-
The depletion of traditional fossil energy resources and the intermittency of renewable energy resources have posed severe challenges to the global energy system. As a crucial solution to these problems, the development of related materials in the field of energy storage technology has become a core topic. Carbon-based electrode materials such as graphite, hard carbon, and porous carbon, which possess numerous advantages such as low cost, stable physical and chemical properties, high electrical conductivity, and easily adjustable structures[1−3], have been widely applied in key energy storage devices such as supercapacitors and lithium-sodium ion batteries.
To achieve high energy density and high cycling stability of energy storage devices, constructing the corresponding crystal and pore structures of carbon-based electrode materials is necessary[4]. Currently, the synthesis of carbon materials mainly relies on the traditional external heating carbonization method, which usually requires several hours for heating, retention, and cooling, presenting drawbacks such as high energy consumption and low efficiency[5−7]. More importantly, due to the inherent defects of traditional methods, such as slow thermal response and low heat transfer efficiency, it is difficult to precisely control the microstructure of carbon materials, and the performance of the final product often fails to meet the requirements of practical applications. The Flash Joule Heating (FJH) technology, as an innovative direct electrical heating method, converts carbon precursors into graphite or hard carbon materials through the instantaneous high temperature (> 3,000 K) generated by the current through the conductor[8]. Compared with traditional methods, FJH technology has the following significant advantages: high temperature (> 3,000 K), rapidity (104−105 K/s), high efficiency, no solvent, and low heat loss[9−11], overcoming the problems of slow response and severe heat transfer limitation in traditional heating conversion. In addition, the extremely high heating and cooling rates of FJH can achieve rapid graphitization, low-defect structure, break through the thermodynamic equilibrium limit, and lock the metastable active sites, optimizing the pore structure and surface chemistry[12,13], which cannot be achieved by the traditional external heating furnace carbonization method.
The surge in research on the synthesis of functionalized carbon materials via FJH technology originated from the research team of James M. Tour[14], who explored the method of synthesizing gram-scale Flash Graphene (FG) from various carbon-based raw materials via FJH. The research results showed that FG had an extremely low defect concentration (I2D/G = 17) and exhibited a unique interlayer disordered arrangement structure, which was expected to be applied in the energy storage field. This research achievement also paved the way for subsequent research work. Since that time, studies on utilizing different carbon sources (coal[15], petroleum coke[16], biomass[17], rubber[18], plastic[19], etc.) to synthesize functionalized carbon materials (such as graphene oxide [GOs][20], carbon nanotubes [CNTs][21], carbon fibers [CF][22], etc.) via FJH have shown an increasing trend year by year. There have also been reports on review articles on the synthesis of functionalized carbon materials using FJH: Dong et al.[23] made a relatively comprehensive summary of the reaction process, product structure, and performance of the early FJH-based carbon material synthesis. Deng et al.[24] conducted a systematic research summary on the application of FJH in environmental pollution remediation in the field of functional carbon materials. Mahmood et al.[25] summarized the research progress of converting different waste materials into graphene through FJH. There have been relatively few comprehensive reviews summarizing the synthesis of high-performance carbon-based electrode materials via FJH and their specific applications. In particular, further elaboration is needed regarding how different raw materials influence the structure and properties of the resulting products, as well as how process optimization can ultimately enhance final product performance. These aspects may not have been thoroughly addressed in existing reviews on FJH-derived carbon materials. Given the growing emphasis on energy storage technologies in industrial applications and the substantial research efforts in recent years, a systematic summary of advances in synthesizing high-performance carbon electrode materials using FJH is both timely and necessary.
Therefore, based on previous research experience, this review outlines the fundamental principles and key characteristics of FJH and highlights recent advances in the synthesis of high-performance carbon-based electrode materials using this technique. It systematically examines how various parameters and raw materials influence the structure of the resulting carbon products and their electrochemical performance. Building on current research progress in synthesizing carbon electrode materials from diverse carbon-based precursors via FJH, the review provides a comprehensive analysis—spanning from process optimization to final performance enhancement. Furthermore, it proposes targeted strategies to address challenges related to precise control and scalable application of carbon-based raw materials during the Joule heating conversion process. This review can help gain a better understanding of the current synthesis of carbon-based electrodes via FJH technology and offer constructive guidance for the development of energy storage materials and high-value utilization of carbon-based solid waste.
-
FJH essentially originates from Joule's law, which states that when current flows through a resistor, the resistance loss in the material within a certain period of time will convert electrical energy into thermal energy[26]. The mathematical expression is:
$ \mathrm{Q}={I}^{2}Rt $ (1) During the FJH process, the generated heat (Q) is directly proportional to the square of the current (I2), the resistance (R), and the heating time (t). Most experimental research utilizes this highly efficient and controllable thermal effect to achieve instantaneous high-temperature sintering. Compared to traditional heating methods (such as tube furnace heating), FJH has fundamental differences in heating principles and processes, which directly lead to its unique performance advantages: Traditional heating relies on external heat sources that transfer heat 'from the outside to the inside' slowly to the materials through radiation and convection. The entire process is limited by the heat conduction rate, resulting in a slow heating rate (typically several degrees Celsius to several dozen degrees Celsius per minute, as shown in Fig. 1a) and significant thermal inertia[27]. In contrast, FJH is an 'internal heating' process, where a strong current directly passes through the material itself, generating heat instantaneously throughout the volume, achieving efficient and uniform energy deposition. This direct 'bulk heating' mode enables it to achieve superb heating rates (up to 103 to 105 K/s, millions of times faster than traditional methods, as shown in Fig. 1b) and extremely high peak temperatures (> 3,000 K), and produces a smaller temperature gradient than traditional tube furnaces (as shown in Fig. 1c)[24,28].
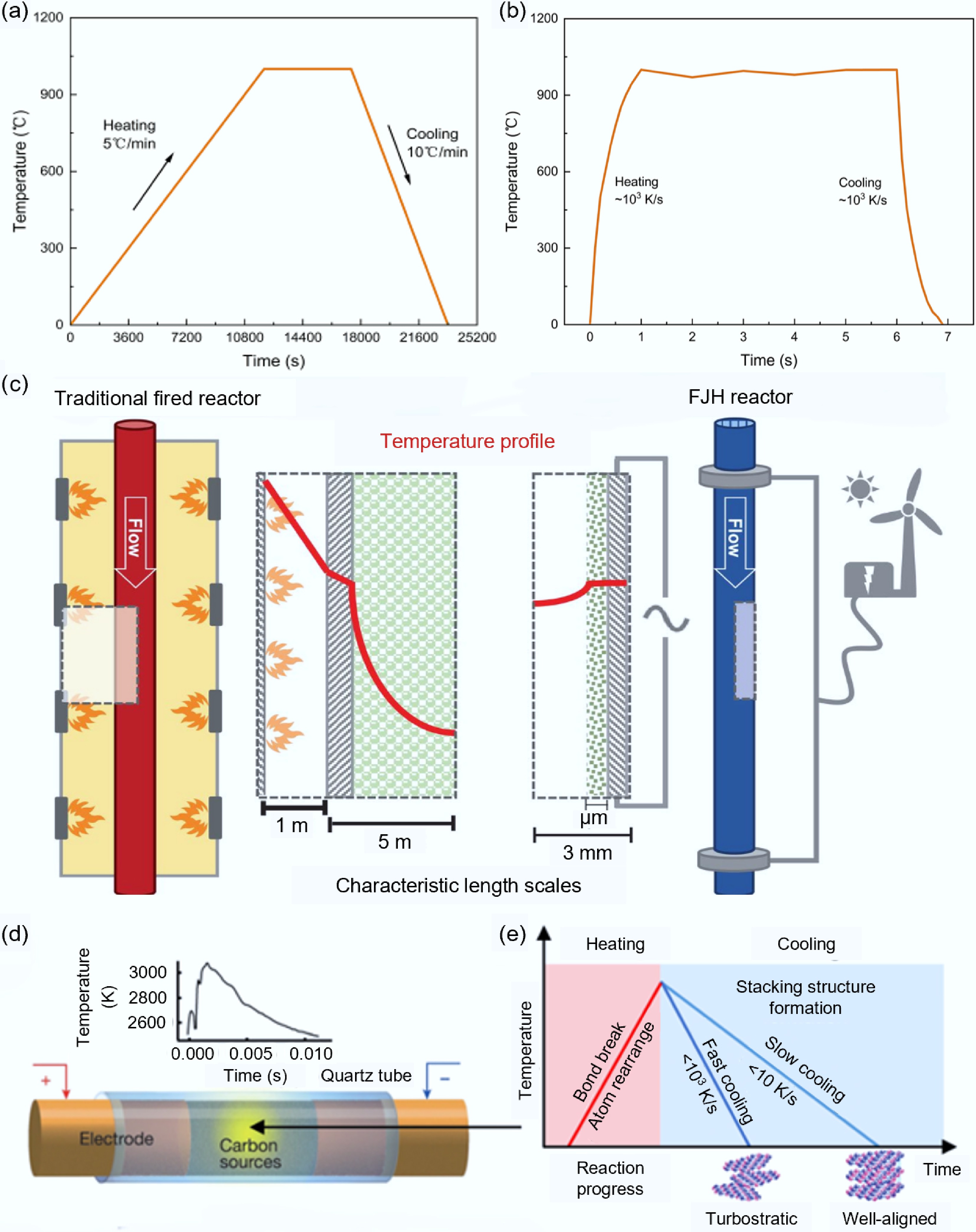
Figure 1.
Temperature characteristics comparison between FJH and traditional heating processes. (a) Heating/cooling rates of traditional heating method[27]. (b) Heating/cooling rates of FJH[24]. (c) Comparison of temperature distributions between traditional combustion devices and FJH reaction devices[28]. (d) Schematic diagram of FJH reaction[14]. (e) Effects of FJH heating/cooling rates on the structure of carbon products[32].
For the synthesis of carbon materials by FJH, the basic process is as follows: Connect the power supply directly to the resistive material and apply a short-term high-intensity current to cause the material to rapidly heat up to an ultra-high temperature state (> 3,000 K) within milliseconds to seconds (as shown in Fig. 1d). Under this extreme temperature condition, carbon atoms undergo intense vibration, chemical bonds break and recombine, thereby completing decomposition, transformation and structural reconstruction in an extremely short period of time, ultimately forming target products such as graphene, porous carbon, or other specific carbon materials[29,30]. The FJH process has two key features: One is that the transient Joule heating reaching the high temperature (> 3,000 K) plays a crucial role in effectively activating stable chemical bonds in the reactants, and the ultrafast heating can precisely achieve the breaking and rearrangement of chemical bonds, avoiding the generation of a large amount of unnecessary by-products (such as tar) during the traditional slow heating process[31]; The other is that the ultrafast rise promotes the reorganization of carbon into thermodynamically more favorable sp2 hybrid graphite layers, while the rapid quenching process hinders the transformation of graphite layers into the traditional AB stacking graphite structure (as shown in Fig. 1e), but forms vortex-shaped, highly graphitized, and low-defect concentration flash graphene (FG)[32].
During the FJH process, the resistive material can either be the carbon precursor itself that is to be carbonized or a conventional resistive component, such as the classic iron/chromium/aluminum (FeCrAl) alloy[33]. The advantage of using the carbon precursor as the resistive material is that the current can directly pass through the carbon material, enabling in-situ driving of the carbon structure evolution[34]. However, this method is limited by the conductivity of the carbon precursor and can be adjusted by pre-carbonization or adding conductive agents to meet the requirements of the FJH process. The use of resistive components is not limited by the material's resistance, but still relies on heat transfer between the component and the carbon material, resulting in significant scale effects[30,35]. Metal alloy resistive components are limited by their melting point and have limitations in applications under ultra-high temperature conditions. For example, the maximum operating temperature of the currently available heat-resistant FeCrAl alloy on the market is typically no more than 1,300 °C[36]. In contrast, carbon-based resistive materials, such as graphite, graphene, carbon fibers, carbon nanotubes, amorphous carbon, etc., have been widely used in the research of FJH carbon material synthesis due to their excellent high-temperature stability (with a tolerance temperature exceeding 3,000 °C)[37].
-
FJH is a direct approach for synthesizing carbon materials through rapid and high-temperature carbonization of carbon-containing raw materials. The final performance of the obtained carbon materials is significantly influenced by various factors such as the characteristics of the raw materials, the parameters of the experimental device, and the methods of quality improvement and modification during the process. This review aims to provide a comprehensive review of the mechanism of action and research progress of these key factors.
Carbon-based electrodes synthesis via different raw materials
-
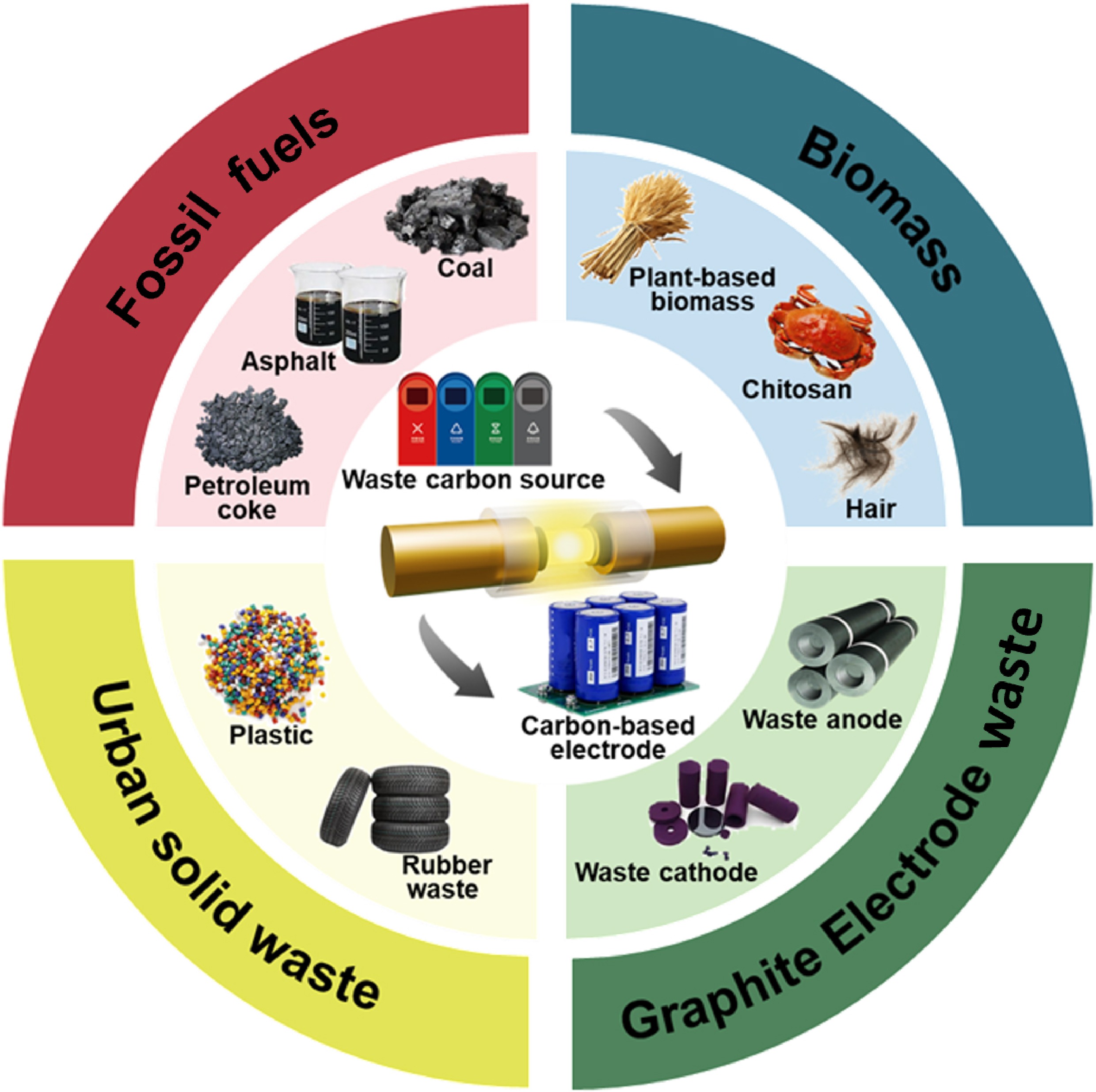
The chemical composition (fixed carbon, volatile matter content), physical properties (electrical conductivity, bulk density), microstructure of carbon-containing raw materials, etc., all affect the performance of the final carbon product and determine whether necessary pre-treatment methods are required to ensure the smooth progress of the FJH process. Table 1 shows the experimental conditions, product characteristics, and related applications (supercapacitor [SC], sodium-ion battery [SIB], lithium-ion battery [LIB]) of carbon materials synthesized through pyrolysis of different carbon-based raw materials by FJH. These raw materials can be classified into the following four categories: fossil fuels (asphalt, coal, etc.), urban solid waste (plastic, rubber, etc.), graphite electrode waste (waste cathode materials, waste anode materials, etc.), and biomass (lignin, cellulose, chitosan, etc.). The following parts will focus on the research progress on the synthesis of functionalized carbon-based electrode materials via FJH for these three main carbon-based raw materials, and analyze the effect of raw material composition on the final material performance.
Table 1. Synthesis of carbon-based electrode materials from different raw materials via FJH
Carbon source Parameter conditions Key structural characteristics
of the productElectrochemical performance
of the productApplication Ref. Asphalt ~3,000 K 3–5 × 105/1 × 104 K/s
Capacitor 160 V, ~1 sd(002) = 3.71 Å, ID/IG = 0.2 Specific capacitance: 210 F/g (1 A/g) SC [41] Bitumite ~3,000 K 8.5 × 104/8 × 103 K/s
Capacitor 100 V, ~1 sd(002) = 3.38 Å, ID/IG = 1.15
surface area: ~620 m2/g
Porosity rate: ~70%Specific capacitance: 136 F/g (1 A/g) SIB [44] Lignite ~3,000 K 8.5 × 104/8 × 103 K/s
Capacitor 140 V, ~1 sd(002) = 3.43 Å, ID/IG = 1.15
surface area: ~450 m2/g
Porosity rate: ~60%Specific capacitance: 95.4 F/g (1 A/g) SIB [44] Anthracite ~1,573 K —/—
DC 42.5A, ~20 sd(002)= 3.789 Å, ID/IG = 1.09
surface area: ~8.01m2/g
Pore volume: 0.018 cm3/gReversible capacity: 256.2 mAh/g capacitance retention rate: 93.2%
after 200 cycles.SIB [45] Anthracite ~1,073 K —/—
Capacitor 150 V, ~0.1 sd(002) = 3.42 Å, ID/IG= 0.94
surface area: ~9 m2/gReversible capacity: 209 mAh/g
(0.05 A/g), with no significant degradation after 500 cyclesSIB [46] Plastic ~2,500 K —/—
Current increased from 0.1 A to 25 A, 50 s
Capacitor 100 V, 0.25 sd(002) = 3.56 Å, ID/IG =0.61
surface area: ~874m2/gDuring the 20-h long-cycle test, overpotential increased by only 19 mV (20 mA/cm²) LIB [55] Waste anode 2,850 K 1.6 × 105/9.2 × 103 K/s
Capacitor 200 V, 0.1 sd(002) = 3.35 Å, ID/IG =0.14
surface area: ~15.4 m2/gCapacitance: 131.1 mAh/g (0.2 C) LIB [60] Waste anode ~3,000 K —/—
Capacitor 200 V, 0.1 sd(002) = 3.389 Å, ID/IG = 0.103
surface area: ~68.345 m2/gCapacitance: 350 mAh/g (1 C)
Capacitance retention rate: ~99% after 500 cyclesLIB [62] Sawdust ~3,100 K —/—
AC 200 V, 6 s; DC
150 V, 0.03 sd(002) = 3.45 Å, ID/IG = 0.2
surface area: ~53 m2/gCapacitance: 280 mAh/g (1 C)
Capacitance retention rate: ~90% after 200 cyclesSC [72] Hair ~1,673 K —/—
Capacitor 400 V, 0.15 sd(002) = 3.357 Å, ID/IG = 0.2
Surface area: ~13.107 m2/g
Pore volume: 0.018 cm3/gCapacitance: 500 mAh/g (0.1 C) LIB [74] Chitosan ~1,573 K —/—
DC 40 A, 1 sd(002) = 3.83 Å, ID/IG = 1.13
Surface area: ~7.33 m2/gDuring low-temperature test, at −20 °C, Reversible capacitance:149 mAh/g, after 1,000 cycles SIB [76] Fossil fuels
-
Early FJH research mainly used coal-based materials derived from fossil fuels as raw materials, such as asphalt, low-rank coal (lignite, bituminous coal), and high-rank coal (anthracite), etc. This type of raw material, due to its high fixed carbon content and good electrical conductivity, can be directly carbonized at high temperatures without the addition of conductive agents, and the final carbon yield is also relatively high (70%–90%)[14,16,38]. The condensed aromatic structure of fossil fuels is conducive to the formation of fibrous graphite, but the uneven particle size and anisotropy lead to an uneven distribution of fibrous graphite layers, which requires staged pretreatment to optimize the process[39]. The following will respectively introduce the carbonization process of asphalt driven by FJH and the application of related products in carbon-based electrode materials, as well as the influence of coal raw materials of different coal grades on the performance of carbon-based electrode materials.
Asphalt is regarded as an ideal precursor for preparing high-performance carbon materials due to its high carbon content and high aromaticity. The FJH process has a decisive influence on the morphology and structure of the final product. Under high-temperature conditions, the coal asphalt molecules undergo branch-chain breakage and active free radicals' generation. These free radicals, under the combined effect of an electric field and high temperature, recombine to form graphene (Fig. 2a). The rapid cooling characteristic of this process effectively inhibits AB stacking, thereby facilitating the formation of high-quality graphene[40]. Zhu et al.[41] confirmed the above mechanism: they used coal asphalt as the carbon source and successfully synthesized the flash porous carbon (F-PC) through a capacitance discharge-type FJH system, and used molecular dynamics simulation and DFT calculations to reveal its graphitization structure evolution. When the obtained FG was applied to supercapacitors, its electrochemical performance significantly improved. As shown in Fig. 2b, c, the closed area of the CV curve of the porous carbon (PC) electrode after FJH treatment significantly increased, indicating that it had a higher specific capacitance. Moreover, the slope of the low-frequency region of its Nyquist plot (Fig. 2d) was greater, further confirming the enhancement of the capacitive behavior.
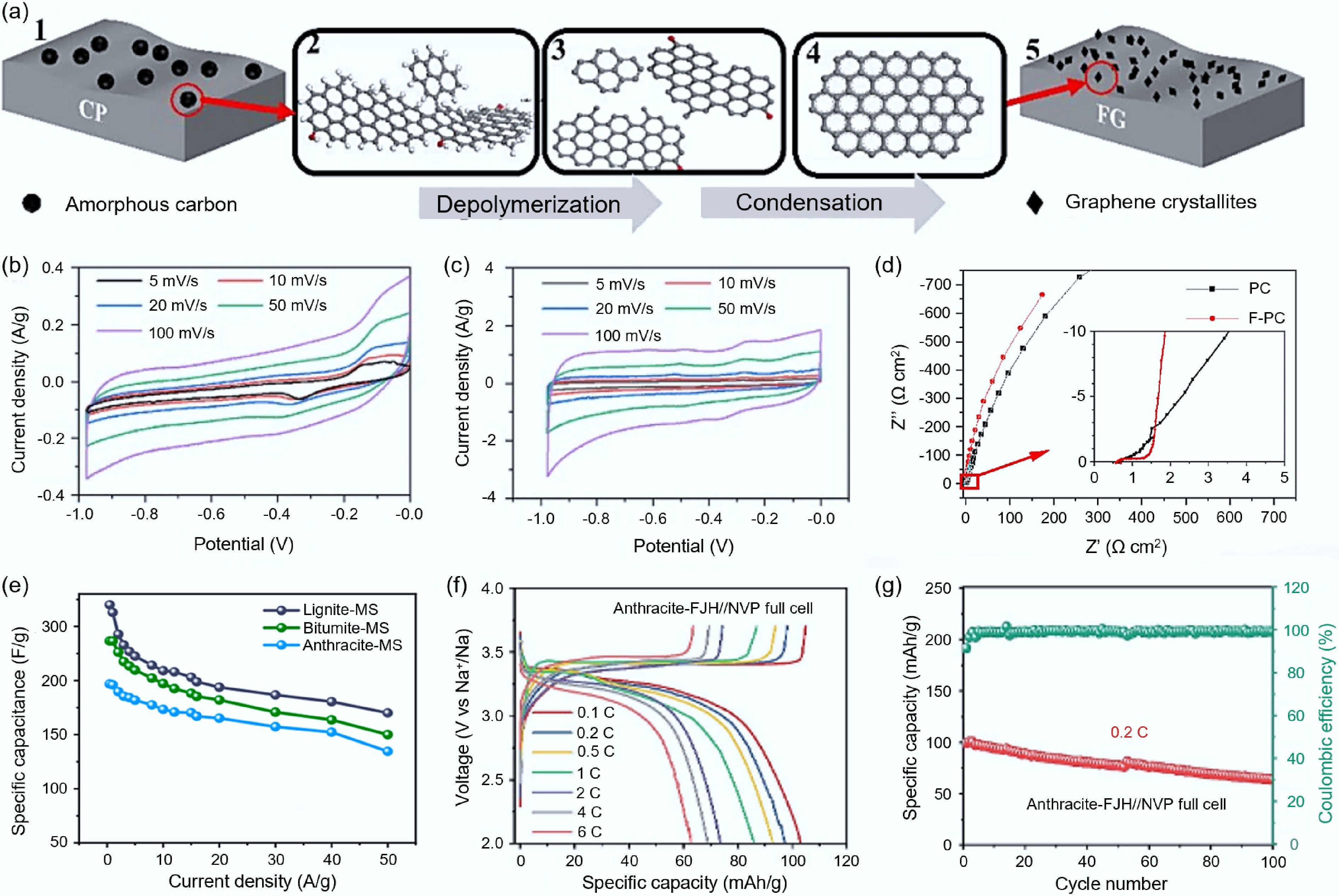
Figure 2.
Synthesis of carbon-based electrode materials from fossil fuels via FJH. (a) Schematic diagram of the formation mechanism of coal-based-FG[41]. (b) CV curve of the supercapacitor battery assembled with PC[41]. (c) CV curve of the supercapacitor battery assembled with F-PC[41]. (d) Nyquist plots of PC and F-PC[41]. (e) Specific capacitance of carbon materials synthesized from lignite, bituminous coal, and anthracite at different current densities[44]. (f) CD spectrum of all batteries assembled with Na3V2(PO4)3 positive electrode and graphite from anthracite at different rates[45]. (g) Cycling performance of all batteries assembled with Na3V2(PO4)3 positive electrode and graphite from anthracite at 0.2 C[45].
The lower rank lignite and bituminous coal have high volatile content characteristics. During the FJH process, these volatile components rapidly escape under the instantaneous high temperature, not only generating abundant pores that provide channels for the subsequent activation agents to enter and act, but also triggering a significant 'thermal expansion effect'[42]. Zhang et al.[42] used bituminous coal as the precursor, and after FJH treatment, they found that the proportion of FG micropores was dominant, and most of the micropores were smaller than 1 nm. The number of mesopores and macropores was relatively small, indicating that the transient high temperature and discharge process of FJH regulated the pore structure of coal-based carbon materials. The porous graphite materials obtained from the FJH transformation of low-rank precursors also exhibited high capacitance in the assembled all-cell batteries. Hu et al.[43] used FJH to instantly convert lignite precursors into high-performance negative electrode materials for sodium-ion batteries in an argon atmosphere. The assembled all-cell battery, when discharged at a high rate (10 C) at room temperature (25 °C), still maintained a capacity of 70.5 mAh/g for the electrode material.
Compared with lower-rank coal, anthracite has a higher fixed carbon content and aromaticity, which is conducive to obtaining graphite carbon materials with the least structural defects and achieving high-rate charge storage. Early studies by Ni et al.[44] showed that under a 30 A/g condition, anthracite tended to form long-range ordered carbon materials, with a high charge storage rate, and its capacity retention rate was as high as 79.1%. While graphite carbon materials derived from lignite and bituminous coal had a lower coalification degree and a larger specific capacitance, with a capacity retention rate lower than that of anthracite (as shown in Fig. 2e). Subsequent research mainly focused on the excellent rate performance and long cycle life of anthracite materials in capacitors and batteries. Wu et al.[45] completed the structural transformation of anthracite using FJH, and the product had a large number of closed nanochannels, significantly enhancing the adsorption and filling capacity for sodium ions. When assembled with the Na3V2(PO4)3 cathode, the energy density reached 225.3 Wh/kg, and at a 6 C rate, it could achieve a high capacity of 62.8 mAh/g. After 100 cycles at a 0.2 C rate, it maintained a high specific capacity of 108.7 mAh/g, with a capacity retention rate of 64.5% (as shown in Fig. 2f, g). Cao et al.[46] used anthracite as the precursor for FJH to synthesize hard carbon anode materials, and the sodium-ion battery assembled with these materials demonstrated a high reversible capacity of 209 mAh/g at a 0.05 A/g current density, and still maintained 115 mAh/g at a 1 A/g current density. After 500 cycles, there was no significant capacity degradation.
Urban solid waste
-
Plastics, rubber, waste graphite electrodes, and other urban solid wastes, due to their large quantities, have posed significant challenges in the field of environmental protection regarding their proper disposal and resource utilization. In recent years, FJH technology has provided a new approach for achieving the high-value conversion of such solid wastes, and has been widely applied in the research of converting them into functionalized carbon materials.
Rubber/plastic polymers, which are high-molecular aliphatic hydrocarbon compounds, have a high carbon content. Common plastics (such as polyolefins [PE, PP]) have a high carbon-hydrogen content and a high conversion rate, and are prone to form sp2 carbon structures[47]; in tire rubber, the inherent sulfur and zinc generate heteroatom-doped graphite in situ, and this material may present better performance than pure graphite in sodium-ion batteries[48]. These characteristics make rubber/plastics more promising for synthesizing high-quality functionalized carbon materials compared to fossil fuels. However, unlike fossil fuels with high conductivity, these raw materials themselves are basically non-conductive. To ensure that the samples have sufficient conductivity to support the rapid discharge current required for high-temperature processes, a two-step FJH method was proposed[18,49]: 3 wt%–5 wt% carbon black was mixed in the sample to increase the electrical conductivity of the material. A long-duration (approximately 10 s) low-current discharge was performed first to carbonize the carbon-containing raw materials and volatilize them. After this low-current FJH, the volatile mass depends on the loss of the raw materials, and the sample resistance is < 10 Ω, allowing for a large current FJH (as shown in Fig. 3a).
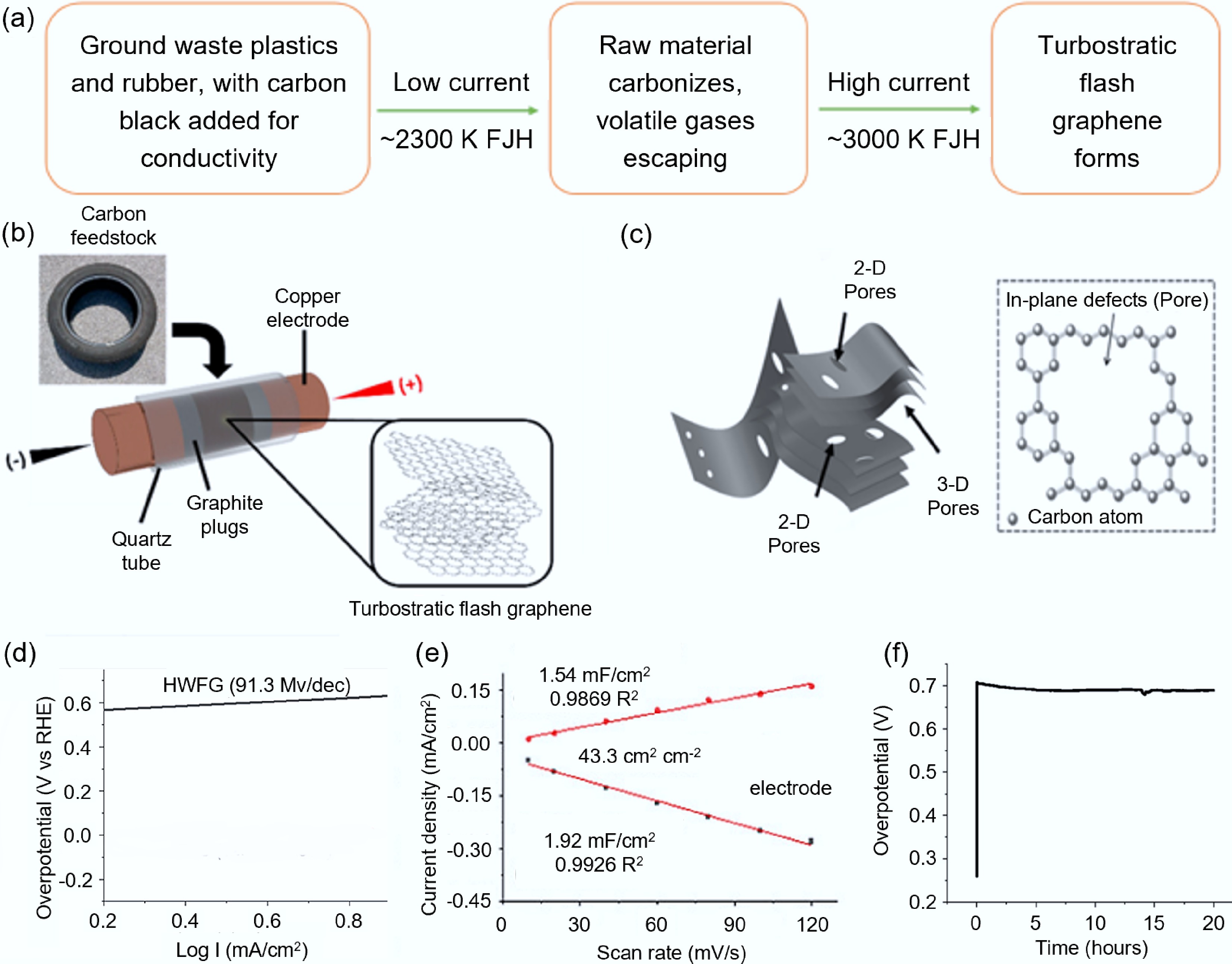
Figure 3.
Synthesis of carbon-based electrode materials from plastics and rubber waste via FJH. (a) Flowchart of the two-step FJH method[18]. (b) Schematic diagram of tFG synthesis[18]. (c) Structural diagram of HWFG[55]. (d) Tafel slope. (e) Capacitance measurement (the inset shows the current density at different scan rates)[55]. (f) Stability test of HWFG at 20 mA/cm2[55].
After the rubber raw materials are treated by FJH, a special turbostratic flash graphene (tFG) with a unique structure can be formed. As shown in Fig. 3b, compared with the conventional flash graphene (FG), tFG exhibits more significant characteristics of a turbostratic layer structure. Usually, an increase in the number of graphene layers leads to a weakening of the ideal two-dimensional properties (such as electronic properties) due to interlayer coupling[50,51]. However, in tFG, the spacing between the layers increases, and the graphene films are arranged in a relative rotational manner. This special structure enables the two-dimensional strength to increase with the increase in the number of layers, while the expansion of the layer spacing inhibits the interlayer electron transmission, allowing the two-dimensional material properties to be retained in the multi-layer system[52]. This characteristic significantly enhances its mechanical properties, making it well-suited for applications in the construction materials field, such as enhancing the mechanical properties of cement mortar[53]. Although the research on directly converting rubber into electrode materials based on the FJH technology is still in its infancy, the achievements of the traditional pyrolysis method have pointed out the direction for it. Existing studies have transformed waste tires into three-dimensional vertical graphene through a two-step pyrolysis method, and this material, as an electrode, has demonstrated excellent reversible capacity (252.7 mAh/g @ 0.2 A/g) and rate performance (105.4 mAh/g @ 5 A/g)[54]. This indicates that if a more efficient and energy-saving FJH technology is adopted, it is expected to further improve the comprehensive performance of the electrode materials while simplifying the process, and has great development potential in fields such as lithium-ion batteries and sodium-ion batteries.
Unlike rubber, the plastic raw materials treated by FJH can usually be synthesized into Holey & Wrinkled Flash Graphene (HWFG) with porous and wrinkled structures, as shown in Fig. 3c. During this process, high temperature of FJH process induces both physical and chemical activation effects, which not only stabilize the porous carbon framework but also promote the formation of high specific surface area, ultimately resulting in high-quality HWFG[55]. Compared to general FG materials (with a specific surface area of approximately 295 m2/g and pores mainly consisting of micropores and mesopores)[14], HWFG has a higher specific surface area (650–874 m2/g) and a wider pore size distribution, covering micropores, mesopores, and macropores. This unique pore structure and surface functional group characteristics enable HWFG to present excellent stability and high discharge rate performance in lithium metal battery anodes. The result of James M. Tour's research team[55] indicated that, when HWFG was used as the negative electrode, the Tafel slope was 91.3 mV/dec, indicating its efficient charge exchange ability; the electrochemically active area of the electrode reached 43.3 cm2. In a 20-h long cycle test, the overpotential of the electrode only increased by 19 mV at a current density of 20 mA/cm2, demonstrating excellent electrochemical stability (Fig. 3d–f).
Graphite electrode waste
-
In addition to the aforementioned solid wastes such as plastics and rubber, electronic solid wastes are also the current research hotspots. With the development and popularization of electronic devices, the demand for various batteries is increasing day by day. The annual generated waste from graphite electrodes has exceeded 50 million tons[56]. How to handle these massive amounts of used lithium-ion battery graphite electrodes and achieve the recycling and reuse of resources has become an urgent problem to be solved at present. The current FJH technology avoids the disadvantages of traditional processing methods, such as high energy consumption, structural defects, and poor electrochemical performance[57,58], and realizes the efficient, low-cost, and environmentally friendly regeneration of used lithium-ion battery graphite negative electrode materials[59]. The following will specifically introduce the research results of FJH processing of waste graphite electrodes from the perspectives of the advantages of FJH compared to traditional processing methods and the improvement of the performance of regenerated waste electrode graphite.
The waste graphite anode regenerated by FJH has significant advantages in terms of structure and electrochemical performance compared to the traditional calcination treatment method. The research team of James M. Tour[60] directly treated the used graphite anode with FJH and compared it with the ordinary calcination treatment. The results showed that the average ID/G and I2D/G strength ratios of the rapidly regenerated graphite were 0.14 and 0.56, respectively, while the values of the calcined regenerated graphite were 0.29 and 0.44 (as shown in Fig. 4a, b), which indicated that the rapid regeneration method can maintain the original structure of the graphite and avoid the formation of defects. The specific capacity of the negative electrode regenerated by FJH (FRA) was significantly higher than that of the untreated waste graphite electrode (AW), although the specific capacity of the calcined recovered negative electrode (CRA) was similar to that of FRA and commercial graphite at low rates (< 0.2 C), but at high rates, the specific capacity of CRA significantly decreased and the overpotential increased (as shown in Fig. 4c). The specific capacity of AW was relatively low, approximately 94.9 mAh/g, with poor stability. After 400 cycles at 0.2 C, the average capacity decay per cycle was 0.17% for AW. The specific capacities of FRA and CRA were 131.1 and 129.8 mAh/g, respectively. However, the electrochemical stability of FRA was better, with a per-cycle decay of 0.078% (as shown in Fig. 4d).
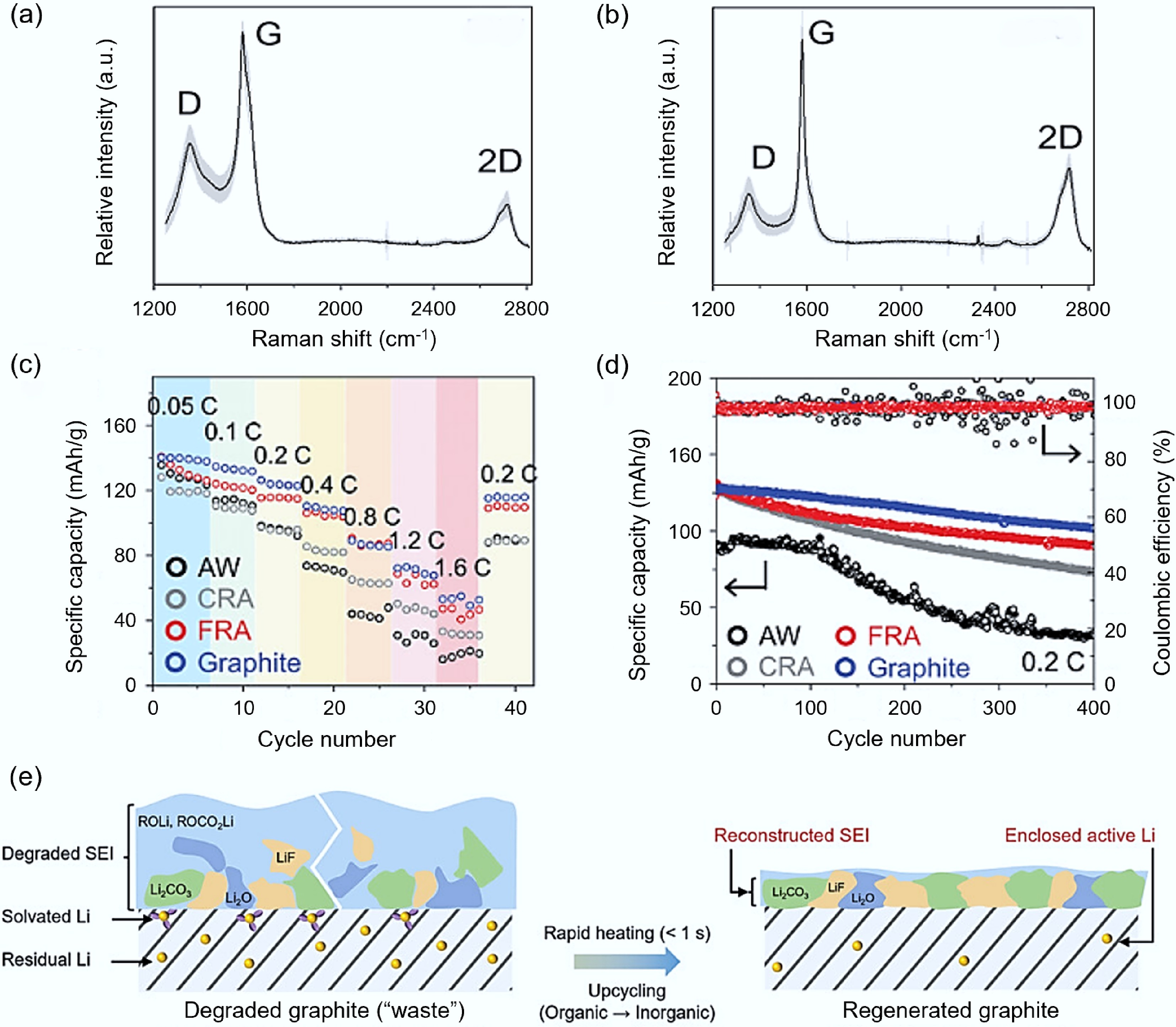
Figure 4.
Regeneration of waste anode graphite via FJH. (a) CRA Raman spectrogram[60]. (b) FRA Raman spectrogram[60]. (c) Comparison of electrode performance among AW (black), CRA (gray), FRA (red), and commercial graphite (blue)[60]. (d) Comparison of cycling performance of AW (black), CRA (gray), FRA (red), and commercial graphite (blue) at 0.2 C current[60]. (e) TEM process diagram of the transformation from thick and loose SEI layer to thin and dense SEI layer[63].
The traditional wet chemical method has difficulties in removing adhesives and optimizing the solid electrolyte interface (SEI) layer, and the recovery of electrochemical performance is slow. The FJH technology utilizes a strong current to promote the repair and reconstruction of the graphite structure, while removing the SEI layer. It also forms a conductive layer on the graphite surface through the thermal decomposition products of conductive agents and adhesives, thereby increasing its conductivity[61]. This characteristic has been well applied in related research. Cao et al.[62] treated recycled waste graphite with FJH. The size of the recycled graphite after FJH treatment slightly decreased, but it did not crack, indicating that the removal of adhesives was successful. The discharged capacity of the recycled graphite at 1 C current density reached 350 mAh/g, and the capacity retention rate after 500 cycles reached 99%, equivalent to a capacity attenuation of 0.02% per cycle, which is much better than that of waste graphite (capacity retention rate of 95%). Ji et al.[63] considering the formation of the SEI layer and the accumulation of residual active lithium during electrochemical cycling, utilized FJH to transform the thick and loose SEI (thickness ≈ 100 nm) into a thin and dense SEI (thickness 10–30 nm) covering the graphite surface (as shown in Fig. 4e). The reconstructed SEI and encapsulated active lithium gave the recycled graphite excellent initial Coulombic efficiency (ICE), and compared with commercial graphene CG in the full battery configuration, ICE (98.8% vs 83.2%) and energy density (309.4 vs 281.4 mAh/g) were significantly improved. Shang et al.[64] synthesized recycled graphite using a similar method and demonstrated excellent performance, showing a specific capacity of 358 mAh/g at 0.1 C, and a capacity retention rate of 94.83% after 100 cycles.
Biomass
-
Biomass, as the only carbon-neutral renewable resource at present, has significantly reduced the environmental impact caused by industrial fossil fuels and some of urban solid waste, aligning with the concept of sustainable development. Therefore, it is one of the optimal paths for achieving the green and low-carbon development of the FJH technology in the future and has attracted the attention of researchers. However, biomass typically contains a high oxygen content, so during the FJH process, a large amount of pyrolysis volatiles are released, accounting for 60%–70% of the carbon emissions, causing the collapse of the carbon skeleton and quality loss, resulting in an output rate of less than 30%[44,65]. In terms of yield and process stability, it is inferior to fossil fuels and urban solid waste[14,41]. Currently, to address these issues, it is usually necessary to pre-pyrolyze to release the volatiles of biomass first, and then conduct the FJH reaction to optimize the structure of the flash graphene[66]. Most biomass, like plastics/rubber, is not conductive by itself, so conductive agents such as carbon black need to be added. At present, the main focus of FJH processing of biomass raw materials is on lignin and cellulose in agricultural and forestry crops, proteins extracted from animals, chitosan, etc. In the following part, the reaction mechanism of these biomass raw materials under the FJH action and the performance characterization of the synthesized functional carbon electrodes will be introduced.
Agricultural and forestry crops contain abundant lignin, which is a renewable aromatic polymer with a high carbon content (> 60%) and is an ideal precursor for carbon materials[67]. However, due to the high branching and oxygen-rich structure of lignin, the degree of graphitization of the derived carbon is usually low, resulting in poor conductivity, thermal conductivity, and stability, which hinders its practical application[68]. The high-temperature pulsed waves above 3,000 K generated by FJH can effectively destroy the aromatic rings in lignin, and subsequent rearrangement and condensation reactions will occur, thereby forming large aromatic clusters. The mechanism is shown in Fig. 5a, b. The decomposition of rigid aromatic rings to provide sufficient free carbon atoms and the rapid and orderly rearrangement of free carbon atoms induced by microcrystals are the main factors promoting the graphitization of lignin during the FJH process[69]. The degree of graphitization and electrical conductivity of the products after FJH treatment have significantly improved[70], making it suitable for applications in the electrochemical field, such as batteries and supercapacitors. Zhao et al.[71] synthesized lignin flash graphene (LFG) by using lignin as the precursor. The supercapacitor based on LFG showed a high capacitance retention rate (~96%) even after 10,000 charge-discharge cycles. Jia et al.[72] converted forestry and agricultural residues into LFG using FJH. The constant current charge-discharge curve of LFG materials showed that the second and third cycles of the material are basically overlapping (as shown in Fig. 5c), indicating that LFG materials had good structural stability and reversibility. After 200 charge-discharge cycles, the capacitance showed almost no attenuation (capacitance retention rate > 90%), approximately 280 mAh/g (as shown in Fig. 5d).
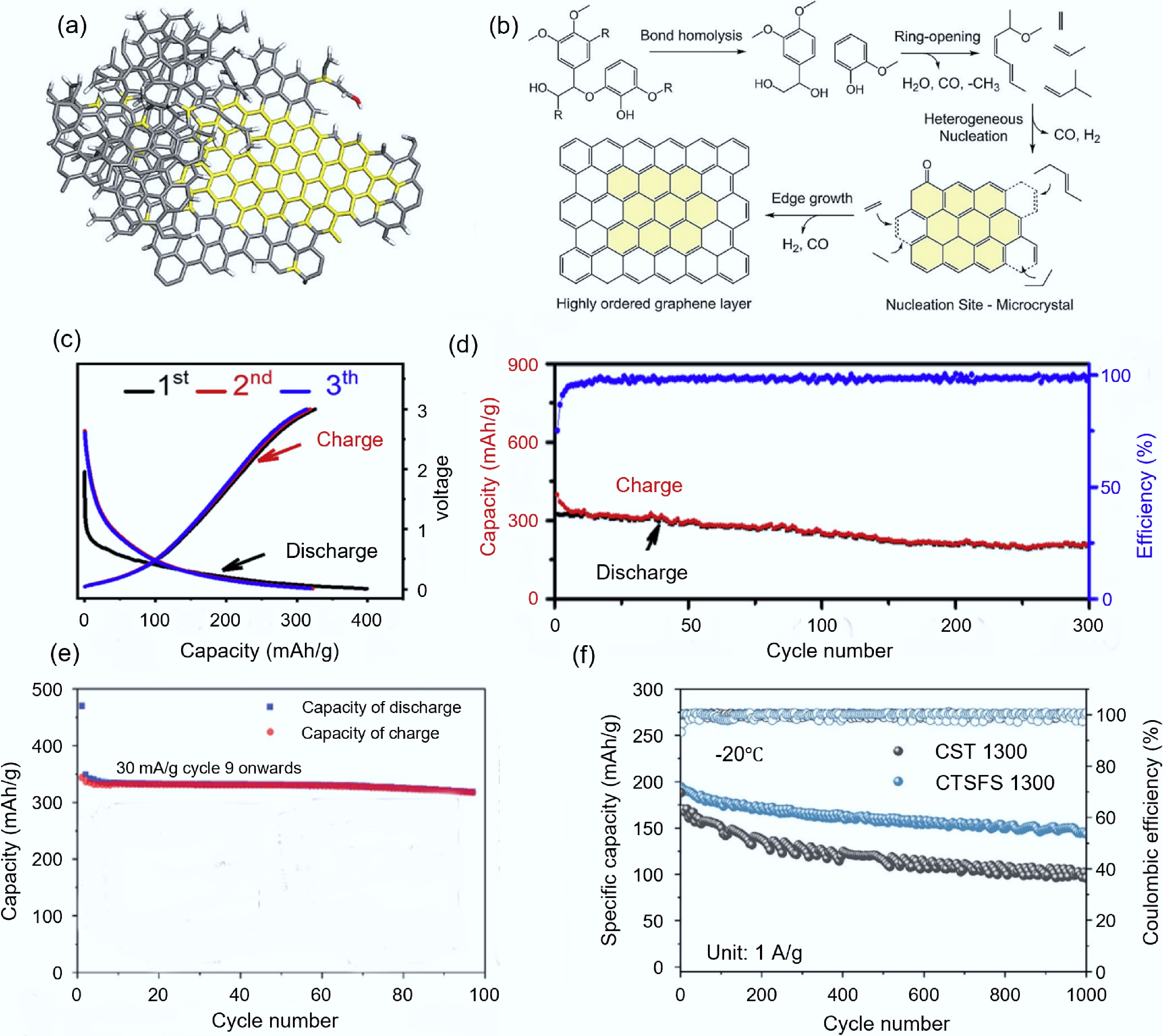
Figure 5.
Synthesis of carbon-based electrode materials from biomass via FJH. (a) Structure diagram of LFG[69]. (b) Mechanism of lignin graphitization[69]. (c) Cycling performance and Coulombic efficiency of LFG transformed from agricultural and forestry residues[72]. (d) Constant current charge-discharge curve of LFG materials[68]. (e) Constant current charge-discharge curve of GH materials[74]. (f) Cycling performance of CTS-1300 and CTSFS-1300 at −20 °C[76].
Moreover, FJH can convert animal-based biomass raw materials into multifunctional carbon materials, such as extracting keratin from human hair and chitin from crustaceans. Human hair, as a biomass carbon source rich in keratin, after being subjected to high-temperature treatment, nitrogen and sulfur elements escape in the form of gaseous oxides, thereby obtaining the purest carbonaceous substance[73]. Jose et al.[74] transformed human hair into high-quality graphene carbon (GH) through FJH. As a negative electrode material for lithium-ion batteries, the GH material, at a discharge rate of approximately 0.1C, ultimately achieved a specific capacity of 500 mAh/g (as shown in Fig. 5e). Chitosan exists in the shells of crustaceans and is widely available, low-cost, and environmentally friendly. The porous carbon-based materials derived from it have the advantages of light weight, tunable intrinsic conductivity, and environmental friendliness[75]. Therefore, it is often used as a precursor for preparing hard carbon electrode materials. Shen et al.[76] synthesized hard carbon materials CTSFS-1300 with smaller particle size, larger interlayer spacing, and larger closed pores through FJH using chitosan as the precursor. Compared with the carbon material CTS-1300 that was not treated by FJH, CTSFS-1300 presented excellent low-temperature performance: at −20 °C, as shown in Fig. 5f, after cycling 1,000 times, the reversible capacities of CTSFS-1300 and CTS-1300 were 149 and 94 mAh/g, respectively, and the capacity retention rates were 76.4% and 55.7%, respectively. CTSFS-1300 presented excellent sodium ion storage performance at low temperatures.
Optimize control factors for high-quality carbon-based electrodes
-
Based on the clarification of the influence of precursor raw materials, it is necessary to consider the influence of process control factors (such as condition parameters and equipment conditions), as these factors serve as the key bridge and regulatory means linking precursor raw materials with the performance of the final product. At the same time, by optimizing these process control factors, the deficiencies of the raw material itself can be compensated, promoting the effective synthesis of target carbon materials through FJH. The following section introduces the impact of process control factors on the performance improvement of carbon-based electrode materials from the perspectives of condition parameters and equipment conditions.
Parameters
-
The carbonization via FJH is a complex process involving multiple physical fields and spatial-temporal dimensions of electricity. The electrical parameters (pulsed current, flash voltage), time parameters (flash time, flash frequency), and flash temperature and other condition parameters are closely related to the structural characteristics of the final carbon products. The structural characteristics of the products affect their electrochemical applications. The following will specifically discuss the influence of these condition parameters on the structure and electrochemistry of the carbon products.
Electrical parameters. Pulsed current during the FJH process has a significant impact on the electrochemical performance of the final product. Under the same flash time, the electrode materials treated with high current intensity showed superior electrochemical performance. Rajendran et al.[77] compared the graphene synthesized under different current intensities (50 and 100 A) after treating carbon nanodots (CNDs) with FJH at a flash evaporation time of 0.5 s. They found that the electrode treated with 100 A current intensity (F100) showed higher crystallinity and a more ordered graphite structure (as shown in Fig. 6a), and its electrochemical performance was significantly improved. Figure 6b shows the 100 mV/s CV curve, and the F100 electrode demonstrated a higher closed area. At a current density of 1 A/g, it presented a high specific capacitance of 279 F/g, significantly superior to the electrode treated with 50 A current intensity (80.9 F/g). Besides the current intensity itself, its frequency is also closely related to the structural evolution of the carbon product. Adjusting the pulse width can control the material's transformation from amorphous carbon to disordered graphene, and ultimately to ordered graphene and graphite (AB and ABC stacking) in a three-step phase transition process[78]. By modulating the pulse width (PWM) of the current frequency, the quality and uniformity of graphene can be improved while reducing energy consumption. The experimental results of James M. Tour's research team[79] showed that the current modulated with PWM at a frequency of 1 kHz could significantly increase the crystallinity of graphene compared to the general DC drive, and reduce its defect density (as shown in Fig. 6c).
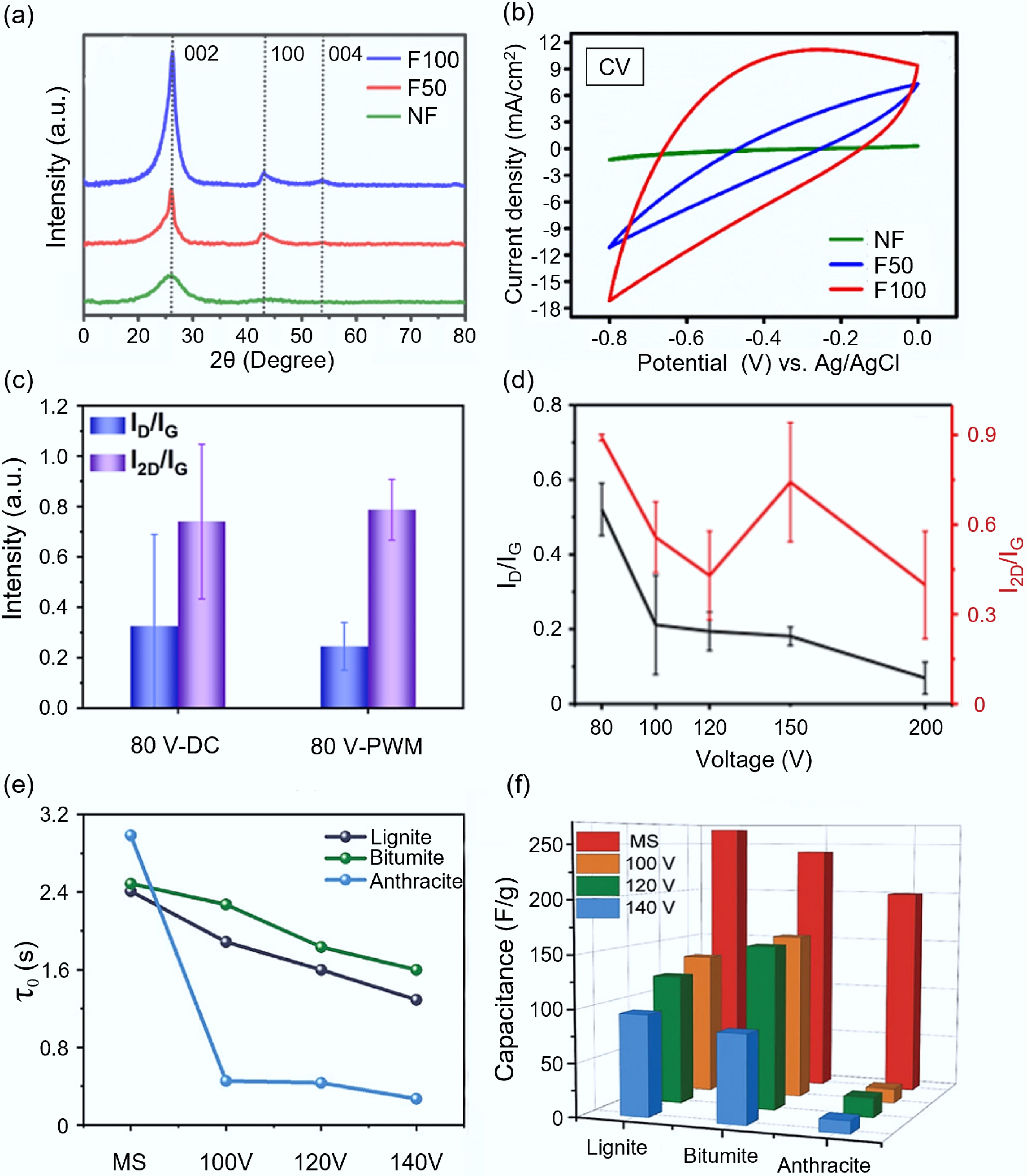
Figure 6.
The influence of electrical parameters on the synthesis of carbon-based electrode materials by FJH. (a) XRD patterns of graphene products under different voltages[77]. (b) CV curves of NF, F50, and F100 electrodes compared at 100 mV/s[77]. (c) The influence of PWM and DC on the structure of graphite products[79]. (d) The effect of flash voltage on the structure of carbon products[72]. (e) The impact of flash voltage on the relaxation time of carbon products[44]. (f) The influence of flash voltage on the specific capacitance of carbon products[44].
During the FJH process, adjusting the flash voltage can maintain the reaction temperature and regulate the physical and chemical properties of graphene as well as the conversion pathway. Figure 6d shows the influence of flash voltage on the structure of carbon products. At medium and low voltages (0–150 V), as the voltage increases, the crystal structure of biochar changes from disordered to ordered. However, in high voltages (> 150 V), the interlayer spacing of graphene will decrease, thereby enhancing the long-range lattice order, and ultimately causing graphene to form an AB stacking structure (the values of ID/G and I2D/2G both decrease)[72]. Within the medium and low voltage range, increasing the voltage can significantly improve the electrochemical performance of carbon materials. Taking coal-based carbon precursors as an example, higher voltages are conducive to the condensation of aromatic rings and the formation of large-sized aromatic fragments[80]. As a result, the relaxation time (τ0) of the carbon material will be reduced, and the lower τ0, the higher the electrical conductivity of the electrode (as shown in Fig. 6e)[44]. At high voltages, the pore size of the carbon material expands, providing space for sodium ion battery storage. The specific capacity of the carbon material has been increased to varying degrees due to the different raw material properties (as shown in Fig. 6f). It is particularly important to note that a higher flash voltage (> 160 V) will lead to a decrease in the specific capacity value. This is because the number of defect sites and oxygen groups decreases at high temperatures[44]. The appropriate flash evaporation voltage should be determined based on the characteristics of different raw materials and the requirements of the application scenario.
Temporal parameters. Temporal parameters mainly manifest in the duration of the flash process and the number of flash times. Jia et al.[72] found that the ratio of I2D/G gradually increased with time within 30 ms, and the degree of graphitization gradually improved. It reached the maximum value around 30 ms, and adjusting the flash time could regulate the energy transfer to the sample and promote the formation of FG with the optimal planar crystallinity and interlayer disorder. After 50 ms, prolonged ultra-high temperature would promote the formation of a thermodynamically driven AB stacking structure, thereby reducing the 2D peak intensity (as shown in Fig. 7a). Stanford et al.[81] found that the flash time has different structural regulation effects on different morphological graphite products. For tFG sheets, the flash evaporation time is around 100 ms, with the lowest defect concentration and the highest graphitization quality. Beyond this time range, the 2D/G ratio decreases. This trend is similar to that of Jia et al.'s research results[72], but for wrinkled graphene prepared by plastic pyrolysis, the 2D/G ratio increases with the increase of flash evaporation time, and once complete graphitization occurs, it basically saturates after more than 100 ms, and the 2D peak intensity will not significantly decrease, and the graphitization structure is well preserved (as shown in Fig. 7b).
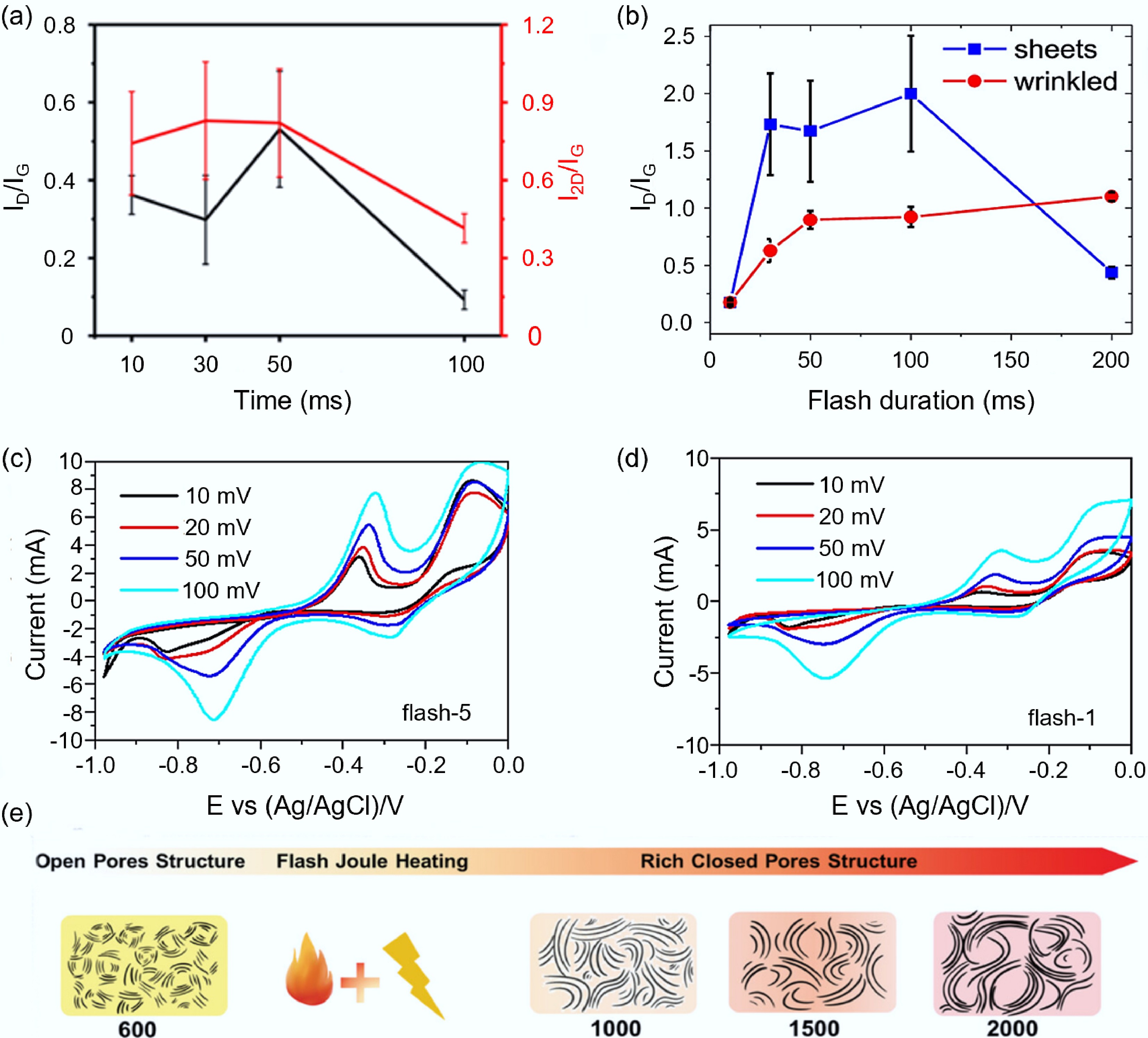
Figure 7.
Effects of temporal parameters and temperature on the synthesis of carbon-based electrode materials by FJH. (a) Influence of flash time on the structure of graphite products[72]. (b) Influence of flash time on the structural properties of tFG sheets and wrinkled graphene[81]. (c) CV curve of the carbon product after flash for only once[82]. (d) CV curve of the carbon product after flash for five times[82]. (e) The regulatory mechanism of FJH temperature on HC pores[85].
In addition to the duration of flash process, the number of flash times also has a significant impact on the carbon products. Multiple flash times can not only effectively remove impurities from the carbon products and increase the carbon content, but also adjust the graphite carbon layer and improve its electrochemical performance. Zhang et al.[82] studied and compared the effects of different flash times on graphite carbon products under the same flash voltage. The carbon content of the graphite product after five flash times increased from 92.57% to 98.11%, and the closed area of the CV curve obtained through electrochemical testing was significantly larger than that of only once, indicating that it had a higher specific capacitance (as shown in Fig. 7c, d). Cao et al.[46] conducted multiple flash evaporation tests using anthracite as the raw material (flash for one, two, and five times). On the one hand, this again confirmed that the increase in the number of flash times significantly improved the graphite orderliness of the carbon layer; on the other hand, the electrochemical performance indicated that the sample with three flash times (FA3) had an appropriate carbon layer thickness, closed pores, and a high reversible capacity (209 mAh/g), good cycle stability (no significant capacity decay after 500 cycles), and was the most ideal anode material for sodium-ion batteries, indicating that choosing an appropriate flash times based on the raw material to obtain the most suitable carbon layer thickness can achieve the best electrochemical performance.
Flash temperature. The temperature during the flash process is closely related to the regulation of pore structure. The structure of the pores directly affects the performance of the energy storage material. Small pores can provide many active sites for energy storage, while medium-sized and large pores are conducive to the transmission and storage of electrolyte ions[83]. Shen et al.[84] used FJH to perform high-temperature modification on methane pyrolyzed carbon black. When the temperature increased from 1,100 to 1,700 °C, the carbon black was restructured from a non-porous structure to a porous structure. However, when the temperature rose to 2,000 °C, the pores inside the carbon black particles began to shrink. As the temperature of the carbon black thermal treatment increased, the orderliness of the carbon black enhanced, so the sample at 2,000 °C had the most stable electrochemical performance (only dropping 18.31% at an extreme current load of 20 A/g), but its surface pores closed, resulting in a lower specific capacitance (7.65 F/g) (less than 11.34 F/g of the sample at 1,700 °C). Qiu et al.[85] developed a closed-pore regulation strategy based on preheating treatment and FJH for biomass-derived hard carbon. This significantly improved the performance of the hard carbon anode material (up to 93.3% initial Coulombic efficiency and 377 mAh/g reversible capacity). The preheating treatment constructed an ultra-microporous structure, converting cellulose into a high-carbonization-capable framework, thereby increasing the final yield of biomass-derived hard carbon. On this basis, FJH treatment rapidly generated abundant closed-pore structures, and by expanding the interlayer spacing, it provided efficient transmission channels for sodium ions (as shown in Fig. 8e).
Reactor design
-
Besides modifying process condition parameters, the adaptability to different raw materials and the flexible adjustment of various working conditions can be achieved through the improvement of the experimental device. The following mainly introduces the influence of the experimental device design from the perspectives of optimizing heat and mass transfer and the reaction process.
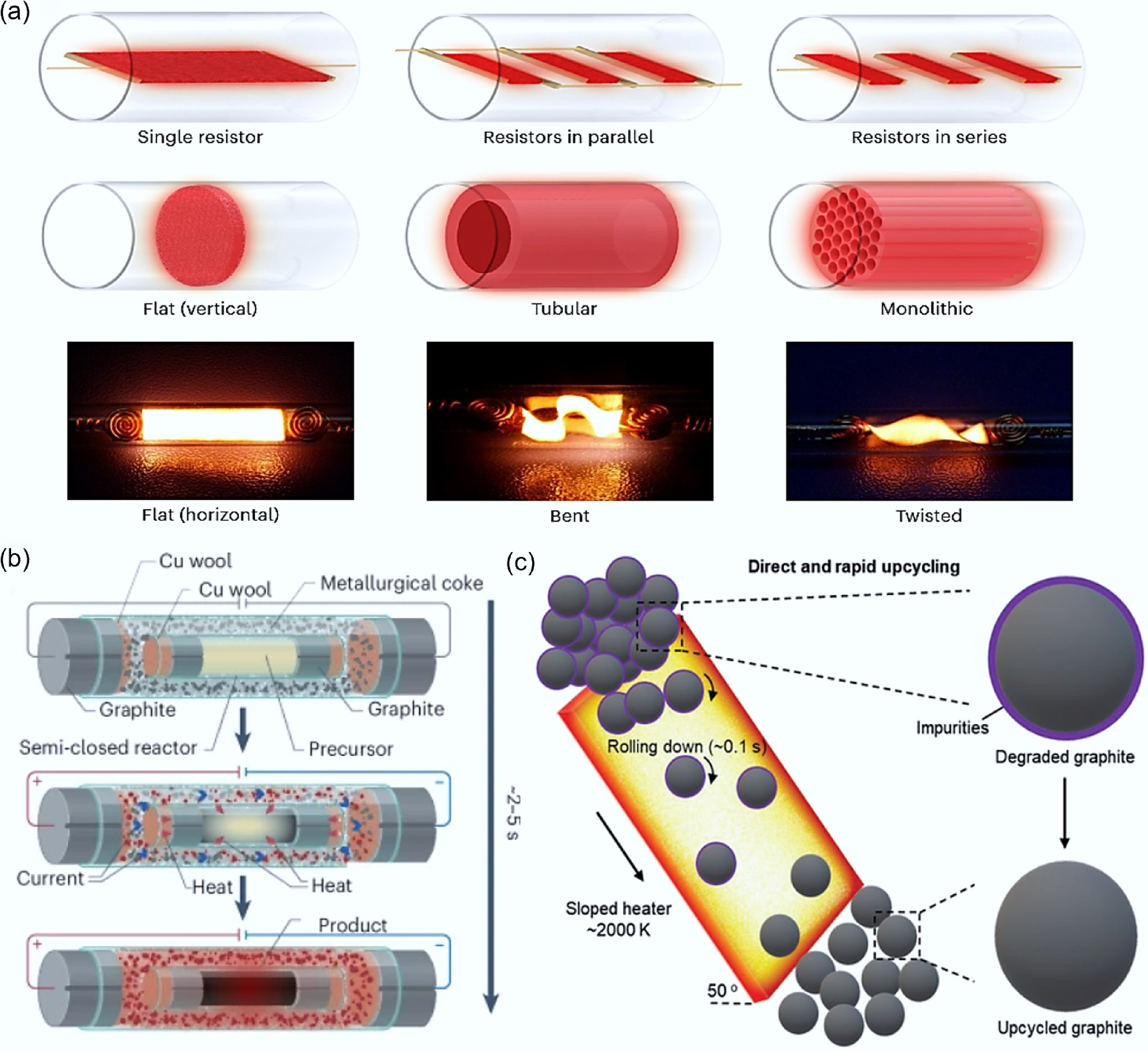
Optimize heat and mass transfer. The FJH process relies on electrothermal conversion to achieve energy supply. The heater serves as the core component and plays a significant role in heat and mass transfer during the FJH process. Table 2 shows different types of heaters' advantages, drawbacks and their applicable scene. A single plate model (as shown in Fig. 8a) with the heater can achieve a simple FJH process. Such simple heater models are easy to operate and suitable for rapid characterization of reaction results. Based on these simple heating elements, they can be combined and integrated to achieve concentrated heat supply and meet the needs of complex reactions[30]. For example, a series type can achieve larger heat supply and uniform temperature distribution, suitable for high resistivity materials, but one must be cautious of the risk of overheating; a parallel type has low requirements for power supply voltage, each parallel branch equipped with an independent switch or controller, enabling separate start/stop or energy regulation for different branches, suitable for parallel processing of multiple samples or studying the effects of different parameters, but the branch currents may not be uniform, resulting in unstable heat transfer and uneven temperature distribution[86]. Additionally, other structural designs, such as cylindrical and tubular heaters (as shown in Fig. 8a), can be fabricated by stretching the sheet material and adding holes and channels[87]. By assembling multiple cylindrical or tubular heaters in an array or honeycomb structure, the heat transfer area can be increased, promoting convective heat transfer, and the honeycomb structure can fully contact the reactants, thereby improving the reaction rate, and by embedding catalysts in the porous structure, the reaction efficiency can be further enhanced. These heating elements are currently mainly limited by the complexity of processing technology and design cost; by bending or twisting planar carbon paper heaters (as shown in Fig. 8a), the thermal conductivity efficiency can be effectively improved, suitable for high-energy consumption reactions, but this structure poses risks of local overheating and potential damage to product morphology[87,88].
Table 2. Comparison of heat transfer and mass transfer effects of different types of heaters
Heater design Advantages Drawbacks Applicable scene Single-panel Easy to set up and with quick heat
responseInsufficient heat supply Rapid characterization of the reaction results Series Greater heat supply and uniform temperature distribution High requirements for component consistency and the risk of overheating Reactions with high requirements for temperature conditions Parallel The power supply voltage requirement is low and the components are independent of each other. The heat transfer is unstable and the temperature field is unevenly distributed. Parallel processing of multiple samples or investigation of the effects of different parameters Monolithic High heat transfer efficiency and flexible catalyst loading The complexity of the processing technology and the design cost Large-scale continuous production Bent/Twisted High heat conduction efficiency The risk of local overheating Reactions with high requirements for temperature conditions Adaptability to raw materials and reaction environments. To meet the conductivity requirements, conductive agents are usually added (especially for plastic and rubber raw materials), which may lead to impurities in the product and a decrease in product purity. Moreover, during the resistance heating process, the introduction of volatile chalcogen elements (such as S and Se) is very difficult, limiting the application of these elements in the material. To address these issues, the 'Flash within Flash' (FWF) device was developed[89]. This device consisted of an external flash-heating container filled with metallurgical coke and a semi-closed internal reactor containing the target reagents (as shown in Fig. 8b). Unlike traditional FJH systems, FWF did not require the addition of conductive agents, reducing the formation of impurities and by-products. At the same time, this method can precisely control the synthesis parameters to produce phase-selective and single-crystal phase powders, demonstrating great flexibility in material modification and the ability to incorporate volatile heteroatoms into graphite materials for quality improvement. Generally, the regeneration of waste graphite requires a system with continuous high-power output. A multi-capacitor-based system can meet this requirement. Li et al.[90] developed a tilted carbon heating recovery process (as shown in Fig. 8c), using gravity to replace horizontal transportation. When degraded graphite rolled down along the heater, a continuous temperature of 2,000 K could remove surface impurities and improve graphitization for 0.1 s. The regenerated anode material showed a reversible capacity of 320 mAh/g at 1 C, with a capacity retention rate of 96.0% after 500 cycles. To ensure that FJH can effectively regenerate graphite under normal air conditions, Dong et al.[62] developed a constant-pressure atmospheric rapid temperature control FJH device system. The binder, SEI components, and lithium nested between the graphite layers rapidly evaporated at high temperatures, and the cooled graphite presented an ordered layered structure (ID/G = 0.103). The graphite anode material presented excellent rate performance (350 mAh/g at 1 C) and cycle performance (99% capacity retention after 500 cycles at 1 C).
External auxiliary means for material quality improvement
-
On the basis of raw material properties influence and process control factors, further research can be conducted on external auxiliary factors (such as doping heteroatoms, incorporating composite phases, coupling other technologies, etc.) to achieve significant modification and improvement of the target products. The following section will specifically introduce the influence of these factors on the quality improvement and modification of the products.
Doping heteroatoms
-
The properties of graphene can be further adjusted by modifying its structure and atomic composition. The substitution of heteroatoms is one of the simplest methods to change the physical and chemical properties of the material[91]. Non-carbon heteroatoms, such as nitrogen, boron, fluorine, and sulfur, have atomic radii comparable to that of carbon and are often introduced into the graphene lattice as dopants[92]. Doping heteroatoms (single elements) and multi-element co-doping into the carbon matrix through FJH is currently one of the main approaches for improving the quality of electrode materials. The following part will discuss the improvement of the electrochemical performance of carbon electrode materials by doping different heteroatoms (single elements) and multi-element co-doping.
Single-element doping. Han et al.[93] reported a nitrogen-doped carbon (FNG) synthesized via FJH (its structure is shown in Fig. 9a), and the assembled symmetrical quasi-solid-state supercapacitor had a specific capacitance of 73.0 mF/cm2 and a capacitance retention rate of 91.2%, which were superior to those of the CB without nitrogen doping (57.1 mF/cm2 and 86.7%), as shown in Fig. 9b, c. Subsequent research teams also synthesized nitrogen-doped carbon nanotubes (N-CNT) using the same method, expanding the application of N-CNT in supercapacitors and zinc-air batteries (ZAB)[94]. After 10,000 cycles of assembly of the symmetrical quasi-solid-state supercapacitor, its initial capacitance remained at 83%, and from the long-term charge-discharge tests, the N-CNT-based ZAB presented excellent stability for 370 h, surpassing commercial Pt/C and most reported non-metal catalysts[95,96] (cycling period 100–200 h). Fan et al.[97] synthesized boron-doped biomass carbon materials (f-Bs-C) through FJH in an actual three-electrode flow cell and tested for 11 h, maintaining a Faraday efficiency of over 80%. James M. Tour's research team[98] used FJH to introduce fluorine atoms into the carbon layer of lithium iron phosphate batteries (LFP) to obtain fluorine-doped carbon F-LFP/C. The capacity of F-LFP/C was 118 mAh/g, and it maintained 84% of the initial capacity after 500 cycles, higher than the undoped LFP (92 mAh/g and 24% retention rate, respectively), and F-LFP/C showed the highest capacity of 96 mAh/g at 10 C, superior to commercial LFP (58 mAh/g at 10 C)[99]. Sheng et al.[100] introduced phosphorus atoms into porous carbon via FJH and synthesized carbon-phosphorus composite materials (MFH-P/C), which showed excellent electrochemical lithium storage performance, including high capacity (1,417 mAh/g under 0.2 A/g conditions), long-term stability (935 mAh/g after 800 cycles at 2 A/g), high-rate performance (730 mAh/g at 20 A/g), and full battery cycle stability (90% retention rate after 100 cycles), with its performance superior to most reported phosphorus-based materials.
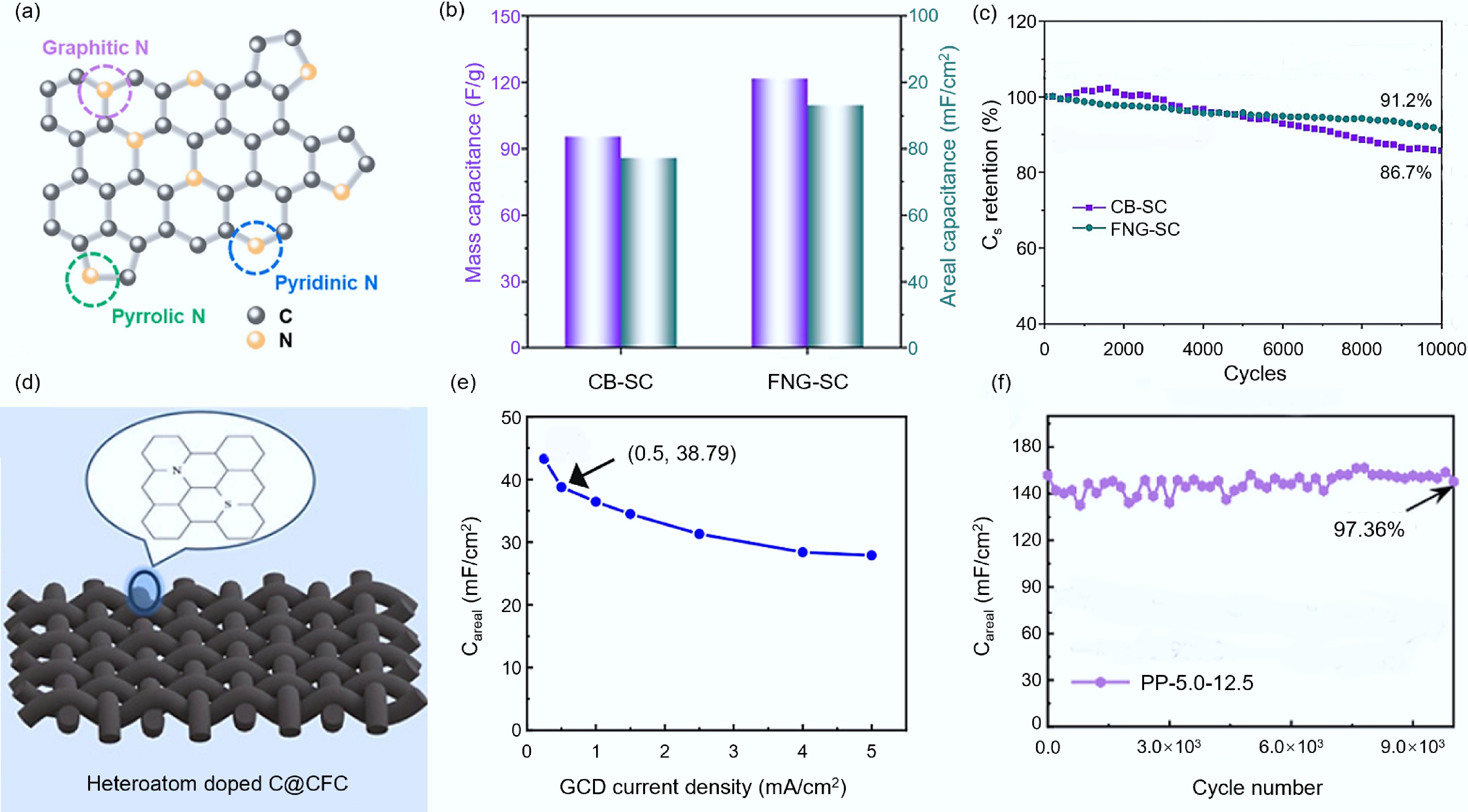
Figure 9.
Doping heteroatoms to enhance the quality of carbon materials. (a) Schematic diagram of nitrogen-doped carbon structure[93]. (b) Comparison of specific capacitance of symmetrical quasi-solid-state supercapacitors assembled by FNG and CB[93]. (c) Comparison of capacity retention rates of symmetrical quasi-solid-state supercapacitors assembled by FNG and CB[93]. (d) Simplified diagram of N-S co-doped carbon structure[101]. (e) Surface capacitance of the supercapacitor assembled with PP-5.0–12.5[101]. (f) Capacity retention rate of supercapacitors assembled with PP-5.0–12.5[101].
Multi-element doping. Heteroatoms from multiple different elements can also be simultaneously introduced into the graphene lattice, with the incorporation ratio of these elements in the graphene lattice being higher than that of single-atom substitution. After multi-element heteroatom substitution, graphene still maintains its crystal structure. However, the yield and crystallinity of graphene decrease, which may be due to the higher atomic proportion of heteroatoms introduced into the graphene lattice[91]. Multi-element doped carbon materials generally have better overall electrochemical performance than single-element doped carbon materials. Guo et al.[101] synthesized nitrogen-sulfur co-doped carbon material PP-5.0–12.5 using FJH (as shown in Fig. 9d). The assembled supercapacitor demonstrated excellent rate performance and cycling stability within a voltage range of 2.0 V. At a current density of 0.5 mA/cm2, the specific capacitance of the supercapacitor was 38.79 mF/cm2, and it still maintained 97.0% of its initial capacitance value after 10,000 cycles (as shown in Fig. 9e, f), which was superior to the supercapacitor assembled with only nitrogen-doped carbon materials (which maintained 83% of its initial capacitance after 10,000 cycles)[93]. Gao et al.[102] synthesized nitrogen-oxygen co-doped activated carbon material with through-pores (FJH-AC) via FJH. FJH-AC presented a high specific capacitance of 313.7 F/g at a current density of 1 A/g. After 10,000 cycles, the specific capacitance retention rate of the symmetric supercapacitor was 99.8%. The button cell assembled with FJH-AC achieved an energy density of 57.8 Wh/kg at a power density of 748.5 W/kg.
Combined with metals and silicon-based materials
-
Besides embedding nitrogen, boron, fluorine, and sulfur in the carbon framework in the form of atoms for modification and improvement, another method is to make metals or metal oxides and silicon-based materials exist in the carbon matrix in the form of nanoparticles, clusters, or specific compounds, thereby forming composite materials. The following will introduce the research results on the improvement and enhancement of carbon-based electrode materials based on the introduction of metals or metal oxides and silicon-based materials.
Metallic oxide. Metal oxides (such as MnOx, NiO, etc.) provide redox active sites, and when combined with carbon materials, they can alleviate the problem of low conductivity. Their high theoretical capacity[103] and pseudo-capacitive properties[104] are often used as one of the ideal electrochemically active materials. Deng et al.[104] reported the MnO2/carbon composite material synthesized by FJH (FJH-Mn-C) (as shown in Fig. 10a). For lithium-ion batteries, after testing, the FJH-Mn-C-1800 electrode performed the best at 1,800 °C (at a current density of 1 A/g, it could maintain a stable capacity of 570.9 mAh/g after 400 cycles, and the Coulombic efficiency was as high as 97.8%) (as shown in Fig. 10b). Andreu et al.[105] synthesized nickel sulfides and nickel oxide nanoparticles (Ni3S2-NiO@BP) on the carbon nanotube substrate (buckypaper, BP) via FJH, and applied them as an electrode material for high-performance flexible supercapacitors. They demonstrated an extremely high specific capacitance of 2,278 F/g at a current density of 1 A/g, and still maintained 1,080 F/g even at a high current density of 10 A/g.
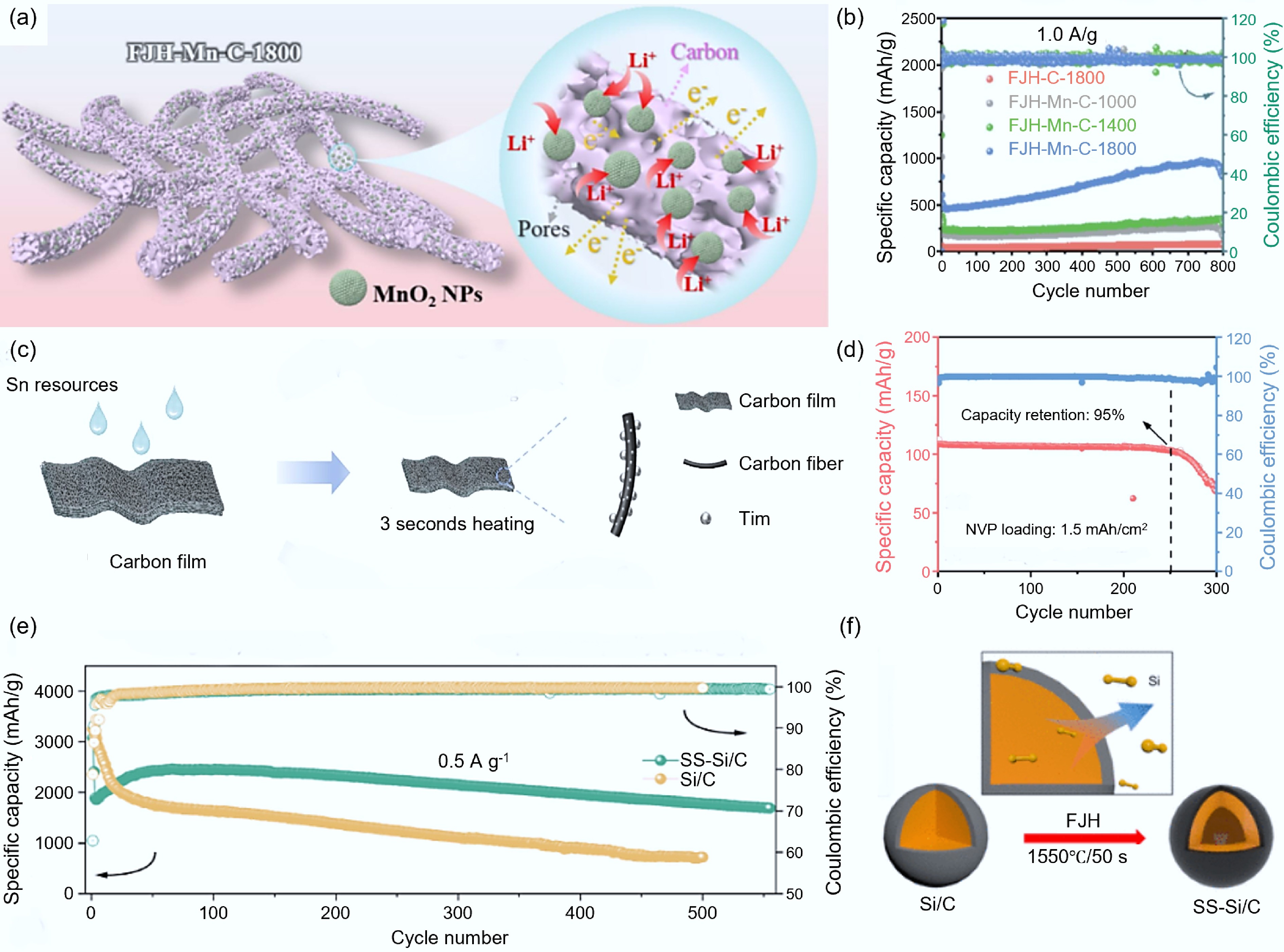
Figure 10.
Combination with metal and silicon-based materials. (a) Structural diagram of FJH-Mn-C[104]. (b) Electrochemical performance of FJH-Mn-C at different temperatures[104]. (c) Schematic diagram of the synthesis of J800-Sn/C[109]. (d) Electrochemical performance of J800-Sn/C[109]. (e) Comparison of electrochemical performance between SS-Si/C and Si/C[111]. (f) Schematic diagram of the synthesis of SS-Si/C[111].
Metal nanoparticles. The combination of metal nanoparticles (such as Pt, Sn, etc.) with carbon materials has become one of the core strategies for improving the performance of carbon electrodes. This method can promote the construction of conductive networks, the introduction of catalytically active sites, and the enhancement of stability[106,107]. Wang et al.[108] used a three-dimensional porous graphene/carbon nanotube aerogel (GCA) as the base material and successfully loaded ultrafine platinum nanoparticles (Pt NPs) on it through FJH, constructing an optimized Pt @ GCA electrode. At a current density of 20 μA/cm2, it had a high area discharge capacity (11.247 mAh/cm2) and achieved excellent energy efficiency (82%). Zeng et al.[109] proposed a modification of the carbon framework with uniform tin (Sn) nanoclusters (J800-Sn/C) through FJH synthesis (as shown in Fig. 10c). The optimized J800-Sn/C electrode presented outstanding cycling stability for over 1,000 h at a current density of 3 mA/cm2. When assembled with the J800-Sn/C @ Na anode and NPV positive electrode, the all-cell battery showed a specific capacity of 1.5 mAh/cm2 and maintained a high-capacity retention rate of 95% after 250 cycles (as shown in Fig. 10d).
Silicon-based materials. The theoretical capacity of silicon-based materials (4,200 mAh/g) is much higher than that of ordinary graphite materials (300–400 mAh/g), but it is limited by the problems such as electrode pulverization and unstable interface caused by the intense volume expansion during charging and discharging[110]. The composite modification of nano-silicon and carbon materials can partially alleviate the volume effect and improve the capacity performance of the carbon anode material. Wang et al.[111] proposed a shell-shell structure silicon-carbon (SS-Si/C) composite material based on FJH synthesis. The SS-Si/C anode exhibits excellent performance, with a specific capacity of 2.05 mAh/cm2 after 400 cycles at 0.5 mA/cm2, and a specific capacity of 1,055.6 mAh/g after 1,000 cycles at 8 A/g (as shown in Fig. 10e & f). Yang et al.[112] based on the Si/C composite material synthesized by FJH, the initial capacity was as high as 1,141.3 mAh/g, and still maintained a capacity of 894.95 mAh/g after 1,000 cycles, with a cycle capacity attenuation rate of only 0.0216%, demonstrating extremely high cycle stability and capacity retention rate.
Coupled with other technologies
-
Coupling FJH with other methods to optimize the reaction process provides a new research approach for the synthesis of high-performance carbon-based electrode materials. Han et al.[113] coupled FJH with the rapid molten salt method, obtaining porous carbon materials with a surface area of up to 1,338 m2/g, which presented excellent (284.6 F/g) specific capacity in an alkaline KOH electrolyte. The symmetrical supercapacitor assembled with TEBAF4/AN electrolyte had a specific energy of 43.9 Wh/kg at 375 W/kg and showed good cycle stability, retaining 90.1% capacity after 10,000 cycles. Wang et al.[114] used FJH combined with molten salt electrolysis of carbon dioxide to synthesize graphite materials (as shown in Fig. 11a), and the graphite anode showed excellent reversible capacity (297.7–378.1 mAh/g) and initial Coulombic efficiency (72.6%–80.5%). Chen et al.[115] coupled FJH with steam activation to synthesize high-graphitized porous carbon foam (as shown in Fig. 11b), which demonstrated a capacity of up to 516.2 mAh/g in lithium-ion battery tests and maintained 92.77% capacity after 1,100 cycles, significantly exceeding the theoretical capacity of traditional graphite. Deng et al.[116] coupled FJH with spheroidal graphite technology (as shown in Fig. 11c), achieving gram-scale production of graphene and regulating its structural properties. The cracked graphene (CFG) after spheroidal graphite treatment exhibited significantly better capacity and rate performance than traditional materials, maintaining a high capacity of 99% at the 500th cycle, and with a charging state of 77% and 62% when assembled with LiFePO4 (LFP) cathode in a 2 and 4 C condition, respectively. Luo et al.[117] introduced the assembly of FJH combined with laser-induced graphene paper-based micro-supercapacitors (LIGP-MSC) (as shown in Fig. 11d), significantly increasing the specific surface area (from 160.97 to 533.49 m2/g) and pore volume (from 0.179 to 0.553 cm3/g) of laser-induced graphene. These structural changes led to a significant improvement in the capacitance performance of LIGP-MSCs, achieving an area-specific capacitance of 13.71 mF/cm2 at a scan rate of 10 mV/s, which is six times that of unheated LIGP-MSCs. Apart from molten salt and laser-assisted processes, other synergistic effects of other electric heating methods like microwave and induction should also be considered. We had previously discussed the issues regarding the removal of adhesives and residual electrolytes for the recycling of waste graphite anodes[118]. Microwave heating, due to its strong microwave absorption capability, can assist FJH in rapidly heating the waste graphite particles, promoting the decomposition of the binders and residual electrolytes, thereby facilitating the efficient detachment from the copper foil. Fan et al.[119] utilized microwave-assisted FJH to recover and regenerate waste anode graphite. The regenerated graphite retained an ordered layered structure and restored electrochemical activity. The initial discharge specific capacity and coulombic efficiency at 0.1 C were 435.2 mAh/g and 80.6%, respectively.
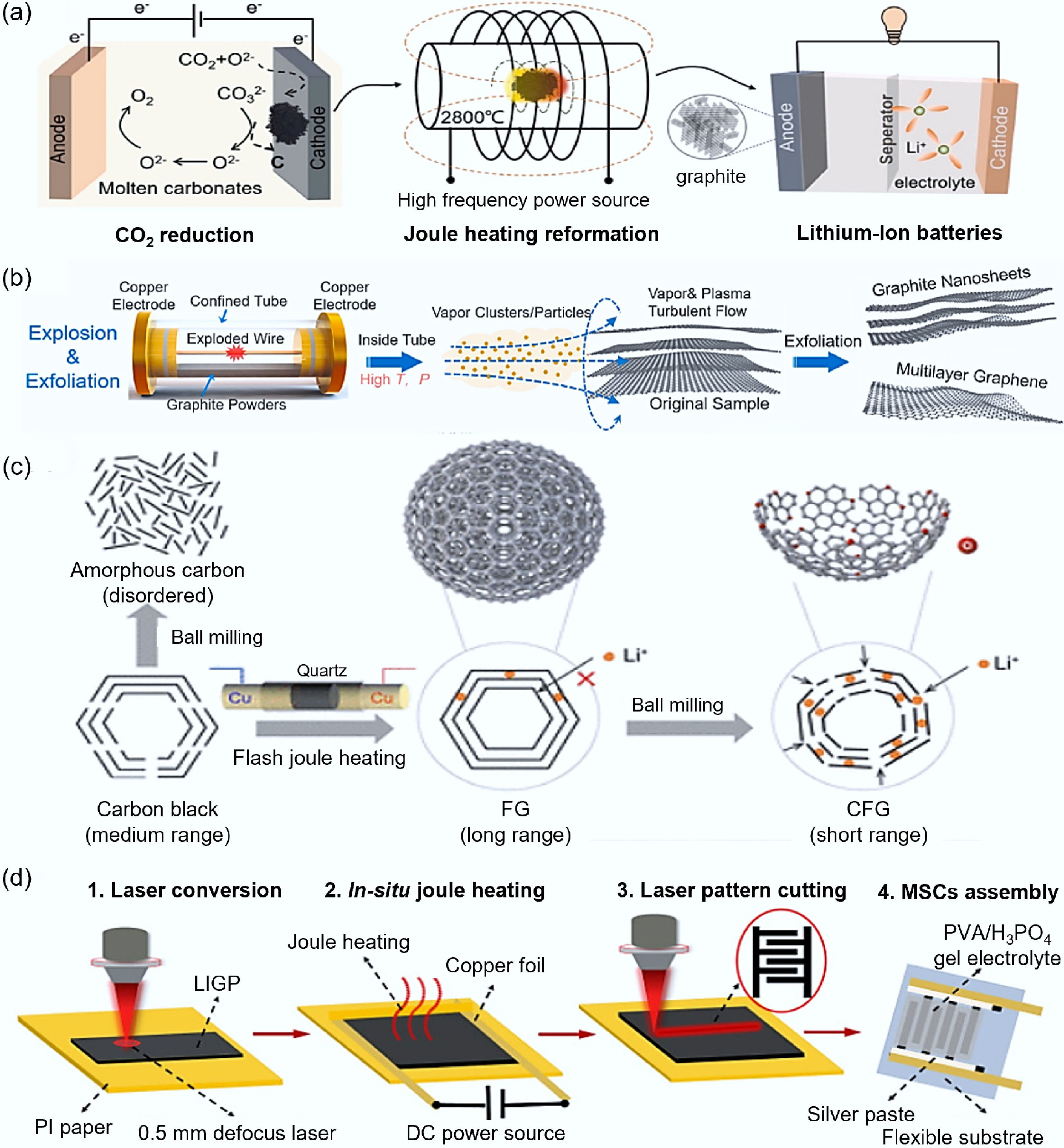
Figure 11.
Coupling other technologies with FJH. (a) Schematic diagram of FJH combined with molten salt electrolysis of carbon dioxide[114]. (b) Schematic diagram of steam activation assisting FJH[115]. (c) Schematic diagram of graphite technology assisting FJH[116]. (d) Schematic diagram of laser-induced assisting FJH[117].
-
The above passage comprehensively summarizes the research progress of synthesizing carbon-based electrode materials from various raw materials via FJH, analyzes key factors, and provides strategies for improving final products. This method is famous for its cleanliness, rapidity, efficiency, and multi-functionality, and has significant advantages in regulating the morphology and performance of carbon products. Although this method has great potential, there are still some challenges in its implementation. These challenges include that the research on the synthesis of certain functional carbon materials using FJH is still at an early stage, the simulation of the internal temperature field is used to optimize the heat and mass transfer process, large-scale industrial production, and how to combine new tools such as AI to achieve the improvement of process technology design. The following section presents several reasonable suggestions and outlooks for the existing problems and challenges:
Explore new carbon-based energy storage materials. Based on the current research achievements of FJH functionalized carbon-based energy storage materials applied in lithium batteries and supercapacitors, in the future, the application of FJH technology in the synthesis of novel carbon-based energy storage materials can be expanded. For example, by leveraging the ultra-fast high-temperature and quenching characteristics of FJH to precisely construct new carbon materials with specific dimensions (such as two-dimensional heterojunctions, three-dimensional hierarchical porous networks) and specific functions (such as high-entropy carbon, topological defect carbon); exploring their applications in cutting-edge systems such as alloy anode composites (such as Si, Sn, Sb), sulfur host materials, and solid-state battery electrolyte frameworks. Through the coupling of the targeted design of precursor chemical structures with the FJH process, it is expected to achieve the controllable synthesis of metastable and non-equilibrium carbon materials that are difficult to synthesize by traditional methods, providing new electrode materials for next-generation high-performance lithium-ion batteries, lithium-sulfur batteries, sodium/kalium batteries, etc.
Strengthen research on mechanisms. At present, FJH technology has achieved remarkable results in non-equilibrium synthesis and the regulation of carbon product defects. However, due to the complexity of the FJH process, which involves strong coupling and transient changes in multiple physical fields such as electricity, heat, and force, there is currently a lack of direct experimental evidence and a unified theoretical model for how these fields interact to cause the breaking and reformation of chemical bonds, as well as whether there are non-thermal effects of the electric field. Therefore, it is necessary to vigorously develop cross-scale simulation and in-situ/operational characterization techniques, integrating multi-field modeling, molecular dynamics simulation, in-situ electron microscopy, and synchrotron radiation, to reveal the nucleation, growth, and phase transformation laws of carbon materials driven by the coupling of 'electric-thermal-force' at the atomic scale[120,121]. At the same time, efforts should be made to analyze the behavioral differences of different raw materials (especially complex component waste materials) in the FJH process, clarify the migration paths of impurities and the formation mechanism of product defects, and lay a theoretical foundation for achieving precise prediction and regulation from raw material characteristics to final product performance.
Industrial, large-scale, and green production. Currently, the main approaches for achieving large-scale production of carbon-based materials include equipping with automated loading and unloading systems[122], installing programmable continuous FJH output devices[123], using the R2R-FJH process to synthesize large-area graphene[124,125], combined with auxiliary arc welding machines to provide better energy transmission capabilities[126], etc. Although these measures have enabled a leap in the production of carbon-based products from gram-level per hour to ton-level per hour, compared with conventional large-scale pyrolysis, the scale of FJH synthesis is still insufficient to meet industrial production requirements. To achieve its industrial-scale application, a series of key issues, such as equipment design, process optimization, and cost control, needs to be addressed. The application of continuous flow reactors in FJH synthesis can be expanded, adopting a modular design concept to facilitate the expansion and upgrade of the production line and meet the production capacity requirements of different scales. At the system level, the synergy between FJH and other electric heating modes such as microwave[127], induction[128], laser[129], and plasma[130] should be explored, and a waste heat recovery system should be integrated to further reduce energy consumption. Currently, for the production of each kilogram of graphene, the traditional method requires an energy consumption of 3,292.2 MJ, while FJH only requires 261.9 MJ[131]. The traditional method generates 102.133 kg of CO2 per kilogram, while FJH production only requires 6.32 kg of CO2 per kilogram, reducing carbon emissions by more than 10 times[132]. Based on technical and economic analysis, the cost of flash graphene is US
${\$} $ ${\$} $ Optimization via machine learning and AI. In the face of the complex process of FJH with multiple parameters and strong coupling, the potential of artificial intelligence and big data analysis should be fully exploited. By constructing a database covering raw material characteristics, process parameters, and material performance, and training machine learning models, rapid reverse design of new materials and optimal processes can be achieved, significantly reducing research and development costs and cycles[133,134]. Combined with advanced simulation and analysis technologies to deeply analyze the microscopic mechanism and guide reactor optimization, a closed-loop research paradigm of 'data-driven discovery—simulation-assisted analysis—experimental verification and optimization' is formed, accelerating the innovation and application of FJH technology in the field of high-end carbon material manufacturing.
-
This review summarizes recent advances in the synthesis of carbon-based electrode materials via FJH. It systematically compares the structural and electrochemical properties of products derived from three primary carbon sources: fossil fuels, municipal solid waste, and biomass, highlighting the critical influence of feedstock composition on material performance. Studies demonstrate that FJH enables rapid and efficient conversion of diverse carbon sources into functional electrodes, whose properties can be flexibly tailored for specific energy storage applications. The review further elucidates the key roles of electrical parameters, pulse duration, temperature control, and reactor design in tuning the microstructure (e.g., degree of graphitization, porosity) and electrochemical behavior of the resulting carbons. Effective performance-enhancement strategies—including heteroatom doping, composite formation with metal/silicon-based materials, and hybrid approaches like molten salt activation or laser assistance—are also summarized. Despite its promise for green and rapid synthesis of high-performance electrodes, FJH still faces challenges in mechanistic understanding and scalable production. Future efforts should focus on elucidating the multi-physics coupling mechanisms, developing continuous and intelligent reactor systems, and expanding applications in next-generation energy storage systems to facilitate the transition of FJH technology from laboratory research to industrial implementation.
The authors express their great appreciation for the support of National Natural Science Foundation of China (Grant No. 52436007), and the National Natural Science Funds for Distinguished Young Scholar (Grant No. 52125601).
-
The authors confirm their contributions to the paper as follows: Zhenyang Ling wrote the original draft and drew the figures; Jiawen Zeng revised the original draft and redesigned the figures and tables; Yichen Dong and Yingquan Chen reviewed the manuscript and provided helpful suggestions; Tong Han and Haiping Yang proposed the overall framework of the review and provided guidance and revision. All authors reviewed the results and approved the final version of the manuscript.
-
All data needed to evaluate the conclusions in the paper are present in the paper. Additional data related to this paper are available from the authors on reasonable request.
-
The authors did not receive any support from any organization for the submitted work.
-
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Ling Z, Zeng J, Dong Y, Chen Y, Han T, et al. 2026. Flash Joule heating towards carbon-based electrode materials: a review and roadmap. Energy & Environment Nexus 2: e011 doi: 10.48130/een-0026-0004
Flash Joule heating towards carbon-based electrode materials: a review and roadmap
- Received: 26 November 2025
- Revised: 27 December 2025
- Accepted: 19 January 2026
- Published online: 12 March 2026
Abstract: Flash Joule heating (FJH), as a new type of heating method, has received increasing attention due to its ultrafast heating and clean, efficient characteristics. Through this method, high-value utilization of carbon-containing raw materials can be achieved, especially for the synthesis of high-performance carbon-based electrode materials. This article provides a systematic review of FJH technology, covering its fundamental principles, process characteristics, and recent advances in synthesizing high-performance carbon-based electrode materials. The impact of various raw materials—including fossil fuels, municipal solid waste, and biomass—on the structure and electrochemical performance of the resulting products is critically analyzed. Furthermore, the regulatory effects of key process parameters (e.g., current, voltage, duration) and reactor design on material properties are examined. Strategies for performance enhancement, such as heteroatom doping, compositing with metal/silicon-based materials, and multi-technology coupling, are also discussed. Finally, the prevailing challenges in expanding material systems, elucidating reaction mechanisms, scaling up production, and implementing intelligent design are outlined, offering prospective solutions to advance the field.