diagnosis. The patient’s clinical characteristics, imaging findings, pathological features, treatment, and outcome are summarized in Table 2.
Table 2. Summary of clinical, pathological, and treatment characteristics of the patient with SMARCA4-deficient hepatocellular carcinoma.
Category Details Demographics Age/sex 75-year-old male Chief complaint and history Admitted due to 'discovered pulmonary space-occupying lesions over one week after TACE therapy for liver malignancy' Physical examination on admission Conscious, fair general condition. Vital signs: T 37 °C, BP 120/79 mmHg, P 57 bpm, R 18 bpm, SpO2 97%. No jaundice. No palpable superficial lymphadenopathy. Clear breath sounds bilaterally without rales. Soft abdomen, no tenderness, liver and spleen not palpable below the costal margin. No pitting edema in lower limbs. Neurological examination normal Imaging findings Liver MRI Residual enhancing nodules in liver segments S7 and S8, suggesting residual tumor post-TACE Chest CT Nodule in the left lower lobe; widespread lymphadenopathy Brain MRI Multiple enhancing nodules throughout the brain, consistent with metastases Whole-body PET-CT Intense hypermetabolism in the liver lesions, left lung nodule, lymph nodes, bones, and brain, confirming multifocal metastatic disease Pathological diagnosis (lung biopsy) Histology (H&E) Poorly differentiated malignant tumor. Cells arranged in sheets and irregular nests, with hyperchromatic nuclei, high nuclear-to-cytoplasmic ratio, eccentric nuclei, and focally eosinophilic cytoplasm. Necrosis present Immunohistochemistry Positive markers: Hepatocyte(+), GPC-3 (focal+), CK7 (partial+), CK19 (+);
Negative markers: TTF-1 (−), Napsin-A (−), P40 (−), CK5/6 (−);
Crucial finding: SMARCA4/BRG1 (−/loss of expression);
Proliferation index: Ki-67 (approx. 60%+);
PD-L1 (Clone E1L3N): TPS = 10%Final diagnosis Metastatic SMARCA4-deficient hepatocellular carcinoma Treatment Lenvatinib (a multi-kinase inhibitor) combined with Atezolizumab (an anti-PD-L1 immune checkpoint inhibitor) Outcome Disease progressed rapidly despite combination therapy. Patient survival was approximately one year from diagnosis -
SMARCA4 is a core catalytic subunit of the SWI/SNF chromatin remodeling complex and plays a critical role in transcriptional regulation, cellular differentiation, and DNA repair. Loss of SMARCA4 function has been associated with highly aggressive, poorly differentiated malignancies, initially described in thoracic tumors and subsequently reported in gynecologic and gastrointestinal cancers[1]. Across multiple organ systems, SMARCA4 deficiency has emerged as a shared molecular driver of aggressive tumor biology, characterized by rapid progression, early metastasis, and poor clinical outcomes[2].
Although alterations in SMARCA4 have been identified in HCC, including copy number loss in a subset of cases, SMARCA4 immunohistochemistry is not routinely incorporated into standard pathological evaluation of liver tumors. Consequently, reports of SMARCA4-deficient HCC remain rare[3,4]. The present case expands the limited literature by illustrating the clinicopathological features and aggressive clinical course of primary SMARCA4-deficient HCC with widespread metastases.
Morphologically, SMARCA4-deficient HCC often presents as a poorly differentiated or undifferentiated carcinoma. As demonstrated in this case, the tumor exhibited sheets and irregular nests of malignant cells with marked nuclear atypia, eccentric nuclei, eosinophilic cytoplasm, and a high proliferation index. These features overlap substantially with those of other high-grade carcinomas, making diagnosis based on morphology alone challenging. Immunohistochemical evaluation is therefore essential. A definitive diagnosis relies on the demonstration of hepatocytic differentiation, indicated by positivity for markers such as Hepatocyte, Arginase-1, or glypican-3, in combination with complete loss of nuclear SMARCA4/BRG1 expression in tumor cells.
Imaging findings in SMARCA4-deficient HCC are non-specific but consistently reflect aggressive disease biology[5]. In this case, contrast-enhanced imaging and PET-CT revealed residual hepatic tumor activity and early, widespread metastatic involvement of the lung, lymph nodes, bone, and brain. This pattern aligns with reports from other organ systems, supporting the concept that SMARCA4 inactivation drives a highly malignant phenotype irrespective of tissue origin.
Therapeutically, SMARCA4 deficiency has been associated with limited responsiveness to conventional chemotherapy and targeted therapies. Although immunotherapy has shown activity in some SMARCA4-altered tumors, the relationship between SMARCA4 deficiency, PD-L1 expression, and response to immune checkpoint inhibition remains heterogeneous and context-dependent[2,3]. In the present case, Programmed Death-ligand 1 (PD-L1) expression was low (tumor proportion score 10%), which may partly explain the limited response to combined Lenvatinib and Atezolizumab therapy. Mechanistically, loss of SMARCA4 may influence chromatin accessibility and transcriptional regulation, potentially affecting genomic stability, antigen presentation, and immune signaling pathways[2]. As a result, responses to immune checkpoint inhibition in this setting are likely heterogeneous.
For patients with SMARCA4-deficient HCC who progress on first-line systemic therapy, subsequent treatment strategies should consider the underlying molecular context. Given the disruption of the SWI/SNF complex, targeting synthetic lethal interactions represents a rational therapeutic approach. Preclinical and early clinical data suggest that EZH2 inhibitors may demonstrate activity in SMARCA4-deficient tumors[6]. In addition, enrollment in clinical trials investigating novel targeted agents or next-generation immunotherapies should be strongly considered. Comprehensive molecular profiling and multidisciplinary management are essential to guide individualized treatment planning in this aggressive tumor subtype.
It should be noted that SMARCA4 deficiency is extremely rare in hepatocellular carcinoma. The present single-case study does not support recommending SMARCA4 as a routine immunohistochemical marker for all HCC cases. Further studies with larger cohorts are needed to better characterize the clinicopathological features and prognostic implications of SMARCA4-deficient HCC. With more robust evidence, we can reasonably discuss whether SMARCA4 immunohistochemistry should be included in the diagnostic panel for poorly differentiated hepatocellular carcinomas.
-
This case highlights the aggressive clinical behavior and distinctive pathological features of SMARCA4-deficient HCC. Morphologically, this tumor subtype frequently presents as a poorly differentiated or undifferentiated carcinoma and lacks specific features that reliably distinguish it from conventional high-grade hepatocellular carcinoma based on routine histology alone. As a result, heightened diagnostic awareness is required, particularly in cases demonstrating unexpected rapid progression or early metastatic dissemination.
We propose that SMARCA4 immunohistochemistry should be considered in poorly differentiated HCC, especially when clinical behavior is disproportionate to histological findings and sufficient tissue is available. Rather than routine universal testing, a risk-stratified diagnostic approach may be appropriate, prioritizing cases with poor differentiation at presentation or unexpectedly rapid progression.
However, given the rarity of SMARCA4 deficiency in hepatocellular carcinoma, larger cohort studies are required to systematically evaluate its clinicopathological features and prognostic significance. Such evidence will be essential to inform future discussions on whether SMARCA4 immunohistochemistry should be incorporated into routine diagnostic workflows for poorly differentiated HCC. Until then, a cautious, case-by-case approach remains warranted.
The authors would like to thank the patient and the clinical colleagues for their contributions to this study. This study was supported by the Discipline Cluster of Oncology, Wenzhou Medical University, China (Grant No. Z2-2023024).
-
This case report was conducted in accordance with ethical standards and received approval from the Ethics Committee of The First Affiliated Hospital of Wenzhou Medical University (Approval No. KY2025-R290, Date of Approval: 2025-09-17).
-
The authors confirm their contributions to the paper as follows: methodology, data curation: Liu Y; visualization, validation, software: Wu J; writing −original draft: Liu Y, Wu J, Zheng Y; writing − review and editing: Mitu MA, Shangguan F, Panashe MB, Zheng Y; supervision: Ma J, Shangguan F, Zheng Y; resources, conceptualization: Ma J, Zheng Y. All authors reviewed the results and approved the final version of the manuscript.
-
The clinical and imaging data supporting the findings of this study are stored in the electronic medical records system of the First Affiliated Hospital of Wenzhou Medical University and are available from the corresponding author upon reasonable request and subject to institutional approval.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Liu Y, Ma J, Wu J, Mitu MA, Shangguan F, et al. 2026. A case of SMARCA4-deficient hepatocellular carcinoma and its pathological characteristics. Gastrointestinal Tumors 13: e008 doi: 10.48130/git-0026-0006
A case of SMARCA4-deficient hepatocellular carcinoma and its pathological characteristics
- Received: 27 January 2026
- Revised: 19 February 2026
- Accepted: 05 March 2026
- Published online: 30 April 2026
Abstract: SMARCA4-deficient tumors represent a group of highly aggressive malignancies defined by the loss of SMARCA4 (BRG1) protein expression and have been most frequently described in thoracic and gynecologic organs. Reports of SMARCA4 deficiency in primary hepatocellular carcinoma (HCC) remain exceedingly rare. We describe a case of SMARCA4-deficient hepatocellular carcinoma with extensive metastatic involvement. A 75-year-old man with a prior diagnosis of HCC developed pulmonary nodules following transarterial chemoembolization (TACE). Histopathological evaluation of a lung biopsy demonstrated metastatic hepatocellular carcinoma with complete loss of SMARCA4 expression on immunohistochemistry. The tumor exhibited poorly differentiated morphology and aggressive clinical behavior, with rapid progression despite systemic therapy. This case highlights the clinicopathological features of SMARCA4-deficient hepatocellular carcinoma, underscores the importance of incorporating molecular pathological evaluation into routine liver cancer diagnostics, and provides insight into the implications of SMARCA4 deficiency for therapeutic decision-making.
-
Key words:
- SMARCA4-deficient tumor /
- HCC /
- Pathology